-
PDF
- Split View
-
Views
-
Cite
Cite
Arie Berkovitz, Fina Eltes, Shlomit Yaari, Nathan Katz, Ilya Barr, Ami Fishman, Benjamin Bartoov, The morphological normalcy of the sperm nucleus and pregnancy rate of intracytoplasmic injection with morphologically selected sperm, Human Reproduction, Volume 20, Issue 1, January 2005, Pages 185–190, https://doi.org/10.1093/humrep/deh545
Close - Share Icon Share
Abstract
BACKGROUND: Our preceding studies have already demonstrated the advantage of intracytoplasmic morphologically selected sperm injection (IMSI) over the conventional IVF-ICSI procedure in terms of pregnancy rate. This study was undertaken to determine whether the increased pregnancy outcome was attributable to the preferred nuclear morphology of the selected spermatozoa, and not to the special sperm preparation technique modified by IMSI. METHODS: Comparison between two matched IMSI groups, i.e. negative comprising 38 cycles, where no spermatozoa with intact nuclei were available for microinjection; and positive, involving ovum microinjection by spermatozoa with strictly defined morphologically normal nuclei. RESULTS: Implantation and pregnancy rates were significantly higher, and abortion rates significantly lower, in the positive group compared with the negative group (25.0±25.9 versus 5.9±12.9%, F=15.8, P≤0.01; 52.6 versus 18.4%, χ2=9.7, P≤0.01; and 10.0 versus 57.1%, χ2=7.1, P≤0.02, respectively). CONCLUSIONS: Implantation and pregnancy by ICSI is associated with morphological nuclear normalcy of sperm. Sperm with a morphologically abnormal nucleus usually have low fertility potential, but some with certain nuclear abnormalities may still be able to produce pregnancy following ICSI.
Introduction
Sperm morphology has been recognized as the best predictor of outcome for natural fertilization (Bartoov et al., 1993, 1994), intra-uterine insemination (IUI) (Berkovitz et al., 1999) and conventional IVF (Kruger et al., 1988; Liu and Baker, 1992; Mashiach et al., 1992). Recently, increasing evidence has suggested that sperm morphology also plays a significant role in ICSI outcome (Bartoov et al., 2001, 2002, 2003; Tesarik et al., 2002; Chemes and Rawe, 2003; De Vos et al., 2003).
In a recent, prospective, feasibility study we demonstrated that fine morphological integrity of human sperm nuclei is positively associated with fertilization and pregnancy rates following IVF-ICSI. These results were obtained by the application of a new method of unstained, real-time, high magnification motile sperm organellar morphology examination (MSOME) to the leftover sperm fraction selected for microinjection in 100 random couples referred for ICSI (Bartoov et al., 2002). Accordingly, we introduced a modified IVF procedure—intracytoplasmic morphologically selected sperm injection (IMSI)—based on microinjection into retrieved oocytes of selected spermatozoa with strictly defined morphologically normal nuclei. The modified IMSI treatment did, in fact, result in significantly higher pregnancy rates, compared with conventional IVF-ICSI (Bartoov et al., 2001, 2003).
In addition to the strict morphological sperm selection, the IMSI method adapted some minor modifications in sperm preparation which were not conducted in the conventional IVF-ICSI process, i.e. use of a density gradient (Sil Select) in the routine preparation prior to selection, use of polyvinyl pyrrolidine (PVP), low temperature and a glass-bottomed dish during selection, prolonged sperm manipulation post-separation from the seminal fluid and sperm storage prior to microinjection.
Thus, one may claim that the advantage of IMSI is caused by these modified techniques, and is not necessarily related to the nuclear normality of the injected sperm cells. The aim of this study was to determine whether the increased pregnancy outcome was, indeed, attributable to the preferred nuclear morphology of the selected spermatozoa and not to the special sperm preparation technique adapted for IMSI. For this purpose, two matched IMSI groups with identical sperm preparation but different selection criteria were compared.
Materials and methods
Patients
Three hundred and nine IVF-IMSI cycles included in our database between the years 2001 and 2003 fit the following criteria: female partner younger than 40 years; three or more retrieved metaphase II ova in the present IVF-IMSI cycle; and IMSI outcome confirmed 3 months after microinjection.
In 38 (12%) of these cycles, we were unable to find any spermatozoa with strictly defined morphologically normal nuclei. Subsequently, all the oocytes within one cycle were injected by ‘second best’ morphologically evaluated sperm cells, selected according to compromised criteria (see MSOME criteria for spermatozoa selected for IMSI). In 126 (41%) of the cycles, the number of retrieved spermatozoa with preferred nuclear morphology was insufficient for all oocytes retrieved within one cycle. Thus, in these 126 cases, the ova were microinjected with spermatozoa with morphologically normal nuclei or with spermatozoa selected under compromised criteria (mixed microinjection). The fertilization rate and the percentage of top embryos at strict microinjection were significantly higher than the parallel variables at compromised microinjection (71.5±23.6 versus 56.6±36.0% and 30.14±24.2 versus 18.7±26.3, respectively, t=4.0, P≤0.01 and t=−2.7, P≤0.02, respectively). The mixed microinjection group was subdivided further into two subgroups: in 98 cycles, embryos derived from both kinds of microinjection were transferred in parallel (mixed transfer), while in the remaining 28 cases only embryos obtained from microinjection into the ova of spermatozoa with strictly defined morphologically normal nuclei were transferred. In 145 cases (47%), all the oocytes were injected with spermatozoa exhibiting the preferred nuclear morphology. Subsequently, all the transferred embryos in these cases were also obtained from microinjection of spermatozoa with strictly defined morphologically normal nuclei.
The pregnancy and abortion rates related to the mixed embryo transfer (n=98) were 26.5 and 30.7%, respectively. The pregnancy and abortion rates related to embryo transfer following the strict sperm selection (n=173) were 46.2 and 8.8%, respectively.
It should be noted that the above IMSI outcome data refer to the overall patient group from which the study group was derived and are not analysed further.
Negative group
The 38 cases where the transferred embryos were obtained from microinjection into the oocytes of the ‘second best’ morphologically evaluated sperm cells, selected according to compromised criteria (see MSOME criteria for spermatozoa selected for IMSI), were designated as the negative group. All the participants in this group were informed that only ‘second best’ morphologically evaluated sperm cells were retrievable for microinjection. Nevertheless, these patients wished to continue with the IMSI procedure. A written, informed, consent was obtained from each couple.
Positive group
A comparative study between the IVF-IMSI cycles, involving transfer of embryos derived following compromised versus strict sperm selection was conducted by matching the 38 negative cases with 38 IVF-IMSI cycles, where only embryos derived from microinjection of spermatozoa with strictly defined morphologically normal nuclei were transferred. The matching criteria were female's age and the number of previous failed ICSI attempts. The time period between the negative and positive matched cases was up to 4 months. The 38 positive matching cases and the remaining 135 cases, where embryos derived from injection of spermatozoa with strictly defined morphologically normal nuclei, were statistically similar in all examined demographic and IMSI outcome parameters.
The examined demographic parameters, i.e. male's and female's age, pregnancy expectation (the period between the first attempt to conceive and the IMSI cycle included in this study) and the numbers of previous failed ICSI and IMSI trials were statistically similar in the negative and positive study groups (Table I). Neither of the study groups included any previously reported data.
Routine preparation of sperm fraction
As previously described (Bartoov et al., 2003), the routine selection procedure of motile high density spermatozoa for IMSI was performed on the basis of a two-layer Sil Select density gradient system (FertiPro N.V. Beernem, Belgium). The obtained motile high density sperm fraction was used for further individual sperm retrieval based on MSOME.
Sperm preparation for individual retrieval based on MSOME
About 4000 motile high density sperm cells, obtained from the above fraction, were transferred to a 4 μl observation microdroplet of SPERM medium (Medi-Cult, Jyllinge, Denmark) containing PVP solution (ICSITM-100, Vitrolife, Kungsbacka, Sweden).
In order to estimate the morphological state of the sperm nucleus, the technician has to follow the motile sperm cell by moving the microscopic stage in the x, y and z directions for at least 20 s. Therefore, the PVP solution was necessary to slow the sperm speed and, thereby, to prevent the highly motile spermatozoa from disappearing from the monitor screen during measurement. To reduce PVP toxicity, the concentration of the PVP was adjusted to a minimum (range 0–8%), which still enables image analysis. The observation microdroplet was placed in a sterile, glass-bottomed dish (Willco wells BV, Amsterdam, The Netherlands) under sterile paraffin oil (OVOIL-100; Vitrolife, Kungsbacka, Sweden). The sperm cells, suspended in the observation microdroplet, were used for individual retrieval by MSOME. For this purpose, the sterile glass-bottomed dish containing the microdroplet was placed on a microscopic stage over the top of a Uplan Apo ×100 oil/1.35 objective lens previously covered by a droplet of immersion oil. In this way, the motile sperm cells, suspended in the observation droplet, could be examined at high magnification by the inverted microscope (Olympus IX 70, Tokyo, Japan) equipped with Nomarski differential interference contrast optics.
The success of the MSOME is associated with four image properties. (i) Optical resolution, which depends on microscope optics and on the light source of the microscope: theoretical resolution is light wavelength divided by numerical aperture of objective (in our case ∼1.5). This resolution is ∼200 nm for blue light and 300–350 nm for red light. When used with the usual white-yellow light, the resolution is ∼300 nm. (ii) Image contrast, which is increased by Nomarski optics (special phase contrasting light processing). (iii) Maximal optical magnification ∼150, which is defined by the following factors: (a) objective magnification (100×); (b) magnification selector (1.5×); and (c) video coupler magnification 0.99 (UPMTV ×0.3, PE ×3.3). (iv) Magnification of the video system, which could be calculated from the following data: (a) CCD chip diagonal dimension 8 mm; and (b) TV monitor diagonal dimension 356 mm.
Thus, the calculated video magnification (b/a) is ∼44.5 (356/8). It is, however, dependent on the size of the display. In our case, the calculated total magnification is ∼150 × 44.5 = 6675×. This number is a ratio between the size of the object on display and the real size. The actual digitally enhanced magnification, as determined by a 0.01 mm Olympus objective micrometer, was 6300×.
The digital image processing provides additional image enhancement (special filters) for detail recognition by the operator and tools for morphological measurements. This video enhancement is very important for the MSOME analysis, which requires morphological definition of the size, counter and content of the sperm nucleus.
MSOME criteria for spermatozoa selected for IMSI
As described in our previous studies, the strict descriptive criteria for normally shaped nuclei were based on those defined by scanning electron microscopy, i.e. smooth, symmetric and oval configuration (Bartoov et al., 1981, 2002, 2003). For MSOME, the average length and width of this configuration were estimated in 100 spermatozoa with an obviously normal nuclear shape and were found to be 4.75±0.28 and 3.28±0.20 μm, respectively. During sperm selection, a fixed, transparent, celluloid form of a sperm nucleus fitting the normal criteria was superimposed on the examined cell; any sperm cell with a nuclear shape was excluded from selection if it varied in length or width by 2 SDs from the normal mean axes values. According to transmission electron microscopy estimations, the nuclear chromatin content was considered normal if it contained no more than one vacuole, which occupies <4% of the nuclear area (Bartoov et al., 1981). To exclude from selection spermatozoa with abnormal chromatin content, a fixed, transparent, celluloid form of a vacuole with a borderline diameter of 0.78±0.18 μm was superimposed on the examined cell. In the cases with few small vacuoles, the estimation was made ‘by eye’. We must emphasize that spermatozoa with doubtful determination were excluded from selection.
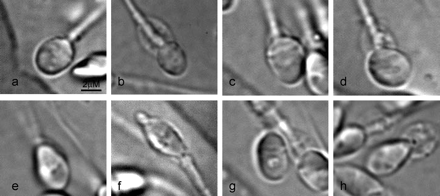
When no spermatozoa with normal nuclei were available for microinjection, the ‘second best’ morphologically evaluated sperm cells with minimally impaired nuclei were retrieved according to the hierarchy presented in Table II and Figure 1. Sperm cells exhibiting an abnormal but oval nuclear shape and a morphologically normal nuclear content were considered for the first choice selection (small or large oval nuclear forms, n=5 and n=2, respectively, Figure 1b and c, respectively). Spermatozoa with non-oval, abnormal nuclear shapes, but with normal nuclear content, were retrieved as a second choice (wide or narrow forms, n=2 and n=7, respectively, Figure 1d and e, respectively) and sperm cells with regional shape disorder (an extrusion or invagination of the nuclear mass, n=2, Figure 1f), as a third choice. Sperm cells exhibiting large nuclear vacuoles but normal oval nuclear shape (n=18, Figure 1g), were considered preferable for selection to those with a non-specific combined nuclear malformation (narrow forms + large nuclear vacuoles, n=2, Figure 1h). It should be noted that, just as in the conventional ICSI trial, sperm cells with pin, amorphous, tapered, round or multinucleated head shapes were totally excluded from retrieval.
Sperm retrieval for IVF-IMSI
Morphologically selected spermatozoa were retrieved from the observation droplet and placed into a recipient selection droplet as described earlier (Bartoov et al., 2003). On average, ∼20 sperm cells were retrieved per cycle (two spermatozoa per expected retrieved ovum). Two technicians, both of whom underwent training in sperm subcellular morphology (transmission and scanning electron microscopy), were involved in each individual sperm selection procedure. The average duration of this process was ∼2.5 h (range 1.5–5.0). During retrieval, the metabolism of the sperm cells was reduced by lowering the temperature to 21°C.
As mentioned above, in the positive group only, spermatozoa with strictly defined morphologically normal nuclei were retrieved and injected into the oocytes. In the negative group, we also tried to inject all the oocytes within one cycle with spermatozoa exhibiting exactly the same nuclear aberration, i.e. all the oocytes of each specific couple were injected with similar sperm. This condition could be accomplished since each semen sample usually exhibited typical nuclear malformations. The distribution of the seven specific aberrations for each patient is demonstrated in Table II.
Sperm storage prior to microinjection
Post-selection, the sterile glass-bottomed dish containing the recipient selection droplet was transferred from the Male Fertility Laboratory in Bar-Ilan University to the IVF centre (∼30 min away) in an insulated box at ∼25°C. The average time between completion of sperm selection and microinjection was ∼2 h (range 1–4). About half an hour before microinjection, the dish was incubated at 37°C with an atmosphere of 5% CO2.
Preparation for IVF-IMSI
The preparation for the IVF-IMSI procedure was conducted according to the method described by Van Steirteghem et al. (1993).
Microinjection
The transferred, retrieved, cumulus-free ova were placed into drops of SPERM medium prepared in the same glass dish with the recipient droplet. As mentioned above, the latter contained the sperm cells morphologically selected for IMSI. The majority of these cells located in the recipient droplet were easily found by the embryologist at the usual magnification of 200–400×. No shortage of selected spermatozoa was reported in any of the IMSI cycles included in this study. Microinjection of the selected spermatozoa into the oocytes was conducted in SPERM medium. Each microinjected oocyte was immediately transferred to a 4-well dish (Nunc), incubated in 0.5 ml of IVF or ISM 1 medium (Medi-Cult, New York, NY), covered with 0.5 ml of mineral oil (Medi-Cult) at 37°C with an atmosphere of 5% CO2.
The definitions and calculations of the fertilization rate, percentage of top embryos and implantation rate were described in detail earlier (Bartoov et al., 2003). The pregnancy rate was calculated per transfer. The abortion rate was calculated per clinical pregnancy occurrence.
Statistical analysis
All statistical analyses were performed using SPSS for Windows Version 11.0 (SPSS Inc., Chicago, IL). Data were presented as means±SD for continuous variables, including the numbers of retrieved and injected oocytes, fertilization rate, percentage of top embryos, number of transferred embryos and implantation rate. Comparisons between the positive and negative matched groups in the above variables were performed using univariate analysis of variance (ANOVA). The data for each cycle have been averaged prior to calculating the means. The discrete variables, including IMSI pregnancy and abortion rates, were presented as percentages. Comparison between the matched groups in the above variables was made by χ2 tests.
Non-parametric statistical anslyses involving Mann–Whitney tests were performed in parallel to the described statistical analyses in order to avoid possible artefacts due to the high standard deviations of some sperm parameters. The non-parametric methods revealed results similar to those reported here.
Results
The total values of the IVF-IMSI outcome parameters obtained in the matched positive and negative groups are demonstrated in Table III. Comparison between these two groups, using one-way ANOVA tests, revealed that three of the six continuous variables examined, namely fertilization rate, percentage of top embryos and implantation rates, were significantly higher in the positive group than in the negative group (F=16.3, P≤0.01; F=4.9, P≤0.03; and F=15.8, P≤0.01 respectively, Table III). Of the two discrete variables, pregnancy rate was significantly higher, and abortion rate significantly lower, in the positive group compared with the negative group (χ2=9.7, P≤0.01 and χ2=7.1, P≤0.02, respectively, Table III).
Of the 38 IVF-IMSI cycles with strict sperm selection, 20 (52.6%) pregnancies were achieved. The distribution of singleton, twin and triplet pregnancies in the positive study group was 12 (60%), six (30%) and two (10%), respectively. In 11 couples, delivery has already taken place, and six pregnancies are ongoing. In two cases (10.0%), the pregnancies resulted in first trimester missed abortions and in one couple an extra-uterine pregnancy occurred.
Of the 38 IVF-IMSI cycles with compromised sperm selection included in the negative group, seven (18.4%) pregnancies were achieved (six singletons and one twin). Three of these pregnancies resulted in normal deliveries while four others resulted in a first trimester missed abortion (Table II). Of the three deliveries, two were achieved following ovum microinjection of sperm cells with large oval nuclei (choice 1, Table II) and the third occurred in a case of narrow formed sperm heads (choice 2). Three missed abortion cases occurred when ovum microinjection was conducted with spermatozoa exhibiting large nuclear vacuoles (choice 4, Table II). In another abortion case, the sperm cells exhibited a combined malformation: large vacuoles associated with narrow formed head shape (choice 5, Table II).
Discussion
In our previous study, we compared the modified IMSI procedure with classic IVF-ICSI (Bartoov et al., 2003) and demonstrated the obvious advantage of the former treatment in pregnancy outcome (Bartoov et al., 2003). In the present study, using a negative control within the IMSI group, we confirmed our previous assumption that the remarkable increase in IVF-IMSI pregnancy rate is associated with the preferred nuclear normalcy of the sperm cells injected into the ova.
Indeed, the significantly higher fertilization rate, percentage of top embryos, implantation and pregnancy rates, and the lower abortion rate obtained following strict sperm selection, compared with the compromised selection, fit our previous findings. The fact that in the excluded ‘mixed microinjection’ group (see ‘Patients’), the fertility and top embryo ratios obtained following injection of strictly selected spermatozoa were significantly higher than those obtained in cases of compromised selection has only strengthened these findings. The present results also confirm the results of studies by Tesarik et al. (2002) and De Vos et al. (2003) who reported that sperm morphological quality influences fertilization, implantation and pregnancy rates following ICSI.
Our decision to use compromised selection criteria in cases where spermatozoa with strict morphologically normal nuclei could not be found was based on the following assumptions. First, it seems logical to suppose that spermatozoa with multiple combined (non-specific) nuclear malformations reflect extra-chromosomal damage compared with those with single specific abnormalities. Secondly, we assumed that within the category of specific morphological malformations, the existence of large vacuoles in the sperm nuclei indicates more damage to the nuclear DNA content and organization than nuclear shape or size impairment. This assumption is based on preliminary unpublished data, obtained in our laboratory, which indicate a significant negative relationship between the size of the nuclear vacuoles and chromatin stability assessed by the sperm chromatin structure assay (SCSA). Since, according to Larson et al. (2000), chromatin stability appears to influence embryonic development, we preferred, where possible, to retrieve sperm cells with a single shape/size nuclear deviation rather than those with a normal nuclear configuration, but with an impaired content. Of the specific nuclear defects with normal nuclear content, we chose to retrieve sperm cells with oval nuclear configuration (either large or small) rather than non-oval sperm cells. The results of Lee et al. (1996), who demonstrated that no increase in chromosome aberrations was found in spermatozoa with large or small heads, support our approach.
Indeed, all three pregnancies in the negative study group which resulted in normal deliveries were achieved when spermatozoa with a single specific nuclear shape malformation were injected into the ova (choices 1 and 2); two of them with large oval heads (choice 1). On the other hand, the four pregnancy cases in the negative study group which terminated in early missed abortions were achieved by microinjection of spermatozoa with large nuclear vacuoles.
The pregnancy rate of 18.4% obtained in the negative study group raises the question of sensitivity regarding the strict sperm selection criteria adapted for IMSI. However, because of the small number of IMSI cycles conducted with spermatozoa retrieved under compromised criteria and the low success rate in the negative group, we were still unable to draw any conclusions concerning the relationship between specific morphological malformations of the sperm nucleus and IMSI pregnancy outcome. Further investigation using compromised sperm selection may prove that spermatozoa with certain so-called ‘morphological nuclear abnormalities’ are still capable of inducing pregnancy following IVF-IMSI. If this is the case, then, in order to reduce the false-negative value of the sperm selection, the preliminary criteria which we have adopted in our laboratory should be re-evaluated. It seems, therefore, that at this stage of the research, the elimination process used in the compromised sperm selection does not infer that one choice is inherently better than another.
Although the advantage of the modified IMSI treatment compared with the classic ICSI approach in pregnancy outcome is quite clear, some important issues have to be considered prior to introducing this method into a routine IVF laboratory: (i) a significant increase in the time taken to complete a case; (ii) the need for additional highly trained man power; and (iii) the additional cost of upgrading for the routine inverted microscope equipment, including high resolution Nomarsky optics (∼€15 000) and a micromanipulator with two memories in space (∼€12 000). Taking into account these issues, a busy IVF centre may prefer to obtain sperm selection services from a separate andrological laboratory. Establishment of special andrological units which will serve several IVF centres seems to us the most practical way to introduce IMSI.
In conclusion, implantation and pregnancy by ICSI is associated with morphological nuclear normalcy of sperm. Sperm with a morphologically abnormal nucleus usually have low fertility potential, but some with certain nuclear abnormalities may still be able to produce pregnancy following ICSI.
The association between specific morphological nuclear abnormalities of the sperm microinjected into the oocyte and IMSI outcome should be investigated further.
Micrograph of spermatozoa obtained by the strict and compromised selection methods: (a) sperm cell with a morphologically normal nucleus; (b) small oval nuclear form; (c) large oval nuclear form; (d) wide nuclear form; (e) narrow nuclear form; (f) regional (acrosomal) nuclear shape disorder; (g) oval nuclear shape + large nuclear vacuoles; (h) abnormal (narrow) nuclear shape + large nuclear vacuoles.
Comparisons between the positive and negative study groups in demographic data
| Demographic dataa . | Study groups . | . | |
|---|---|---|---|
| . | Positive (n=38) . | Negative (n=38) . | |
| Female's age (years) | 32.3±3.3 | 33.3±4.5 | |
| Male's age (years) | 35.1±4.5 | 36.1±4.5 | |
| Pregnancy expectation (years) | 4.4±2.9 | 5.4±2.6 | |
| No. of previous ICSI cycles | 3.0±2.6 | 3.0±2.6 | |
| No. of previous IMSI cycles | 0.8±1.4 | 1.2±1.9 | |
| Demographic dataa . | Study groups . | . | |
|---|---|---|---|
| . | Positive (n=38) . | Negative (n=38) . | |
| Female's age (years) | 32.3±3.3 | 33.3±4.5 | |
| Male's age (years) | 35.1±4.5 | 36.1±4.5 | |
| Pregnancy expectation (years) | 4.4±2.9 | 5.4±2.6 | |
| No. of previous ICSI cycles | 3.0±2.6 | 3.0±2.6 | |
| No. of previous IMSI cycles | 0.8±1.4 | 1.2±1.9 | |
Values are means±SD.
Comparisons between the positive and negative study groups in demographic data
| Demographic dataa . | Study groups . | . | |
|---|---|---|---|
| . | Positive (n=38) . | Negative (n=38) . | |
| Female's age (years) | 32.3±3.3 | 33.3±4.5 | |
| Male's age (years) | 35.1±4.5 | 36.1±4.5 | |
| Pregnancy expectation (years) | 4.4±2.9 | 5.4±2.6 | |
| No. of previous ICSI cycles | 3.0±2.6 | 3.0±2.6 | |
| No. of previous IMSI cycles | 0.8±1.4 | 1.2±1.9 | |
| Demographic dataa . | Study groups . | . | |
|---|---|---|---|
| . | Positive (n=38) . | Negative (n=38) . | |
| Female's age (years) | 32.3±3.3 | 33.3±4.5 | |
| Male's age (years) | 35.1±4.5 | 36.1±4.5 | |
| Pregnancy expectation (years) | 4.4±2.9 | 5.4±2.6 | |
| No. of previous ICSI cycles | 3.0±2.6 | 3.0±2.6 | |
| No. of previous IMSI cycles | 0.8±1.4 | 1.2±1.9 | |
Values are means±SD.
Main nuclear malformations of the sperm cells retrieved for microinjection in the IMSI cycles of the negative group (n=38)
| Choice . | Specific nuclear malformations . | No. of patients . | Frequency (%) of malformation within each patient . | . | Pregnancy outcome . | . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
| . | . | . | Mean±SD . | Range . | D . | O . | A . | |||
| 1 | Large oval | 5 | 77.9±8.6 | 68–89 | 2 | 0 | 0 | |||
| 1 | Small oval | 2 | 80.0±28.3 | 60–100 | 0 | 0 | 0 | |||
| 2 | Wide forms (>3.7 μm width) | 2 | 100 | 0 | 0 | 0 | ||||
| 2 | Narrow forms (<2.9 μm width) | 7 | 83.5±15.5 | 60–100 | 1 | 0 | 0 | |||
| 3 | Regional disorder | 2 | 80.0±28.3 | 60–100 | 0 | 0 | 0 | |||
| 4 | Large vacuoles+normal shape/size | 18 | 86.1±15.4 | 63–100 | 0 | 0 | 3 | |||
| 5 | Narrow forms+large vacuoles | 2 | 70.9±15.5 | 60–82 | 0 | 0 | 1 | |||
| Choice . | Specific nuclear malformations . | No. of patients . | Frequency (%) of malformation within each patient . | . | Pregnancy outcome . | . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
| . | . | . | Mean±SD . | Range . | D . | O . | A . | |||
| 1 | Large oval | 5 | 77.9±8.6 | 68–89 | 2 | 0 | 0 | |||
| 1 | Small oval | 2 | 80.0±28.3 | 60–100 | 0 | 0 | 0 | |||
| 2 | Wide forms (>3.7 μm width) | 2 | 100 | 0 | 0 | 0 | ||||
| 2 | Narrow forms (<2.9 μm width) | 7 | 83.5±15.5 | 60–100 | 1 | 0 | 0 | |||
| 3 | Regional disorder | 2 | 80.0±28.3 | 60–100 | 0 | 0 | 0 | |||
| 4 | Large vacuoles+normal shape/size | 18 | 86.1±15.4 | 63–100 | 0 | 0 | 3 | |||
| 5 | Narrow forms+large vacuoles | 2 | 70.9±15.5 | 60–82 | 0 | 0 | 1 | |||
D = delivered; O ongoing pregnancy; A = abortion.
Main nuclear malformations of the sperm cells retrieved for microinjection in the IMSI cycles of the negative group (n=38)
| Choice . | Specific nuclear malformations . | No. of patients . | Frequency (%) of malformation within each patient . | . | Pregnancy outcome . | . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
| . | . | . | Mean±SD . | Range . | D . | O . | A . | |||
| 1 | Large oval | 5 | 77.9±8.6 | 68–89 | 2 | 0 | 0 | |||
| 1 | Small oval | 2 | 80.0±28.3 | 60–100 | 0 | 0 | 0 | |||
| 2 | Wide forms (>3.7 μm width) | 2 | 100 | 0 | 0 | 0 | ||||
| 2 | Narrow forms (<2.9 μm width) | 7 | 83.5±15.5 | 60–100 | 1 | 0 | 0 | |||
| 3 | Regional disorder | 2 | 80.0±28.3 | 60–100 | 0 | 0 | 0 | |||
| 4 | Large vacuoles+normal shape/size | 18 | 86.1±15.4 | 63–100 | 0 | 0 | 3 | |||
| 5 | Narrow forms+large vacuoles | 2 | 70.9±15.5 | 60–82 | 0 | 0 | 1 | |||
| Choice . | Specific nuclear malformations . | No. of patients . | Frequency (%) of malformation within each patient . | . | Pregnancy outcome . | . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
| . | . | . | Mean±SD . | Range . | D . | O . | A . | |||
| 1 | Large oval | 5 | 77.9±8.6 | 68–89 | 2 | 0 | 0 | |||
| 1 | Small oval | 2 | 80.0±28.3 | 60–100 | 0 | 0 | 0 | |||
| 2 | Wide forms (>3.7 μm width) | 2 | 100 | 0 | 0 | 0 | ||||
| 2 | Narrow forms (<2.9 μm width) | 7 | 83.5±15.5 | 60–100 | 1 | 0 | 0 | |||
| 3 | Regional disorder | 2 | 80.0±28.3 | 60–100 | 0 | 0 | 0 | |||
| 4 | Large vacuoles+normal shape/size | 18 | 86.1±15.4 | 63–100 | 0 | 0 | 3 | |||
| 5 | Narrow forms+large vacuoles | 2 | 70.9±15.5 | 60–82 | 0 | 0 | 1 | |||
D = delivered; O ongoing pregnancy; A = abortion.
Comparisons between the positive and negative study groups in IVF- IMSI outcome parameters
| . | Study groups . | . | ||
|---|---|---|---|---|
| . | Positive (n= 38) per cyclea (total) . | Negative (n=38) per cyclea (total) . | ||
| Continuous variables | ||||
| No. of retrieved ova | 13.3±5.7 (506) | 13.1±6.8 (488) | ||
| No. of injected ova | 10.1±4.5 (382) | 9.2±4.0 (334) | ||
| Fertilization rate (%) | 71.3±20.8 (264) | 50.3±24.1b (174) | ||
| Percentage of top embryos (%) | 34.9±31.3 (81) | 19.4±27.1b (32) | ||
| No. of embryos transferred | 3.3±1.2 (125) | 3.5±1.4 (113) | ||
| Implantation rate (%) | 25.0±25.9 (31) | 5.9±12.9b (8) | ||
| Discrete variables | ||||
| Pregnancy rate per transfer (%) | 52.6 (20) | 18.4b (7) | ||
| Abortion rate (%) | 10.0 (2) | 57.1b (4) | ||
| . | Study groups . | . | ||
|---|---|---|---|---|
| . | Positive (n= 38) per cyclea (total) . | Negative (n=38) per cyclea (total) . | ||
| Continuous variables | ||||
| No. of retrieved ova | 13.3±5.7 (506) | 13.1±6.8 (488) | ||
| No. of injected ova | 10.1±4.5 (382) | 9.2±4.0 (334) | ||
| Fertilization rate (%) | 71.3±20.8 (264) | 50.3±24.1b (174) | ||
| Percentage of top embryos (%) | 34.9±31.3 (81) | 19.4±27.1b (32) | ||
| No. of embryos transferred | 3.3±1.2 (125) | 3.5±1.4 (113) | ||
| Implantation rate (%) | 25.0±25.9 (31) | 5.9±12.9b (8) | ||
| Discrete variables | ||||
| Pregnancy rate per transfer (%) | 52.6 (20) | 18.4b (7) | ||
| Abortion rate (%) | 10.0 (2) | 57.1b (4) | ||
Values are means±SD per cycle.
Significantly different from the positive group (P≤0.05).
Comparisons between the positive and negative study groups in IVF- IMSI outcome parameters
| . | Study groups . | . | ||
|---|---|---|---|---|
| . | Positive (n= 38) per cyclea (total) . | Negative (n=38) per cyclea (total) . | ||
| Continuous variables | ||||
| No. of retrieved ova | 13.3±5.7 (506) | 13.1±6.8 (488) | ||
| No. of injected ova | 10.1±4.5 (382) | 9.2±4.0 (334) | ||
| Fertilization rate (%) | 71.3±20.8 (264) | 50.3±24.1b (174) | ||
| Percentage of top embryos (%) | 34.9±31.3 (81) | 19.4±27.1b (32) | ||
| No. of embryos transferred | 3.3±1.2 (125) | 3.5±1.4 (113) | ||
| Implantation rate (%) | 25.0±25.9 (31) | 5.9±12.9b (8) | ||
| Discrete variables | ||||
| Pregnancy rate per transfer (%) | 52.6 (20) | 18.4b (7) | ||
| Abortion rate (%) | 10.0 (2) | 57.1b (4) | ||
| . | Study groups . | . | ||
|---|---|---|---|---|
| . | Positive (n= 38) per cyclea (total) . | Negative (n=38) per cyclea (total) . | ||
| Continuous variables | ||||
| No. of retrieved ova | 13.3±5.7 (506) | 13.1±6.8 (488) | ||
| No. of injected ova | 10.1±4.5 (382) | 9.2±4.0 (334) | ||
| Fertilization rate (%) | 71.3±20.8 (264) | 50.3±24.1b (174) | ||
| Percentage of top embryos (%) | 34.9±31.3 (81) | 19.4±27.1b (32) | ||
| No. of embryos transferred | 3.3±1.2 (125) | 3.5±1.4 (113) | ||
| Implantation rate (%) | 25.0±25.9 (31) | 5.9±12.9b (8) | ||
| Discrete variables | ||||
| Pregnancy rate per transfer (%) | 52.6 (20) | 18.4b (7) | ||
| Abortion rate (%) | 10.0 (2) | 57.1b (4) | ||
Values are means±SD per cycle.
Significantly different from the positive group (P≤0.05).
References
Bartoov B, Fisher J, Eltes F, Langzam J and Lunenfeld B (
Bartoov B, Eltes F, Pansky M, Lederman H, Caspi E and Soffer Y (
Bartoov B, Eltes F, Pansky M, Langzam J, Reichart M and Soffer Y (
Bartoov B, Berkovitz A and Eltes F (
Bartoov B, Berkovitz A, Eltes F, Kogosowski A, Menezo Y and Barak Y (
Bartoov B, Berkovitz A, Eltes F, Kogosovsky A, Yagoda A, Lederman H, Artzi S, Gross M and Barak Y (
Berkovitz A, Eltes F, Soffer Y, Zabludovsky N, Beyth Y, Farhi J, Levran D and Bartoov B (
Chemes EH and Rawe YV (
De Vos A, Van De Velde H, Joris H, Verheyen G, Devroey P and Van Steirteghem A (
Kruger TF, Acosta AA, Simmons KF, Swanson JR, Matta JF and Oehninger S (
Larson KL, DeJonge CJ, Barnes AM, Jost LK and Evenson DP (
Lee JD, Kamiguchi Y and Yanagimachi R (
Liu DY and Baker HW (
Mashiach R, Fisch B, Eltes F, Tadir Y, Ovadia J and Bartoov B (
Tesarik J, Mendoza C and Greco E (
Author notes
1IVF Unit, Department of Obstetrics and Gynaecology, Sapir Medical Centre, Meir Hospital, Kfar Saba, 2Male Fertility Laboratory, Faculty of Life Sciences, Bar-Ilan University, Ramat Gan, 3Bar Ilan University, Center for Infrastructures and IS, Ramat Gan and 4Fertility Medical Centre, Rishon-Le-Zion, Israel