-
PDF
- Split View
-
Views
-
Cite
Cite
Alexandros Moschovas, Paulo A. Amorim, Mariana Nold, Gloria Faerber, Mahmoud Diab, Tobias Buenger, Torsten Doenst, Percutaneous cannulation for cardiopulmonary bypass in minimally invasive surgery is associated with reduced groin complications, Interactive CardioVascular and Thoracic Surgery, Volume 25, Issue 3, September 2017, Pages 377–383, https://doi.org/10.1093/icvts/ivx140
Close - Share Icon Share
Abstract
Femoral cutdown is standard in most centres if groin cannulation is used for cardiopulmonary bypass in minimally invasive cardiac surgery (MICS). Arterial closure devices (ACDs) allow placement of larger cannulas percutaneously, but its benefit in MICS is unclear. We assessed our results with percutaneous groin cannulation using ACDs in comparison with conventional surgical access in patients undergoing MICS.
We reviewed 445 consecutive patients having undergone MICS between October 2010 and March 2015. Of those, 92 (21%) were performed with conventional surgical access to the groin vessels and 353 (79%) with the use of ACDs.
Operative risk was higher in the ACD group [logistic EuroSCORE 7.9% (SD: 8.1) vs 10.6% (SD: 12.3); P = 0.010]. The use of ACDs significantly reduced operation time [193 min (SD: 43.8) vs 173 min (SD: 47.1); P < 0.001] and hospital stay [Cutdown: median 9 days (8, 14); ACD: median 9 days (7, 12), P = 0.040] without affecting the time to full mobilization. The incidence of any complication was significantly lower in the ACD group (2.3% vs 8.7%; P = 0.007). Complications with conventional cannulation consisted of lymphatic fistulae (n = 4), wound infections (n = 2), stenosis (n = 1) and haematoma (n = 1). In the ACD group, there were local dissections (n = 2) and stenoses (n = 3). There was 1 haematoma in both groups. There were 2 vascular injuries in the ACD group (n = 2), leading to conversion to surgical access.
Percutaneous groin cannulation using ACDs for establishing cardiopulmonary bypass in minimally invasive valve surgery significantly reduces groin complications, operation time and hospital stay. However, the remaining complications are mainly of vascular nature versus wound infection and lymph fistulae with cutdown.
INTRODUCTION
Minimally invasive valve surgery is a continuously growing field. In Germany, almost 50% of all mitral valve operations are performed through a mini-thoracotomy [1]. Most of these surgical techniques include cannulation of the groin for the establishment of cardiopulmonary bypass (CPB), requiring a second incision for exposure to the femoral vessels.
For interventional and endovascular procedures, percutaneous approaches have long been used but have been limited by the size of the device entering the artery [2, 3]. Arterial closure devices (ACDs) have emerged allowing the use of larger sheaths and devices percutaneously [4], thus permitting the placement of larger cannulae, which may also be used for the percutaneous set-up of CPB. The potential advantages of the percutaneous approach, such as faster patient ambulation and higher rates of patient satisfaction [5, 6], may also apply to the surgical setting. However, the use of these devices in cardiac surgery has been limited thus far and its safety has been questioned [7, 8]. One of the major differences with the use of ACD in the surgical setting is the absence of routine fluoroscopy control in the operating room [9]. We have started to use ACDs for groin cannulation in minimally invasive cardiac surgery (MICS) in 2011 without using fluoroscopy. We here describe our results with ACDs since implementation in our department and compare the results to conventional surgical access in patients undergoing MICS during the same time period.
PATIENTS AND METHODS
The population included 445 consecutive patients operated in our department in MICS between October 2010 and March 2015. These patients include all patients with percutaneous cannulation (starting from the first use of ACD and the stepwise adoption of the technique) in parallel to conventional cutdown, up to complete adoption of ACDs in more recent years. Of these 445 cases, 92 (21%) were performed with conventional surgical access to the groin vessels and 353 (79%) with the use of ACDs. Baseline data include clinical, demographic, procedural characteristics; medications used and in-hospital outcomes.
Successful outcome was defined as achievement of haemostasis, prompt wound healing and the absence of neurological or arterial complications during the in-hospital stay. Serious complications included disturbed wound healing (infections and lymphatic fistula) and vascular complications defined as acute bleeding, vascular lesion, arterial stenosis with limb ischaemia and/or arteriovenous fistula.
Preoperative diagnostics and arterial closure devices
During the observation period, the cannulated groin was preoperatively assessed by palpation, auscultation and history taking. The records did not contain any preoperative groin imaging. There was also no intraoperative fluoroscopy. The decision to use ACDs was always made by the operating surgeon. The cannulated groin did not show a history of severe peripheral arterial disease, previous femoral surgery or stent intervention in any of the patients.
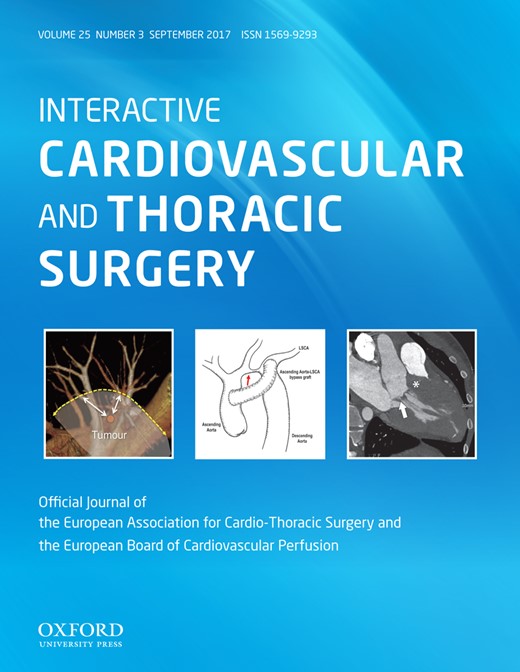
ProGlide Perclose system (Abbott Vascular) was used. The company was not involved in the decision making for ACD use or any other process relevant to the use in the patients of this report. In addition, there has been no training by the company for the use of the device. The technique was adopted from the hybrid operating room where we use ACDs routinely for transcatheter aortic valve replacement, and all devices were regularly purchased by the hospital. Figure 1 shows the principle of the ‘pre-closure’ technique. Percutaneous access is achieved by transcutaneous needle puncture of the femoral artery and vein, and guide wires are placed under echocardiographic guidance in the descending aorta and the superior vena cava. After heparin is given, 2 ProGlide systems are used. The devices are introduced into the femoral artery using the arterial wire as a guide. The wire is removed and the device is deployed at a 45° angle to the left (first device) and to the right (second device) (Fig. 1, for details see http://www.abbottvascular.com/docs/ifu/vessel_closure/eIFU_Perclose_ProGlide.pdf). Deployment consists of placing a suture through the vessel that can be tightened after the cannula has been removed at the end of the case. After both preclosure systems have been applied, a dilator with a size of 16 Fr is used, and the 20-Fr cannula is placed in the femoral artery again over the arterial guide wire. The guide wire and internal sheath are removed, and the cannula is connected to the arterial line of the CPB. After the arterial line has been secured with sutures, the venous cannula is introduced into the superior vena cava using again the Seldinger technique and echocardiographic guidance. At the end of CPB, the venous line is removed first under manual compression. After re-transfusion of remaining blood from CPB, the arterial line is removed, the ProGlide preformed knots are tightened and a pressure bandage is applied for 4–6 h postoperatively [10].
‘Pre-closure’ technique. (A) Device is rotated approximately 30° towards the patient’s right side (approximately 10 o’clock). Device is positioned at 45° angle. Procedure is repeated at 2 o’clock (B). See (http://www.abbottvascular.com/docs/ifu/vessel_closure/eIFU_Perclose_ProGlide.pdf. (reproduced with permission).
Statistical analysis
Patients were divided into 2 categories according to whether or not they were treated with ACDs during MICS. The 2 sets of patients were then compared on baseline variables, summarizing continuous variables in terms of their means and discrete variables using percentages. Independent samples t-test was conducted for the continuous data and Cox regression for the skewed censored data. Fisher’s exact and χ2 tests were performed for categorical data. Where all expected numbers are at least 1, we analysed by the ‘n−1’ χ2 test. Otherwise, we analysed using Fisher’s exact test. For the above-described statististical analyses, we used SPSS (http://www-01.ibm.com/support/docview.wss?uid=swg21476197). In the multivariable analysis, we used both binary logistic regression and penalized logistic regression to assess independent variables for complications using the statistical program R (http://cran.r-project.org/doc/FAQ/R-FAQ.html#Citing-R). Significance was set as P-value <0.05. Data are presented as mean (standard deviation) or median (interquartile range).
For both analyses, we conducted a logit transformation for the variable Logistic EuroSCORE, in order to obtain more symmetry in our statistical model [11].
In the first method and according to ‘the rule of thumb’ of 10 events per variable, several logistic regression analyses were fitted to adjust the effect of 1 stable covariate for 1 further covariate [12]. We used the femoral closure method as stable covariate. In the second method, due to the small absolute number of complications—namely 16—we chose to apply the Firth method in combination with backward selection, a penalized likelihood (bias-corrected) method that was described to exhibit superior small-sample properties [13–15]. The backward selection algorithm that we used is a trade-off between automated computer algorithms and a skilled analyst, making decisions at each step of the modelling process [16]. After univariable analysis, we started with the logistic regression including all potentially relevant variables: Femoral access method + Age + BMI + log (Logistic EuroSCORE) + Peripheral artery Disease + Stroke + Warfarin + Dialysis + Previous cardiac operation + Diabetes. The estimation procedure used was the logistf function of the R package logistf. We removed variables stepwise. Every decision for removal had 2 steps: the variable with the highest P-value can be removed (Step 1) unless the value of one of the remaining variable estimates changed greater than 15% (Step 2). When the change was greater than 15%, then the candidate variable for removal remained the confounding factor. This resulted in a final model with confounders and significant parameters. Results of logistic regression analysis are presented by odds ratios (ORs) and 95% confidence intervals.
RESULTS
Table 1 shows the preoperative characteristics of both groups. Gender distribution was equal among the groups (55% male). There was no difference between both populations with regard to ejection fraction, diabetes, hypertension, atrial fibrillation, the need for dialysis or requirement for immunosuppression or warfarin. However, there was a trend to more endocarditis and more redo operations in the ACD group (3.3% vs 9.6%, P = 0.056) and accordingly a higher preoperative risk as demonstrated by the logistic EuroSCORE [7.9% (SD: 8.1) vs 10.6% (SD: 12.3); P = 0.010]. Mortality in the groups at 30 days were 2.2% in the Cutdown group and 2.7% in the ACD group (P = 0.74). None of the patients who died had a complication at the cannulation site.
Preoperative characteristics of all patients who underwent groin cannulation for CPB either by femoral cutdown (Cutdown) or percutaneously with the use of ACDs
| . | Cutdown . | ACD . | P-value . |
|---|---|---|---|
| (n = 92) . | (n = 353) . | ||
| Male | 51 (55.4%) | 194 (55.0%) | 0.79 |
| Age (years) | 65 (SD:12) | 66 (SD:12) | 0.46 |
| Diabetes | 22 (23.9%) | 72 (20.4%) | 0.47 |
| Ejection fraction (%) | 56 (SD:14) | 55 (SD:13) | 0.46 |
| BMI (kg/m2) | 27.5 (SD:5.2) | 27.0 (SD:5.2) | 0.77 |
| Arterial hypertension | 68 (73.9%) | 273 (77.3%) | 0.58 |
| Immunosuppression | 3 (3.3%) | 20 (5.7%) | 0.59 |
| Atrial fibrillation | 35 (38.0%) | 150 (42.5%) | 0.90 |
| Peripheral artery disease | 5 (5.4%) | 31 (8.8%) | 0.39 |
| Endocarditis | 3 (3.4%) | 30 (8.5%) | 0.17 |
| Dialysis | 5 (5.4%) | 26 (7.4%) | 0.65 |
| Warfarin | 18 (19.6%) | 93 (26.3%) | 0.22 |
| Previous cardiac operation | 3 (3.3%) | 34 (9.6%) | 0.056 |
| Logistic EuroSCORE (%) | 7.9 (SD:8.1) | 10.6 (SD:12.3) | 0.010 |
| . | Cutdown . | ACD . | P-value . |
|---|---|---|---|
| (n = 92) . | (n = 353) . | ||
| Male | 51 (55.4%) | 194 (55.0%) | 0.79 |
| Age (years) | 65 (SD:12) | 66 (SD:12) | 0.46 |
| Diabetes | 22 (23.9%) | 72 (20.4%) | 0.47 |
| Ejection fraction (%) | 56 (SD:14) | 55 (SD:13) | 0.46 |
| BMI (kg/m2) | 27.5 (SD:5.2) | 27.0 (SD:5.2) | 0.77 |
| Arterial hypertension | 68 (73.9%) | 273 (77.3%) | 0.58 |
| Immunosuppression | 3 (3.3%) | 20 (5.7%) | 0.59 |
| Atrial fibrillation | 35 (38.0%) | 150 (42.5%) | 0.90 |
| Peripheral artery disease | 5 (5.4%) | 31 (8.8%) | 0.39 |
| Endocarditis | 3 (3.4%) | 30 (8.5%) | 0.17 |
| Dialysis | 5 (5.4%) | 26 (7.4%) | 0.65 |
| Warfarin | 18 (19.6%) | 93 (26.3%) | 0.22 |
| Previous cardiac operation | 3 (3.3%) | 34 (9.6%) | 0.056 |
| Logistic EuroSCORE (%) | 7.9 (SD:8.1) | 10.6 (SD:12.3) | 0.010 |
Data are presented as mean (SD) or n (%).
ACD: arterial closure device; BMI: body mass index; SD: standard deviation.
Preoperative characteristics of all patients who underwent groin cannulation for CPB either by femoral cutdown (Cutdown) or percutaneously with the use of ACDs
| . | Cutdown . | ACD . | P-value . |
|---|---|---|---|
| (n = 92) . | (n = 353) . | ||
| Male | 51 (55.4%) | 194 (55.0%) | 0.79 |
| Age (years) | 65 (SD:12) | 66 (SD:12) | 0.46 |
| Diabetes | 22 (23.9%) | 72 (20.4%) | 0.47 |
| Ejection fraction (%) | 56 (SD:14) | 55 (SD:13) | 0.46 |
| BMI (kg/m2) | 27.5 (SD:5.2) | 27.0 (SD:5.2) | 0.77 |
| Arterial hypertension | 68 (73.9%) | 273 (77.3%) | 0.58 |
| Immunosuppression | 3 (3.3%) | 20 (5.7%) | 0.59 |
| Atrial fibrillation | 35 (38.0%) | 150 (42.5%) | 0.90 |
| Peripheral artery disease | 5 (5.4%) | 31 (8.8%) | 0.39 |
| Endocarditis | 3 (3.4%) | 30 (8.5%) | 0.17 |
| Dialysis | 5 (5.4%) | 26 (7.4%) | 0.65 |
| Warfarin | 18 (19.6%) | 93 (26.3%) | 0.22 |
| Previous cardiac operation | 3 (3.3%) | 34 (9.6%) | 0.056 |
| Logistic EuroSCORE (%) | 7.9 (SD:8.1) | 10.6 (SD:12.3) | 0.010 |
| . | Cutdown . | ACD . | P-value . |
|---|---|---|---|
| (n = 92) . | (n = 353) . | ||
| Male | 51 (55.4%) | 194 (55.0%) | 0.79 |
| Age (years) | 65 (SD:12) | 66 (SD:12) | 0.46 |
| Diabetes | 22 (23.9%) | 72 (20.4%) | 0.47 |
| Ejection fraction (%) | 56 (SD:14) | 55 (SD:13) | 0.46 |
| BMI (kg/m2) | 27.5 (SD:5.2) | 27.0 (SD:5.2) | 0.77 |
| Arterial hypertension | 68 (73.9%) | 273 (77.3%) | 0.58 |
| Immunosuppression | 3 (3.3%) | 20 (5.7%) | 0.59 |
| Atrial fibrillation | 35 (38.0%) | 150 (42.5%) | 0.90 |
| Peripheral artery disease | 5 (5.4%) | 31 (8.8%) | 0.39 |
| Endocarditis | 3 (3.4%) | 30 (8.5%) | 0.17 |
| Dialysis | 5 (5.4%) | 26 (7.4%) | 0.65 |
| Warfarin | 18 (19.6%) | 93 (26.3%) | 0.22 |
| Previous cardiac operation | 3 (3.3%) | 34 (9.6%) | 0.056 |
| Logistic EuroSCORE (%) | 7.9 (SD:8.1) | 10.6 (SD:12.3) | 0.010 |
Data are presented as mean (SD) or n (%).
ACD: arterial closure device; BMI: body mass index; SD: standard deviation.
Table 2 demonstrates the perioperative parameters of the 2 groups. There were more aortic valve procedures in the ACD group. This is due to the recent establishment of aortic valve surgery through a right-sided mini-thoracotomy with groin cannulation in our department. Note also that double (aortic and mitral) and triple valve surgery (minimally invasive access) was only performed in the ACD group, again due to the recent introduction of this technique. Although surgical characteristics were mainly similar among groups including bypass times and cannula sizes, operation time was significantly reduced with the use of ACDs [93 min (SD: 43.8) vs 173 min (SD: 47.1), P < 0.001]. Since minimally invasive procedures including the aortic valve are only present in the ACD group, we also analysed operation times excluding these procedures. The result still shows a significant difference between groups [Cutdown 193 min (SD: 43), ACD 174 min (SD: 47), P = 0.001].
Perioperative characteristics of all patients who underwent groin cannulation for CPB either by femoral cutdown (Cutdown) or percutaneously with use of ACDs
| . | Cutdown . | ACD . | P-value . |
|---|---|---|---|
| Single-valve operation | |||
| MV Rec | 35 (38.0%) | 131 (37.1%) | 0.90 |
| MV Rep | 9 (9.8%) | 35 (9.9%) | 1.00 |
| TV Rec | 7 (7.6%) | 38 (10.8%) | 0.44 |
| AV Rep | 0 | 42 (11.9%) | 0.001 |
| Double-valve operation | |||
| MV Rec-TV Rec | 28 (30.4%) | 66 (18.7%) | 0.010 |
| MV Rep-TV Rec | 4 (4.3%) | 24 (6.8%) | 0.48 |
| AV Rep-MV Rec | 0 | 4 (1.1%) | 0.59 |
| Triple-valve operation | |||
| AV Rep-MV Rec-TV Rec | 0 | 3 (0.8%) | 1.00 |
| Ablation | 32 (34.8%) | 135 (38.2%) | 0.63 |
| ASD closure | 28 (30.4%) | 105 (29.7%) | 0.90 |
| Bypass time (min) | 132 ± 39 | 132 ± 41 | 0.92 |
| Operation time (min) | 193 (SD:43) | 172 (SD:47) | <0.001 |
| Arterial cannulation 20 Fr | 53 (57.6%) | 171 (48.4%) | 0.13 |
| . | Cutdown . | ACD . | P-value . |
|---|---|---|---|
| Single-valve operation | |||
| MV Rec | 35 (38.0%) | 131 (37.1%) | 0.90 |
| MV Rep | 9 (9.8%) | 35 (9.9%) | 1.00 |
| TV Rec | 7 (7.6%) | 38 (10.8%) | 0.44 |
| AV Rep | 0 | 42 (11.9%) | 0.001 |
| Double-valve operation | |||
| MV Rec-TV Rec | 28 (30.4%) | 66 (18.7%) | 0.010 |
| MV Rep-TV Rec | 4 (4.3%) | 24 (6.8%) | 0.48 |
| AV Rep-MV Rec | 0 | 4 (1.1%) | 0.59 |
| Triple-valve operation | |||
| AV Rep-MV Rec-TV Rec | 0 | 3 (0.8%) | 1.00 |
| Ablation | 32 (34.8%) | 135 (38.2%) | 0.63 |
| ASD closure | 28 (30.4%) | 105 (29.7%) | 0.90 |
| Bypass time (min) | 132 ± 39 | 132 ± 41 | 0.92 |
| Operation time (min) | 193 (SD:43) | 172 (SD:47) | <0.001 |
| Arterial cannulation 20 Fr | 53 (57.6%) | 171 (48.4%) | 0.13 |
Data are presented as mean (SD) or n (%).
ACD: arterial closure device; MV: mitral valve; TV: tricuspid valve; AV: aortic valve; Rep: replacement; Rec: reconstruction; ASD: atrial septum defect.
Perioperative characteristics of all patients who underwent groin cannulation for CPB either by femoral cutdown (Cutdown) or percutaneously with use of ACDs
| . | Cutdown . | ACD . | P-value . |
|---|---|---|---|
| Single-valve operation | |||
| MV Rec | 35 (38.0%) | 131 (37.1%) | 0.90 |
| MV Rep | 9 (9.8%) | 35 (9.9%) | 1.00 |
| TV Rec | 7 (7.6%) | 38 (10.8%) | 0.44 |
| AV Rep | 0 | 42 (11.9%) | 0.001 |
| Double-valve operation | |||
| MV Rec-TV Rec | 28 (30.4%) | 66 (18.7%) | 0.010 |
| MV Rep-TV Rec | 4 (4.3%) | 24 (6.8%) | 0.48 |
| AV Rep-MV Rec | 0 | 4 (1.1%) | 0.59 |
| Triple-valve operation | |||
| AV Rep-MV Rec-TV Rec | 0 | 3 (0.8%) | 1.00 |
| Ablation | 32 (34.8%) | 135 (38.2%) | 0.63 |
| ASD closure | 28 (30.4%) | 105 (29.7%) | 0.90 |
| Bypass time (min) | 132 ± 39 | 132 ± 41 | 0.92 |
| Operation time (min) | 193 (SD:43) | 172 (SD:47) | <0.001 |
| Arterial cannulation 20 Fr | 53 (57.6%) | 171 (48.4%) | 0.13 |
| . | Cutdown . | ACD . | P-value . |
|---|---|---|---|
| Single-valve operation | |||
| MV Rec | 35 (38.0%) | 131 (37.1%) | 0.90 |
| MV Rep | 9 (9.8%) | 35 (9.9%) | 1.00 |
| TV Rec | 7 (7.6%) | 38 (10.8%) | 0.44 |
| AV Rep | 0 | 42 (11.9%) | 0.001 |
| Double-valve operation | |||
| MV Rec-TV Rec | 28 (30.4%) | 66 (18.7%) | 0.010 |
| MV Rep-TV Rec | 4 (4.3%) | 24 (6.8%) | 0.48 |
| AV Rep-MV Rec | 0 | 4 (1.1%) | 0.59 |
| Triple-valve operation | |||
| AV Rep-MV Rec-TV Rec | 0 | 3 (0.8%) | 1.00 |
| Ablation | 32 (34.8%) | 135 (38.2%) | 0.63 |
| ASD closure | 28 (30.4%) | 105 (29.7%) | 0.90 |
| Bypass time (min) | 132 ± 39 | 132 ± 41 | 0.92 |
| Operation time (min) | 193 (SD:43) | 172 (SD:47) | <0.001 |
| Arterial cannulation 20 Fr | 53 (57.6%) | 171 (48.4%) | 0.13 |
Data are presented as mean (SD) or n (%).
ACD: arterial closure device; MV: mitral valve; TV: tricuspid valve; AV: aortic valve; Rep: replacement; Rec: reconstruction; ASD: atrial septum defect.
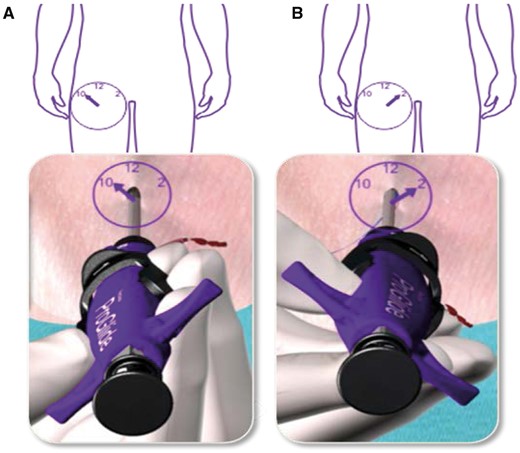
Figure 2 shows the rate of encountered complications from groin cannulation (A: for all patients, B for the time period of the first 100 ACDs, ‘learning curve’). The use of ACDs was associated with a significant reduction in overall complications in both populations (8.7% vs 2.3%, P = 0.007 and 6.2% vs 0%, P = 0.016, respectively). Note that during the first 100 cases, there were no complications in the ACD group, suggesting a minimum impact of the learning curve on patient’s outcome. The demographic data for the patients in this comparison are listed in Supplementary Material, Table S1.
Overall complication rates comparing surgical access to the groin vessel and the use of ACDs for all cases (A) and for the time period of the first 100 ACD cases (B).
When assessing the individual complications for each cannulation approach, as seen in Table 3, it is noticeable that the type of complication was different for each group. While the surgical group presented fistulae and wound infections as main complication, the use of ACDs led to more vessel injury, aneurysm and stenosis. Importantly, of the 6 patients with ACD complications, 2 were due to a cannula problem and not due to problems with the ACD. In these cases, a softer polyurethane cannula with a stiff plastic tip was used (Sorin 19 and 21 Fr), from which the plastic tip separated upon removal. The tip then remained stuck in the vessel below the fascia lata (very stiff in these 2 patients) and had to be surgically removed. ACD complications were resolved without sequalae. Vascular injuries (i.e. femoral artery stenosis and arteriovenous fistula) were resolved without sequale by conventional cutdown and repair either directly or by performing a patch reconstruction. Aneurysms were treated by ultrasound-guided fibrin injection, and haematomas could be managed conservatively. The clinical outcome was characterized by a significantly shorter hospital stay in the ACD group [Cutdown: median 9 days (8, 14); ACD: median 8 days (7, 12), P = 0.040]. There was no difference in the speed to full mobilization [Cutdown: 1.9 days (SD: 0.8); ACD: 2.0 days (SD: 1.5); P = 0.80].
List of complications in all patients who underwent groin cannulation for CPB either by femoral cutdown (Cutdown) or percutaneously with the use of ACDs
| . | Cutdown . | ACD . |
|---|---|---|
| (n = 92) . | (n = 353) . | |
| Lymphatic fistula | 4 | 0 |
| Acute bleeding | 0 | 0 |
| Access site infection | 2 | 0 |
| Haematoma | 1 | 1 |
| Femoral/iliac stenosis | 1 | 3 |
| Aneurysm/arteriovenous fistula | 0 | 2 |
| Vascular injury | 0 | 2 |
| Total | 8 (8.7%) | 8 (2.3%) |
| . | Cutdown . | ACD . |
|---|---|---|
| (n = 92) . | (n = 353) . | |
| Lymphatic fistula | 4 | 0 |
| Acute bleeding | 0 | 0 |
| Access site infection | 2 | 0 |
| Haematoma | 1 | 1 |
| Femoral/iliac stenosis | 1 | 3 |
| Aneurysm/arteriovenous fistula | 0 | 2 |
| Vascular injury | 0 | 2 |
| Total | 8 (8.7%) | 8 (2.3%) |
Data are presented as number of complications (%).
ACD: arterial closure device.
List of complications in all patients who underwent groin cannulation for CPB either by femoral cutdown (Cutdown) or percutaneously with the use of ACDs
| . | Cutdown . | ACD . |
|---|---|---|
| (n = 92) . | (n = 353) . | |
| Lymphatic fistula | 4 | 0 |
| Acute bleeding | 0 | 0 |
| Access site infection | 2 | 0 |
| Haematoma | 1 | 1 |
| Femoral/iliac stenosis | 1 | 3 |
| Aneurysm/arteriovenous fistula | 0 | 2 |
| Vascular injury | 0 | 2 |
| Total | 8 (8.7%) | 8 (2.3%) |
| . | Cutdown . | ACD . |
|---|---|---|
| (n = 92) . | (n = 353) . | |
| Lymphatic fistula | 4 | 0 |
| Acute bleeding | 0 | 0 |
| Access site infection | 2 | 0 |
| Haematoma | 1 | 1 |
| Femoral/iliac stenosis | 1 | 3 |
| Aneurysm/arteriovenous fistula | 0 | 2 |
| Vascular injury | 0 | 2 |
| Total | 8 (8.7%) | 8 (2.3%) |
Data are presented as number of complications (%).
ACD: arterial closure device.
Table 4 shows the result of the multivariable analysis. This risk adjustment revealed that percutaneous groin cannulation for CPB with the use of ACDs significantly lowers the risk of complications [P = 0.004, OR = 0.22, 95% confidence interval = −2.56 to −0.47). We used a penalized regression model, in order to fit all co-variables as possible predictors in 1 model as described in detail in Methods. Our model demonstrated 5 significant parameters (femoral access method, BMI, dialysis, previous cardiac operation and diabetes) and 1 confounder, the log (Logistic EuroSCORE). This strongly confirmed that percutaneous groin cannulation with ACD is independently associated with a reduced risk of complications (OR = 0.23, 95% confidence interval = −2.59 to −0.33). We further identified previous cardiac operations as a relevant influence.
Multivariable analysis for total groin complications with binary logistic regression and penalized logistic regression
| . | Regression coefficient . | Standard error . | Odds ratio . | Confidence interval . | P-value . |
|---|---|---|---|---|---|
| Binary regression | |||||
| Femoral access method | −1.51 | 0.52 | 0.22 | −2.56, −0.47 | 0.004 |
| Log (Logistic EuroSCORE) | 0.43 | 0.27 | 1.54 | −0.09, 0.97 | 0.11 |
| Penalized regresion | |||||
| Femoral access method | −1.46 | 0.55 | 0.23 | −2.59, −0.33 | 0.013 |
| Log (Logistic EuroSCORE) | 0.23 | 0.26 | 1.26 | −0.31, 0.78 | 0.41 |
| Dialysis | 1.12 | 0.73 | 3.06 | −0.57, 2.45 | 0.17 |
| Previous cardiac operation | 1.53 | 0.67 | 4.62 | 0.06,2.82 | 0.042 |
| Body mass index (kg/m2) | −0.07 | 0.06 | 0.93 | −0.20, 0.03 | 0.17 |
| Diabetes | 0.94 | 0.55 | 2.56 | 0.76, 7.39 | 0.12 |
| . | Regression coefficient . | Standard error . | Odds ratio . | Confidence interval . | P-value . |
|---|---|---|---|---|---|
| Binary regression | |||||
| Femoral access method | −1.51 | 0.52 | 0.22 | −2.56, −0.47 | 0.004 |
| Log (Logistic EuroSCORE) | 0.43 | 0.27 | 1.54 | −0.09, 0.97 | 0.11 |
| Penalized regresion | |||||
| Femoral access method | −1.46 | 0.55 | 0.23 | −2.59, −0.33 | 0.013 |
| Log (Logistic EuroSCORE) | 0.23 | 0.26 | 1.26 | −0.31, 0.78 | 0.41 |
| Dialysis | 1.12 | 0.73 | 3.06 | −0.57, 2.45 | 0.17 |
| Previous cardiac operation | 1.53 | 0.67 | 4.62 | 0.06,2.82 | 0.042 |
| Body mass index (kg/m2) | −0.07 | 0.06 | 0.93 | −0.20, 0.03 | 0.17 |
| Diabetes | 0.94 | 0.55 | 2.56 | 0.76, 7.39 | 0.12 |
Multivariable analysis for total groin complications with binary logistic regression and penalized logistic regression
| . | Regression coefficient . | Standard error . | Odds ratio . | Confidence interval . | P-value . |
|---|---|---|---|---|---|
| Binary regression | |||||
| Femoral access method | −1.51 | 0.52 | 0.22 | −2.56, −0.47 | 0.004 |
| Log (Logistic EuroSCORE) | 0.43 | 0.27 | 1.54 | −0.09, 0.97 | 0.11 |
| Penalized regresion | |||||
| Femoral access method | −1.46 | 0.55 | 0.23 | −2.59, −0.33 | 0.013 |
| Log (Logistic EuroSCORE) | 0.23 | 0.26 | 1.26 | −0.31, 0.78 | 0.41 |
| Dialysis | 1.12 | 0.73 | 3.06 | −0.57, 2.45 | 0.17 |
| Previous cardiac operation | 1.53 | 0.67 | 4.62 | 0.06,2.82 | 0.042 |
| Body mass index (kg/m2) | −0.07 | 0.06 | 0.93 | −0.20, 0.03 | 0.17 |
| Diabetes | 0.94 | 0.55 | 2.56 | 0.76, 7.39 | 0.12 |
| . | Regression coefficient . | Standard error . | Odds ratio . | Confidence interval . | P-value . |
|---|---|---|---|---|---|
| Binary regression | |||||
| Femoral access method | −1.51 | 0.52 | 0.22 | −2.56, −0.47 | 0.004 |
| Log (Logistic EuroSCORE) | 0.43 | 0.27 | 1.54 | −0.09, 0.97 | 0.11 |
| Penalized regresion | |||||
| Femoral access method | −1.46 | 0.55 | 0.23 | −2.59, −0.33 | 0.013 |
| Log (Logistic EuroSCORE) | 0.23 | 0.26 | 1.26 | −0.31, 0.78 | 0.41 |
| Dialysis | 1.12 | 0.73 | 3.06 | −0.57, 2.45 | 0.17 |
| Previous cardiac operation | 1.53 | 0.67 | 4.62 | 0.06,2.82 | 0.042 |
| Body mass index (kg/m2) | −0.07 | 0.06 | 0.93 | −0.20, 0.03 | 0.17 |
| Diabetes | 0.94 | 0.55 | 2.56 | 0.76, 7.39 | 0.12 |
DISCUSSION
We demonstrate, in this study, that the use of percutaneous groin cannulation using ACDs for establishing CPB in minimally invasive valve surgery is associated with a significant reduction of groin complications, operation time and hospital stay. However, the remaining complications are mainly of vascular nature versus wound infection and lymph fistulae with cutdown.
Efficacy and safety of ACD have been evaluated in a number of clinical trials in the setting of percutaneous coronary intervention [17–20], but data on its use in MICS are scarce. Because of the specifics of the procedure to achieve haemostasis, which does not allow blinding, there is also a lack of randomized clinical trials with sample sizes large enough to reveal superiority or non-inferiority of ACD compared with mechanical compression. However, in MICS, due to the larger size of the cannulae, mechanical compression alone may not be sufficient to achieve haemostasis, so surgical access is conventionally performed by cutdown and direct exposure of the vessels. Nevertheless, surgical access presents different sources of complications, demanding further improvement of the operative technique. Others have reported overall complication rates after groin cannulation for setting up CPB between 5% and 14.9% [21], which is fully consistent with the findings in our Cutdown group with a complication rate of 8.7%.
ACDs have been shown to improve major complication rates in interventional procedures compared with mechanical compression [22, 23] and are routinely used for transcatheter valve implantation where larger sheath diameters are required [24–26]. However, during these procedures, great care is usually taken to visualize the groin vessels prior to vessel access, and fluoroscopy is routinely used to guide the valve devices into the vessel as well as their removal [25]. It is important to note that in our patient population undergoing MICS (not performed in the hybrid operating room), we did not use fluoroscopy in any of our patients. Furthermore, we did not routinely image the groin vessels for neither access (percutaneous or direct access). Using this ‘blind’ approach, we achieved a significant reduction in groin complications. In addition, the complications experienced only occurred after the first 100 cases, strongly arguing against a learning curve of the procedure. Most importantly, we achieved an overall complication rate of 2.3% with the use of ACD, which is well comparable to other reports using ACDs with fluoroscopy reporting a complication rate between 2.8% and 3.6% [22, 27].
Patients undergoing MICS also require anticoagulation during and after surgery and many patients are later also on oral anticoagulation. Therefore, the observed complications may not be due to the use of bigger sized cannulae but inherent to the procedure, so cannulation poses no limitation to its use. Lee et al. [4] have achieved successful haemostasis in 94% of 292 patients undergoing percutaneous endovascular aortic repairs using 2 ProGlide devices simultaneously. Similarly, our data show a significant decrease in overall incidence of complications with the use of 2 ACDs per patient (Fig. 3).
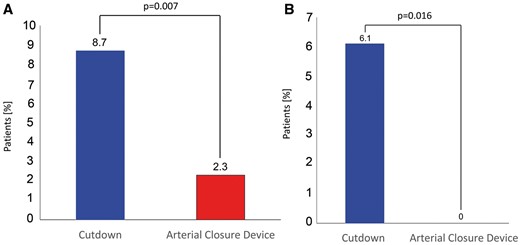
Pictures of the surgical setting after surgical access to the groin vessels (A) and transcutaneous placement of the cannulas and the use of ACDs (B) as well as their respective aesthetic result (C, D respectively). Note that in Panel D the 2 mini-cuts in the lower half of the picture compared with the percutaneous cannulation. In the upper half is the site of puncture for diagnostic cardiac catheterization.
Some studies report a decreased hospital stay with the use of ACDs after percutaneous coronary intervention [3, 6, 28]. In addition, a cost analysis performed by Rickli et al. suggested that ACDs reduced post-percutaneous coronary intervention costs by 13%, by facilitating earlier discharge, compared with manual compression [6]. We observed a difference in time to discharge that was just significant (P = 0.04) and experienced significantly shorter operation times using ACDs. We did not specifically calculate the impact for these time savings on cost-effectiveness. However, the cost of 21 min of OR time and 1 day of hospital stay should generally suffice to cover the additional cost of the 2 ProGlide devices (∼250€). It therefore appears safe to assume that the use of the devices is not associated with an increase in total cost.
We adopted the ProGlide device from the hybrid OR without further proctoring or guidance by the company. The operating team was soon familiarized with the standard application protocol. In order to address the impact of our learning curve on outcome, we analysed our first 100 cases separately. Interestingly, we observed no complications in this ACD subgroup (Fig. 3B). With this positive experience, we widened patient selection. While during the time period of the first 100 ACD cases, we used groin access in 82 patients, the time period of the next 253 ACD patients experienced only 10 cutdowns. Accordingly, comparison of the first 100 cases with the last 253 cases showed a significantly higher operative risk also evident by standard risk scores [Logistic EuroSCORE 8.6% (SD: 9.2) vs 11.9% (SD: 13.2); P = 0.042]. Nevertheless, despite experiencing complications in the ACD group in the later patients, the overall rate was still less than with cutdown in the entire patient populations.
However, an important finding has to be considered when using ACDs in this setting: the type of complication changes. While cutdown and cannulation under direct vision usually allows controlling for vessel complications (there was only 1 stenosis), wound infections, specifically in obese patients, and lymph fistulae are the dominating sources of complication. With ACDs, these complications are practically absent, but vessel complications occur. Malplacement of the device may cause stenosis upon removal of the cannula or false aneurysms may occur if the device is not placed properly or tears out. The vascular complications we had were all solved by cutdown and did not lead to any lasting sequelae. It may be argued that using vessel repairs using patches or direct (as done in our cases) may cause long-term complications. However, there is no evidence in the literature for any significant long-term detrimental effects of patch plasties or direct closures [29, 30], and some surgeons even cut the vessel open routinely for direct cannulation and repair each vessel after removal of the cannula. We therefore believe that using ACDs is safe and complications can be addressed by a cutdown, which otherwise would have been done in the first place.
Limitations
This study was limited because of its retrospective, non-randomized design and the resulted residual confounding of backward selection and probable variable overfitting. However, we have enlisted all patients operated with groin cannulation during this time, and there was no exclusion or exception made.
CONCLUSION
We conclude that percutaneous groin cannulation using ACDs for establishing CPB in minimally invasive valve surgery is associated with a significant reduction of groin complications, operation time and hospital stay. However, the remaining complications are mainly of vascular nature versus wound infection and lymph fistulae with cutdown.
SUPPLEMENTARY MATERIAL
Supplementary material is available at ICVTS online.
Conflict of interest: none declared.
REFERENCES
Author notes
The first two authors contributed equally to this work.
- cardiopulmonary bypass
- surgical procedures, minimally invasive
- cardiac surgery procedures
- hematoma
- pathologic fistula
- catheterization
- constriction, pathologic
- lymph
- surgical procedures, operative
- wound infections
- groin
- lymphatic vessels
- heart valve surgery
- european system for cardiac operative risk evaluation
- malnutrition-inflammation-cachexia syndrome