-
PDF
- Split View
-
Views
-
Cite
Cite
Tiina Vilkuna-Rautiainen, Pirkko J Pussinen, Merja Roivainen, Tuula Petäys, Pekka Jousilahti, Tapani Hovi, Erkki Vartiainen, Sirkka Asikainen, Serum antibody response to periodontal pathogens and herpes simplex virus in relation to classic risk factors of cardiovascular disease, International Journal of Epidemiology, Volume 35, Issue 6, December 2006, Pages 1486–1494, https://doi.org/10.1093/ije/dyl166
Close - Share Icon Share
Abstract
Background Increasing evidence links chronic infections, especially burden of several infections, with increased risk for cardiovascular diseases (CVD). We studied joint immune response against two major periodontal pathogens and herpes simplex virus (HSV) in relation to established risk factors of CVD.
Methods Serum antibody levels to HSV, Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis were determined by ELISA. The study included 1107 subjects, 734 from Finland and 373 from Russia.
Results Combined antibody response to periodontal pathogens was associated inversely (OR, 95% CI) with high-density lipoprotein (HDL) cholesterol concentration (β = 0.35; 0.20, 0.60; P < 0.001) and directly with HSV antibody quartiles: compared with the first quartile, ORs (95% CI) for quartiles 2–4 were 1.43 (0.88–2.32), 1.74 (1.07–2.82), and 1.89 (1.18–3.02), respectively (P for trend <0.001), after adjusting for age, gender, area, education, smoking, BMI, alcohol, triglycerides, and number of teeth. In linear regression analysis, the 3-pathogen antibody score (comprising antibody levels against periodontal pathogens and HSV) was inversely associated with HDL cholesterol concentration (β = −0.067/1 mmol/l; −0.235, −0.018; P < 0.05).
Conclusions HSV infection may promote infection by periodontal pathogens. Furthermore, the infectious burden comprising HSV and periodontitis may increase the risk for CVD by clearly decreasing HDL cholesterol concentrations.
Substantial amount of evidence links infections to cardiovascular diseases (CVD).1 Epidemiological and mechanistic studies imply that infectious agents, such as Chlamydia pneumoniae, Helicobacter pylori, herpes viruses, and periodontal pathogens, enhance atherogenesis.2–5 Pathogen burden consisting of multiple microbial species increases the risk for CVD more than any single pathogen.4,6 The relationship between infections and CVD is, however, intricate, since a variety of common underlying factors contribute to both conditions. For instance, the acute-phase response to infection is accompanied with a complex cascade leading to changes in concentrations of serum proteins and alterations in lipoprotein metabolism.7,8 Changes in lipid profiles during infection are related to the type, severity, and duration of the infection.8
Increased triglyceride and decreased high-density lipoprotein (HDL) cholesterol concentrations are the most notable unfavourable changes in the plasma lipid profile during both chronic and acute infections.7–10 High triglyceride concentration is an independent risk factor for CVD in the general population.11 It has also been shown in numerous epidemiological studies that low HDL cholesterol concentration increases the risk for CVD.12 Even though acute-phase reactions influence metabolism in all lipoprotein classes, HDL is the first lipoprotein class to be affected by infection.9 The favourable, antiatherogenic potential of HDL is owing to its role in reverse cholesterol transport and in its ability to protect low density lipoprotein (LDL) from oxidation. Additionally, HDL participates in detoxification of bacterial lipopolysaccharide (LPS) in circulation, thereby, reducing provocation of proinflammatory reactions in infections caused by gram-negative bacteria.8
Periodontitis is a common bacterial biofilm infection causing chronic inflammation in tooth-supporting tissues. In addition to the compositional shift of oral normal flora into predominance of gram-negative species in periodontitis, the number of bacteria also increases in diseased periodontal pockets.13 Thus, persistent periodontitis provides a continuous source of gram-negative bacteria and LPS, which may have access to circulation. Certain oral gram-negative species, such as Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis, are important pathogens in aggressive, as well as chronic forms of periodontitis. It has been recently suggested that herpes viruses can promote subgingival growth of periodontal pathogens, thereby, contributing to the development of periodontitis.14–16 There are no seroepidemiological studies available on the association between infections by periodontal pathogens and herpes viruses.
Discrepant results have been published on the relationship between periodontitis and CVD.17 Potential reasons include the difficulty in controlling for confounders, since periodontitis and CVD share a variety of background factors. For instance, like CVD, the prevalence of periodontitis is associated with ageing, male gender, low socioeconomic status, and smoking habit. Another complicating factor may derive from the methods used to verify periodontitis. In previous studies, periodontitis has been diagnosed by measuring signs of tissue destruction ascribed to periodontitis, such as periodontal pocket depth and loss of periodontal attachment.18,19 Little attempt has been made to determine systemic host response dedicated to periodontitis. However, a relationship between serum antibody levels to periodontal pathogens and risk factors for CVD, such as unfavourable lipid profile, was reported by us recently.20
The aims of the present study were (i) to clarify whether the same individuals were affected by periodontitis and herpes simplex virus (HSV) infections as assessed by serum IgG antibody levels against two major periodontal pathogens and HSV, and (ii) to determine the association between an aggregate infection burden formed by these pathogens and the classic CVD risk factors in two culturally and socioeconomically different study populations.
Materials and methods
Study subjects
The present study included 1107 study subjects (446 males, 661 females), 734 from North Karelia, Finland (309 males, 425 females), and 373 (137 males, 236 females) from the Republic of Karelia, Russia. The participation rates were 73.8 and 69.1% in Finland and Russia, respectively. Random population samples stratified for age and gender were obtained from the population registers. Subjects aged 25–55 years were included in the present study. The Finnish study material originates from the National FINRISK Study—survey that has been carried out at 5 year intervals to monitor risk factors of non-communicable diseases in Finland. In 1998, the same research team performed a survey in the Republic of Karelia in Russia using the same study protocol as in Finland.21
Anamnestic data and physical examination
The self-administrated questionnaire for data on the socioeconomic status, medical history, perceived health, and lifestyle was mailed to each study subject. The filled questionnaires were checked by trained study nurses at an appointment for physical examination.21 The physical examination included counting the number of natural teeth and measurements for height, weight, and blood pressure. The body mass index (BMI) was computed as weight/height2 (kg/m2). The blood pressure measurement was repeated and the mean of the two measurements was used.
Laboratory measurements
Serum total cholesterol, HDL cholesterol, and triglyceride concentrations were assayed by standard enzymatic methods. The LDL concentrations were calculated according to the Friedewald's formula. Serum carbohydrate-deficient transferrin (CDT) concentrations were determined by a double antibody kit (CDTect™; Pharmacia & Upjohn Diagnostics, Uppsala, Sweden) to estimate the alcohol consumption.22 The reference values for heavy alcohol consumption were ≥20 U/l for males and ≥26 U/l for females.
Serum IgG levels against HSV were determined with a test kit (Enzygnost® Anti HSV/IgG, Dade Behring Marburg, Germany). Before handling, the serum samples were heat-treated at 56°C for 30 min to reduce the risk of infection and subsequently diluted 1:100. Behring ELISA processor III (BEP III) was used to analyse the samples according to the manufacturer's instructions. The results are expressed as titers directly given by the processor.
Serum IgG antibody levels against two periodontal pathogens, A. actinomycetemcomitans and P. gingivalis, were determined using multiserotype-ELISAs.23 Formalin-fixed, whole bacterial cells were used as antigens. A. actinomycetemcomitans antigen mixture contained six strains representing serotypes a, b, c, d, e, and a non-serotypeable strain (ATCC 29523, ATCC 43718, ATCC 33384, IDH 781, IDH 1705, and C59A, respectively). Porphyromonas gingivalis antigen mixture comprised three strains representing serotypes a, b, and c (ATCC 33277, W50, and OMGS 434, respectively). Two dilutions (1:1500 and 1:3000 for A. actinomycetemcomitans and 1:100 and 1:200 for P. gingivalis) of each serum sample were run in duplicates. On each plate, a control for non-specific binding (no serum sample), and a high and a low control were included in duplicates to monitor the inter-assay variations. The inter-assay coefficients of variation were 6.3 and 6.1% for A. actinomycetemcomitans and P. gingivalis, respectively. The ELISA results of each plate were corrected for the mean of high control value after the whole material had been analysed. Every set of ELISA contained samples from both Finnish and Russian study populations.
Combined antibody response against periodontal pathogens was obtained by summing up individual results (the antibody level against A. actinomycetemcomitans + the antibody level against P. gingivalis). The combined antibody response was considered ‘high’, when it exceeded 14 EU, which represents the corresponding mean value +3 × SD of periodontally healthy subjects.20,23 Using this cut-off limit, the assay has a sensitivity and specificity of 71 and 90% for finding periodontitis.23 Titers below 230 EU and value <5 EU were considered seronegative for HSV and periodontal pathogens,20,23 respectively.
In order to estimate the microbial burden brought about by the two chronic infections, a designated 3-pathogen (HSV, A. actinomycetemcomitans, and P. gingivalis) antibody score was calculated by dividing the serum IgG levels against each pathogen into quartiles. The quartiles were given a value of 1, 2, 3, or 4. For each study subject, a 3-pathogen antibody score (high, intermediate, or low) was obtained by summing up the quartile values (1 through 4). Thus, each study subject received a 3-pathogen antibody score from a minimum of 3 to a maximum of 12. Values from 3 to 5 were classified as low, from 6 to 9 as intermediate, and from 10 to 12 as high.
Statistical analyses
The statistical significance of the differences in the continuous variables between the study groups was tested with the Mann–Whitney U-test. A linear by linear association test (Jonckheere-Terpstra) was used to obtain P-values for trends. The two-tailed Pearson correlation was used for correlation analyses. Multivariate logistic and linear regression analyses were used to examine the independent relations of different antibody levels with each other and with CVD risk factors studied. In the multivariate logistic and linear regression models, combined antibody response to periodontal pathogens was used as a dependent and HDL cholesterol concentration, blood pressure values, and HSV antibody quartiles as independent variables. In another model with 3-pathogen antibody score as a dependent variable, the independent variables included HDL cholesterol concentration and blood pressure values. All regression analyses were adjusted for age, gender, smoking, BMI, area (Finland or Russia), years of education, CDT, serum triglyceride concentrations, and number of teeth. The statistical analyses were carried out with SPSS 10.0 for Windows program.
Results
Characteristics and comparisons of the Finnish and Russian study populations
Table 1 shows demographic and other background characteristics of the Finnish and the Russian study populations. Except for age, gender, and BMI statistically significant (P < 0.001) differences were found between the two study populations in all variables compared. The frequency of current smokers and subjects with serum CDT concentration exceeding the reference value for heavy alcohol consumption were significantly lower in the Finnish than in the Russian population. High socioeconomic status was significantly more prevalent in the Finnish than in the Russian population.
Frequency distributions of the background variables of the Finnish and Russian study populations
| . | Whole study population (n = 1107) . | Finnish population (n = 734) . | Russian population (n = 373) . | . |
|---|---|---|---|---|
| Variable . | % (n) . | % (n) . | % (n) . | P-valuea . |
| Gender | ns | |||
| Females | 59.7 (661) | 57.9 (425) | 63.3 (236) | |
| Males | 40.7 (446) | 42.1 (309) | 36.7 (137) | |
| Age (years) | ns | |||
| 25–35 | 28.4 (314) | 26.3 (193) | 32.4 (121) | |
| 36–45 | 34.0 (376) | 35.0 (257) | 31.9 (119) | |
| 46–55 | 37.6 (417) | 38.7 (284) | 35.7 (133) | |
| Current smoker | 31.0 (343) | 25.6 (188) | 41.6 (155) | <0.001 |
| Increased CDTb | 14.8 (164) | 8.3 (61) | 27.6 (103) | <0.001 |
| Obesec | 16.3 (180) | 15.0 (110) | 18.8 (70) | ns |
| Socioeconomic status | <0.001 | |||
| Low | 21.1 (234) | 16.5 (121) | 30.0 (113) | |
| Intermediate | 59.0 (653) | 54.4 (399) | 68.1 (254) | |
| High | 19.9 (207) | 29.1 (201) | 1.6 (6) | |
| Number of natural teeth | <0.001d | |||
| Edentulous | 4.1 (46) | 6.0 (44) | 0.5 (2) | |
| 1–7 | 2.9 (32) | 3.1 (23) | 2.4 (9) | |
| 8–15 | 7.0 (78) | 5.3 (39) | 9.7 (39) | |
| 16–23 | 13.3 (147) | 4.6 (34) | 30.3 (113) | |
| 23–32 | 72.7 (755) | 81.0 (591) | 57.1 (209) |
| . | Whole study population (n = 1107) . | Finnish population (n = 734) . | Russian population (n = 373) . | . |
|---|---|---|---|---|
| Variable . | % (n) . | % (n) . | % (n) . | P-valuea . |
| Gender | ns | |||
| Females | 59.7 (661) | 57.9 (425) | 63.3 (236) | |
| Males | 40.7 (446) | 42.1 (309) | 36.7 (137) | |
| Age (years) | ns | |||
| 25–35 | 28.4 (314) | 26.3 (193) | 32.4 (121) | |
| 36–45 | 34.0 (376) | 35.0 (257) | 31.9 (119) | |
| 46–55 | 37.6 (417) | 38.7 (284) | 35.7 (133) | |
| Current smoker | 31.0 (343) | 25.6 (188) | 41.6 (155) | <0.001 |
| Increased CDTb | 14.8 (164) | 8.3 (61) | 27.6 (103) | <0.001 |
| Obesec | 16.3 (180) | 15.0 (110) | 18.8 (70) | ns |
| Socioeconomic status | <0.001 | |||
| Low | 21.1 (234) | 16.5 (121) | 30.0 (113) | |
| Intermediate | 59.0 (653) | 54.4 (399) | 68.1 (254) | |
| High | 19.9 (207) | 29.1 (201) | 1.6 (6) | |
| Number of natural teeth | <0.001d | |||
| Edentulous | 4.1 (46) | 6.0 (44) | 0.5 (2) | |
| 1–7 | 2.9 (32) | 3.1 (23) | 2.4 (9) | |
| 8–15 | 7.0 (78) | 5.3 (39) | 9.7 (39) | |
| 16–23 | 13.3 (147) | 4.6 (34) | 30.3 (113) | |
| 23–32 | 72.7 (755) | 81.0 (591) | 57.1 (209) |
Chi-square test.
CDT reference value: males ≥20 U/l, females ≥26 U/l.
BMI ≥30 kg/m2.
Mann–Whitney test.
Frequency distributions of the background variables of the Finnish and Russian study populations
| . | Whole study population (n = 1107) . | Finnish population (n = 734) . | Russian population (n = 373) . | . |
|---|---|---|---|---|
| Variable . | % (n) . | % (n) . | % (n) . | P-valuea . |
| Gender | ns | |||
| Females | 59.7 (661) | 57.9 (425) | 63.3 (236) | |
| Males | 40.7 (446) | 42.1 (309) | 36.7 (137) | |
| Age (years) | ns | |||
| 25–35 | 28.4 (314) | 26.3 (193) | 32.4 (121) | |
| 36–45 | 34.0 (376) | 35.0 (257) | 31.9 (119) | |
| 46–55 | 37.6 (417) | 38.7 (284) | 35.7 (133) | |
| Current smoker | 31.0 (343) | 25.6 (188) | 41.6 (155) | <0.001 |
| Increased CDTb | 14.8 (164) | 8.3 (61) | 27.6 (103) | <0.001 |
| Obesec | 16.3 (180) | 15.0 (110) | 18.8 (70) | ns |
| Socioeconomic status | <0.001 | |||
| Low | 21.1 (234) | 16.5 (121) | 30.0 (113) | |
| Intermediate | 59.0 (653) | 54.4 (399) | 68.1 (254) | |
| High | 19.9 (207) | 29.1 (201) | 1.6 (6) | |
| Number of natural teeth | <0.001d | |||
| Edentulous | 4.1 (46) | 6.0 (44) | 0.5 (2) | |
| 1–7 | 2.9 (32) | 3.1 (23) | 2.4 (9) | |
| 8–15 | 7.0 (78) | 5.3 (39) | 9.7 (39) | |
| 16–23 | 13.3 (147) | 4.6 (34) | 30.3 (113) | |
| 23–32 | 72.7 (755) | 81.0 (591) | 57.1 (209) |
| . | Whole study population (n = 1107) . | Finnish population (n = 734) . | Russian population (n = 373) . | . |
|---|---|---|---|---|
| Variable . | % (n) . | % (n) . | % (n) . | P-valuea . |
| Gender | ns | |||
| Females | 59.7 (661) | 57.9 (425) | 63.3 (236) | |
| Males | 40.7 (446) | 42.1 (309) | 36.7 (137) | |
| Age (years) | ns | |||
| 25–35 | 28.4 (314) | 26.3 (193) | 32.4 (121) | |
| 36–45 | 34.0 (376) | 35.0 (257) | 31.9 (119) | |
| 46–55 | 37.6 (417) | 38.7 (284) | 35.7 (133) | |
| Current smoker | 31.0 (343) | 25.6 (188) | 41.6 (155) | <0.001 |
| Increased CDTb | 14.8 (164) | 8.3 (61) | 27.6 (103) | <0.001 |
| Obesec | 16.3 (180) | 15.0 (110) | 18.8 (70) | ns |
| Socioeconomic status | <0.001 | |||
| Low | 21.1 (234) | 16.5 (121) | 30.0 (113) | |
| Intermediate | 59.0 (653) | 54.4 (399) | 68.1 (254) | |
| High | 19.9 (207) | 29.1 (201) | 1.6 (6) | |
| Number of natural teeth | <0.001d | |||
| Edentulous | 4.1 (46) | 6.0 (44) | 0.5 (2) | |
| 1–7 | 2.9 (32) | 3.1 (23) | 2.4 (9) | |
| 8–15 | 7.0 (78) | 5.3 (39) | 9.7 (39) | |
| 16–23 | 13.3 (147) | 4.6 (34) | 30.3 (113) | |
| 23–32 | 72.7 (755) | 81.0 (591) | 57.1 (209) |
Chi-square test.
CDT reference value: males ≥20 U/l, females ≥26 U/l.
BMI ≥30 kg/m2.
Mann–Whitney test.
The mean serum IgG antibody levels against each of the three pathogens, HSV, A. actinomycetemcomitans, and P. gingivalis, were significantly (P < 0.001) lower in the Finnish than in the Russian study population (Table 2). Similarly, the proportions of seronegative subjects for the pathogens were higher (P < 0.001) in the Finnish than in the Russian population. The two study populations also differed significantly as regards all serum lipid parameters as well as systolic and diastolic blood pressure: the Finnish subjects had higher (P < 0.001) mean serum concentrations of the total, LDL, and HDL cholesterol, as well as concentrations of triglycerides, whereas they had lower values for systolic (P < 0.05) and diastolic (P < 0.001) blood pressure (Table 2).
Mean results from the serum determinations and mean blood pressure values in the Finnish and Russian study populations
| . | Whole population . | Finnish population . | Russian population . | . | ||||
|---|---|---|---|---|---|---|---|---|
. | n = 1107 . | n = 734 . | n = 373 . | P-valuea . | ||||
| Serum IgG antibody to (EU) | ||||||||
| HSV, mean (SD) | 5103 (4212) | 4437 (4537) | 6414 (3100) | <0.001 | ||||
| Seronegative, n (%)b | 239 (22) | 236 (32) | 3 (1) | <0.001 | ||||
| A. actinomycetemcomitans, mean (SD) | 5.6 (3.1) | 5.2 (3.0) | 6.5 (3.1) | <0.001 | ||||
| Seronegative, n (%)c | 562 (51) | 430 (59) | 132 (35) | <0.001 | ||||
| P. gingivalis, mean (SD) | 5.7 (3.1) | 4.9 (2.7) | 7.2 (3.3) | <0.001 | ||||
| Seronegative, n (%)c | 645 (58) | 524 (71) | 121 (32) | <0.001 | ||||
| Serum lipids, mean (SD) | ||||||||
| Total cholesterol (mmol/l) | 5.23 (1.05) | 5.42 (1.05) | 4.87 (0.97) | <0.001 | ||||
| LDL cholesterol (mmol/l) | 3.24 (0.92) | 3.35 (0.92) | 3.01 (0.87) | <0.001 | ||||
| HDL cholesterol (mmol/l) | 1.40 (0.35) | 1.44 (0.37) | 1.33 (0.32) | <0.001 | ||||
| Triglycerides (mmol/l) | 1.30 (0.87) | 1.38 (0.97) | 1.15 (0.60) | <0.001 | ||||
| Blood pressure, mean (SD) | ||||||||
| Systolic (mmHg) | 131.4 (20) | 130.2 (18) | 133.8 (22) | <0.05 | ||||
| Diastolic (mmHg) | 80.2 (12) | 79.2 (11) | 82.2 (13) | <0.001 | ||||
| . | Whole population . | Finnish population . | Russian population . | . | ||||
|---|---|---|---|---|---|---|---|---|
. | n = 1107 . | n = 734 . | n = 373 . | P-valuea . | ||||
| Serum IgG antibody to (EU) | ||||||||
| HSV, mean (SD) | 5103 (4212) | 4437 (4537) | 6414 (3100) | <0.001 | ||||
| Seronegative, n (%)b | 239 (22) | 236 (32) | 3 (1) | <0.001 | ||||
| A. actinomycetemcomitans, mean (SD) | 5.6 (3.1) | 5.2 (3.0) | 6.5 (3.1) | <0.001 | ||||
| Seronegative, n (%)c | 562 (51) | 430 (59) | 132 (35) | <0.001 | ||||
| P. gingivalis, mean (SD) | 5.7 (3.1) | 4.9 (2.7) | 7.2 (3.3) | <0.001 | ||||
| Seronegative, n (%)c | 645 (58) | 524 (71) | 121 (32) | <0.001 | ||||
| Serum lipids, mean (SD) | ||||||||
| Total cholesterol (mmol/l) | 5.23 (1.05) | 5.42 (1.05) | 4.87 (0.97) | <0.001 | ||||
| LDL cholesterol (mmol/l) | 3.24 (0.92) | 3.35 (0.92) | 3.01 (0.87) | <0.001 | ||||
| HDL cholesterol (mmol/l) | 1.40 (0.35) | 1.44 (0.37) | 1.33 (0.32) | <0.001 | ||||
| Triglycerides (mmol/l) | 1.30 (0.87) | 1.38 (0.97) | 1.15 (0.60) | <0.001 | ||||
| Blood pressure, mean (SD) | ||||||||
| Systolic (mmHg) | 131.4 (20) | 130.2 (18) | 133.8 (22) | <0.05 | ||||
| Diastolic (mmHg) | 80.2 (12) | 79.2 (11) | 82.2 (13) | <0.001 | ||||
Chi-square test for frequency distributions, Mann–Whitney test for differences in the mean.
Titer <230 EU.
Level <5.0 EU.
Mean results from the serum determinations and mean blood pressure values in the Finnish and Russian study populations
| . | Whole population . | Finnish population . | Russian population . | . | ||||
|---|---|---|---|---|---|---|---|---|
. | n = 1107 . | n = 734 . | n = 373 . | P-valuea . | ||||
| Serum IgG antibody to (EU) | ||||||||
| HSV, mean (SD) | 5103 (4212) | 4437 (4537) | 6414 (3100) | <0.001 | ||||
| Seronegative, n (%)b | 239 (22) | 236 (32) | 3 (1) | <0.001 | ||||
| A. actinomycetemcomitans, mean (SD) | 5.6 (3.1) | 5.2 (3.0) | 6.5 (3.1) | <0.001 | ||||
| Seronegative, n (%)c | 562 (51) | 430 (59) | 132 (35) | <0.001 | ||||
| P. gingivalis, mean (SD) | 5.7 (3.1) | 4.9 (2.7) | 7.2 (3.3) | <0.001 | ||||
| Seronegative, n (%)c | 645 (58) | 524 (71) | 121 (32) | <0.001 | ||||
| Serum lipids, mean (SD) | ||||||||
| Total cholesterol (mmol/l) | 5.23 (1.05) | 5.42 (1.05) | 4.87 (0.97) | <0.001 | ||||
| LDL cholesterol (mmol/l) | 3.24 (0.92) | 3.35 (0.92) | 3.01 (0.87) | <0.001 | ||||
| HDL cholesterol (mmol/l) | 1.40 (0.35) | 1.44 (0.37) | 1.33 (0.32) | <0.001 | ||||
| Triglycerides (mmol/l) | 1.30 (0.87) | 1.38 (0.97) | 1.15 (0.60) | <0.001 | ||||
| Blood pressure, mean (SD) | ||||||||
| Systolic (mmHg) | 131.4 (20) | 130.2 (18) | 133.8 (22) | <0.05 | ||||
| Diastolic (mmHg) | 80.2 (12) | 79.2 (11) | 82.2 (13) | <0.001 | ||||
| . | Whole population . | Finnish population . | Russian population . | . | ||||
|---|---|---|---|---|---|---|---|---|
. | n = 1107 . | n = 734 . | n = 373 . | P-valuea . | ||||
| Serum IgG antibody to (EU) | ||||||||
| HSV, mean (SD) | 5103 (4212) | 4437 (4537) | 6414 (3100) | <0.001 | ||||
| Seronegative, n (%)b | 239 (22) | 236 (32) | 3 (1) | <0.001 | ||||
| A. actinomycetemcomitans, mean (SD) | 5.6 (3.1) | 5.2 (3.0) | 6.5 (3.1) | <0.001 | ||||
| Seronegative, n (%)c | 562 (51) | 430 (59) | 132 (35) | <0.001 | ||||
| P. gingivalis, mean (SD) | 5.7 (3.1) | 4.9 (2.7) | 7.2 (3.3) | <0.001 | ||||
| Seronegative, n (%)c | 645 (58) | 524 (71) | 121 (32) | <0.001 | ||||
| Serum lipids, mean (SD) | ||||||||
| Total cholesterol (mmol/l) | 5.23 (1.05) | 5.42 (1.05) | 4.87 (0.97) | <0.001 | ||||
| LDL cholesterol (mmol/l) | 3.24 (0.92) | 3.35 (0.92) | 3.01 (0.87) | <0.001 | ||||
| HDL cholesterol (mmol/l) | 1.40 (0.35) | 1.44 (0.37) | 1.33 (0.32) | <0.001 | ||||
| Triglycerides (mmol/l) | 1.30 (0.87) | 1.38 (0.97) | 1.15 (0.60) | <0.001 | ||||
| Blood pressure, mean (SD) | ||||||||
| Systolic (mmHg) | 131.4 (20) | 130.2 (18) | 133.8 (22) | <0.05 | ||||
| Diastolic (mmHg) | 80.2 (12) | 79.2 (11) | 82.2 (13) | <0.001 | ||||
Chi-square test for frequency distributions, Mann–Whitney test for differences in the mean.
Titer <230 EU.
Level <5.0 EU.
Bivariate correlations between variables in the whole study population
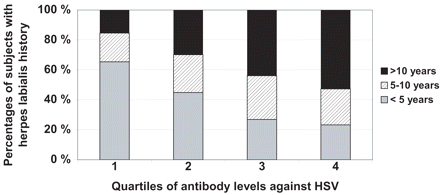
A two-tailed Pearson correlation analysis was performed to examine whether serum IgG antibody levels against HSV, A. actinomycetemcomitans, and P. gingivalis were associated with each other. Positive correlations (P < 0.001) were found between the antibody levels to HSV and periodontal pathogens, A. actinomycetemcomitans (r = 0.182) and P. gingivalis (r = 0.167), between the two periodontal pathogens (r = 0.323) as well as between the antibody level against HSV and the number of years from the first herpes labialis experience (r = 0.256). As presented in Figure 1, the subjects with high antibody level against HSV had frequently a long herpes labialis history (Figure 1).
Percentage values of subjects (N = 409) with indicated length of herpes labialis history in quartiles of antibody levels against HSV. The length of herpes labialis experience was obtained from the questionnaires filled by the study subjects
Comparison of subjects with a high and low combined antibody response to periodontal pathogens
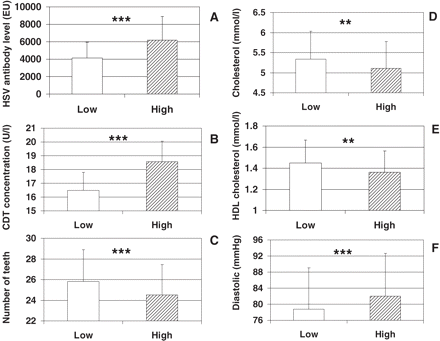
To study the relationship between periodontitis and HSV infection as well as periodontitis and risk factors for atherosclerosis, the whole study population was divided into two groups according to their combined antibody response to periodontal pathogens. The group of subjects with the value ≥14 EU (high combined antibody response; n = 305) had significantly lower mean total cholesterol (P < 0.01) and HDL cholesterol concentrations (P < 0.01) as well as higher mean diastolic blood pressure (P < 0.001) than the group with the value <14 EU (low combined antibody response; n = 802). The former group of subjects also had a higher mean antibody level against HSV (P < 0.001) and a higher mean CDT value (P < 0.001) but fewer teeth (P < 0.001) than the latter group (Figure 2). When the Finnish and Russian populations were analysed separately, the results tended to be similar, but only diastolic blood pressure values (P < 0.05) and HSV antibody levels (P < 0.01) remained significantly higher in the group with a high combined antibody response compared with those with a low response in both countries.
Comparison of mean (SD) serum HSV antibody levels (a), serum carbohydrate-deficient transferrin (CDT) concentration (b), number of teeth (c), total cholesterol concentration (d), HDL cholesterol concentration (e), and diastolic blood pressure (f) between subjects with a low (<14.0 EU, n = 803) and a high (≥14.0 EU, n = 304) combined antibody response to periodontal pathogens, A. actinomycetemcomitans and P. gingivalis. The statistically significant differences are shown in the figure by stars: **P < 0.01, ***P < 0.001
Serum lipid profiles in the 3-pathogen antibody categories
Since the antibody levels against A. actinomycetemcomitans, P. gingivalis, and HSV were positively correlated with each other, we also studied the joint association of these antibody levels with the traditional risk factors of atherosclerosis (Table 3). When the whole study population (n = 1107) was divided into three categories (low, intermediate, high) according to the 3-pathogen antibody score, significant trends were seen in both serum lipid profiles and blood pressure (Table 3). Total cholesterol (P = 0.012) and HDL cholesterol (P < 0.001) concentrations had significant descending trends, whereas both systolic (P = 0.007) and diastolic (P = 0.001) blood pressure had ascending trends towards the higher 3-pathogen antibody category.
Lipid profiles and blood pressure values (mean and SD) in relation to infection burden by HSV and two periodontal pathogens
| . | Serum lipids (mmol/l) . | . | . | . | Blood pressure (mmHg) . | . | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3-pathogen score . | Cholesterol . | LDL . | HDL . | Triglycerides . | Systolic . | Diastolic . | ||||||
| All subjects, n = 1094 | ||||||||||||
| Low, n = 237 | 5.42 (1.00) | 3.36 (0.91) | 1.46 (0.36) | 1.34 (0.77) | 128 (17) | 78 (11) | ||||||
| Interm., n = 618 | 5.19 (1.08) | 3.19 (0.93) | 1.41 (0.36) | 1.29 (0.87) | 132 (20) | 80 (12) | ||||||
| High, n = 239 | 5.17 (1.01) | 3.25 (0.87) | 1.34 (0.33) | 1.30 (0.94) | 134 (20) | 82 (11) | ||||||
| P for trenda | 0.012 | 0.248 | <0.001 | 0.265 | 0.007 | 0.001 | ||||||
| Subjects without medication for hypertension, n = 913 | ||||||||||||
| Low, n = 222 | 5.43 (1.02) | 3.37 (0.92) | 1.46 (0.35) | 1.31 (0.75) | 127 (16) | 78 (11) | ||||||
| Interm., n = 518 | 5.20 (1.06) | 3.19 (0.92) | 1.43 (0.36) | 1.26 (0.89) | 129 (18) | 79 (11) | ||||||
| High, n = 173 | 5.05 (0.97) | 3.12 (0.83) | 1.36 (0.33) | 1.25 (0.99) | 130 (17) | 80 (10) | ||||||
| P for trenda | <0.001 | 0.006 | 0.008 | 0.082 | 0.106 | 0.027 | ||||||
| . | Serum lipids (mmol/l) . | . | . | . | Blood pressure (mmHg) . | . | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3-pathogen score . | Cholesterol . | LDL . | HDL . | Triglycerides . | Systolic . | Diastolic . | ||||||
| All subjects, n = 1094 | ||||||||||||
| Low, n = 237 | 5.42 (1.00) | 3.36 (0.91) | 1.46 (0.36) | 1.34 (0.77) | 128 (17) | 78 (11) | ||||||
| Interm., n = 618 | 5.19 (1.08) | 3.19 (0.93) | 1.41 (0.36) | 1.29 (0.87) | 132 (20) | 80 (12) | ||||||
| High, n = 239 | 5.17 (1.01) | 3.25 (0.87) | 1.34 (0.33) | 1.30 (0.94) | 134 (20) | 82 (11) | ||||||
| P for trenda | 0.012 | 0.248 | <0.001 | 0.265 | 0.007 | 0.001 | ||||||
| Subjects without medication for hypertension, n = 913 | ||||||||||||
| Low, n = 222 | 5.43 (1.02) | 3.37 (0.92) | 1.46 (0.35) | 1.31 (0.75) | 127 (16) | 78 (11) | ||||||
| Interm., n = 518 | 5.20 (1.06) | 3.19 (0.92) | 1.43 (0.36) | 1.26 (0.89) | 129 (18) | 79 (11) | ||||||
| High, n = 173 | 5.05 (0.97) | 3.12 (0.83) | 1.36 (0.33) | 1.25 (0.99) | 130 (17) | 80 (10) | ||||||
| P for trenda | <0.001 | 0.006 | 0.008 | 0.082 | 0.106 | 0.027 | ||||||
Lipid profiles and blood pressure values (mean and SD) in relation to infection burden by HSV and two periodontal pathogens
| . | Serum lipids (mmol/l) . | . | . | . | Blood pressure (mmHg) . | . | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3-pathogen score . | Cholesterol . | LDL . | HDL . | Triglycerides . | Systolic . | Diastolic . | ||||||
| All subjects, n = 1094 | ||||||||||||
| Low, n = 237 | 5.42 (1.00) | 3.36 (0.91) | 1.46 (0.36) | 1.34 (0.77) | 128 (17) | 78 (11) | ||||||
| Interm., n = 618 | 5.19 (1.08) | 3.19 (0.93) | 1.41 (0.36) | 1.29 (0.87) | 132 (20) | 80 (12) | ||||||
| High, n = 239 | 5.17 (1.01) | 3.25 (0.87) | 1.34 (0.33) | 1.30 (0.94) | 134 (20) | 82 (11) | ||||||
| P for trenda | 0.012 | 0.248 | <0.001 | 0.265 | 0.007 | 0.001 | ||||||
| Subjects without medication for hypertension, n = 913 | ||||||||||||
| Low, n = 222 | 5.43 (1.02) | 3.37 (0.92) | 1.46 (0.35) | 1.31 (0.75) | 127 (16) | 78 (11) | ||||||
| Interm., n = 518 | 5.20 (1.06) | 3.19 (0.92) | 1.43 (0.36) | 1.26 (0.89) | 129 (18) | 79 (11) | ||||||
| High, n = 173 | 5.05 (0.97) | 3.12 (0.83) | 1.36 (0.33) | 1.25 (0.99) | 130 (17) | 80 (10) | ||||||
| P for trenda | <0.001 | 0.006 | 0.008 | 0.082 | 0.106 | 0.027 | ||||||
| . | Serum lipids (mmol/l) . | . | . | . | Blood pressure (mmHg) . | . | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3-pathogen score . | Cholesterol . | LDL . | HDL . | Triglycerides . | Systolic . | Diastolic . | ||||||
| All subjects, n = 1094 | ||||||||||||
| Low, n = 237 | 5.42 (1.00) | 3.36 (0.91) | 1.46 (0.36) | 1.34 (0.77) | 128 (17) | 78 (11) | ||||||
| Interm., n = 618 | 5.19 (1.08) | 3.19 (0.93) | 1.41 (0.36) | 1.29 (0.87) | 132 (20) | 80 (12) | ||||||
| High, n = 239 | 5.17 (1.01) | 3.25 (0.87) | 1.34 (0.33) | 1.30 (0.94) | 134 (20) | 82 (11) | ||||||
| P for trenda | 0.012 | 0.248 | <0.001 | 0.265 | 0.007 | 0.001 | ||||||
| Subjects without medication for hypertension, n = 913 | ||||||||||||
| Low, n = 222 | 5.43 (1.02) | 3.37 (0.92) | 1.46 (0.35) | 1.31 (0.75) | 127 (16) | 78 (11) | ||||||
| Interm., n = 518 | 5.20 (1.06) | 3.19 (0.92) | 1.43 (0.36) | 1.26 (0.89) | 129 (18) | 79 (11) | ||||||
| High, n = 173 | 5.05 (0.97) | 3.12 (0.83) | 1.36 (0.33) | 1.25 (0.99) | 130 (17) | 80 (10) | ||||||
| P for trenda | <0.001 | 0.006 | 0.008 | 0.082 | 0.106 | 0.027 | ||||||
In the subsequent analyses we excluded subjects, who had medication for hypertension (n = 183, 17%) (Table 3). Since only 9 (1.6%) subjects had a cholesterol-lowering medication, all these study subjects were included in the analyses. The analyses still revealed descending trends towards the higher 3-pathogen antibody category in total cholesterol (P < 0.001), HDL cholesterol (P = 0.008), and LDL cholesterol (P = 0.006) concentrations but an ascending trend (P = 0.027) in diastolic blood pressure. Thus, after excluding the subjects with medication for hypertension, only the trend in systolic blood pressure towards the higher 3-pathogen antibody category was no longer significant (P = 0.106) (Table 3).
Multivariate analyses
Multivariate logistic and linear regression analyses were performed to examine the effect of confounding factors on the association between antibody levels with each other and with CVD risk factors. All models were adjusted for age, gender, smoking, BMI, area (Finland or Russia), years of education, CDT, triglycerides, and number of teeth. In the logistic regression model with the combined antibody response to periodontal pathogens as a dependent variable, the ORs (95% CI) for HSV antibody quartiles 2–4 compared with the first quartile were 1.43 (0.88–2.32), 1.74 (1.07–2.82), and 1.89 (1.18–3.02), respectively, P < 0.001. In the same model, the OR for HDL cholesterol concentration was 0.35 (0.20–0.60), P < 0.001. The association with diastolic blood pressure as an independent variable was not statistically significant (P = 0.354). In the linear regression model with the 3-pathogen antibody category as a dependent variable, a negative association with HDL cholesterol concentration was found, β = −0.067/1 mmol/l (−0.235; −0.018), P < 0.05. The association as regards diastolic blood pressure was not significant.
Discussion
We studied the relationship between infection burden and classic risk factors for atherosclerosis in a large random study population from Finland and Russia. The infection burden was evaluated using combined serum IgG antibody response to periodontal pathogens and HSV as a joint variable. Elevated antibody levels against these pathogens and low HDL cholesterol concentration were significantly associated with each other and also after adjusting for confounding factors.
A serological approach was chosen for the present study for two reasons: on one hand it made it possible to clarify the antibody response in two common chronic infections, periodontitis and HSV infection. On the other hand, it allowed the analysis of a large study population. To enhance the detection of seropositive individuals for A. actinomycetemcomitans and P. gingivalis, we used multiserotype ELISAs. Previous results from our laboratory have shown that the method has sensitivity and specificity of 71 and 90% for finding clinically diagnosed periodontitis.23 Methodological factors may at least partly explain the discrepant findings reported on the compatibility of clinical periodontal status and serum IgG levels against these species.24,25 For instance, using a single strain of a species as the source of ELISA antigen is likely to lead to underestimation of the number of seropositive individuals.26,27 However, the serological approach may enclose a complexity of variables other than those deriving from the laboratory methods. This can be reflected by the inconsistent results from previous seroepidemiological studies on the relationship between other chronic infections and risk factors for atherosclerosis.10,28 In the present study, we attempted to avoid bias by selecting the study population, which was random, population-based, and heterogeneously collected and stratified only by age and gender. Both males and females were included to allow balanced data analyses regarding gender.
HSV was included in the study because it causes a recurrent infection, which commonly occurs in the mouth. Additionally, HSV infection has been associated with both periodontitis14 and CVD.29,30 Previous studies on herpes viruses and periodontitis have particularly focused on cytomegalovirus or Epstein–Barr virus.31 Therefore, we chose the less-studied HSV as a representative of herpes viruses. Even so, the results from multivariate logistic regression analysis on antibody levels against periodontal pathogens and HSV support an independent association between herpes virus and periodontitis infections.
Because both HSV-1 and HSV-2 can cause oral infections, instead of type-specific kits we used an assay that detects serum antibodies against HSV. HSV-1 infection is common already from childhood and may protect adults from genital HSV-1 infection.32 Oral HSV-2 infection only rarely recurs and is almost always associated with concomitant primary genital infection.33 The present anamnestic data of the subjects revealed that the prevalence of genital herpes infection was very low (2%). Thus, we assume that most of the antibodies against HSV in our study were targeted against HSV-1. Further evidence was found from the result that there was a positive correlation between antibody levels against HSV and years of having herpes labialis. The observation that serum IgG antibody levels against HSV are significantly higher in subjects with recurrent herpes labialis than in asymptomatic seropositive subjects34 suggests that also in our study HSV infection may have been recurrent throughout the years in the subjects with elevated antibodies against HSV.
Seroprevalence of HSV infection is globally well documented, but the data from Russia are limited.34 Recent results indicate that the prevalence of seropositivity to another bacterium causing persistent infections, H. pylori, is higher among adolescents in Russia than in developed countries35 and resembles the situation in developing countries. The mean serum IgG levels against all three pathogens were significantly higher in the present Russian than in the Finnish study population suggesting that periodontitis and HSV infection may also be more common in Russia than in Finland.
If we assume that the subjects with high combined antibody response to periodontal pathogens suffer from periodontitis, our results on plasma lipid profiles in periodontitis and non-periodontitis groups differ from those of previous studies.36–38 These studies found no statistically significant difference in HDL cholesterol concentrations but higher total cholesterol, LDL cholesterol, and triglyceride concentrations in subjects with periodontitis than in those without periodontitis.36–38 In our study, the subjects with ‘serologically defined periodontitis’, i.e. high combined antibody response to periodontal pathogens, had lower serum total cholesterol concentration than those without it. This was most likely due to lower HDL cholesterol concentration, as also shown by us earlier,20 because no difference in the LDL cholesterol concentrations was found. The differences may be partly explained by the selection of the study populations, age distribution, and criteria used for determining periodontitis. In the present study we combined two culturally and socioeconomically distinct populations collected from population registers. It resulted in a wider spectrum of the target infections, background variables, and individual infection responses than what has been possible in the aforementioned studies. Additionally, there is a strong positive correlation between ageing and prevalence of periodontitis as well as ageing and serum concentrations of total cholesterol, LDL cholesterol, and triglycerides.39 Finally, the methods used in studies of Cutler et al.36, Lösche et al.38, and Katz et al.38 for determining periodontitis were different from ours. They used clinical examination, whereas our study utilized serum antibody response against periodontal pathogens. Measuring systemic immune response to periodontal pathogens may reflect the systemic effects of the disease better than the clinical examination does. Our approach may be particularly efficient for picking up individuals who raise high immune responses in early phases of periodontal infection. Identification of these individuals may enhance possibilities to find relevant associations between periodontitis and CVD risk factors. Of special interest is that the high responders to infection may also be particularly susceptible to factors considered proatherogenic.40
Recent studies emphasize an aggregate pathogen load rather than single pathogens as infectious triggers for proatherogenic phenomena.1,41 Several cross-sectional and case–control studies link chronic infections, among them also periodontitis and HSV infection, to CVD.17,30,42,43 The strong correlation between antibody levels against periodontal pathogens and HSV indicated that the joint effect of these infections may be weighty more than their single effects on the traditional risk factors for atherosclerosis. Earlier the joint effect of HSV and C. pneumoniae infections on CHD risk was shown to be higher than their single effects.30 Additionally, the aggregate number of anti-pathogen antibodies in general has been associated with the extent of atherosclerosis.6
In the present study the infectious burden, as determined by combined antibody response to periodontal pathogens and HSV, was associated with a major established risk factor for atherosclerosis, low serum HDL cholesterol concentration.12 Interestingly, this negative association remained statistically significant after adjusting for age, gender, area, years of education, smoking, BMI, CDT, and number of teeth. Notable is that periodontitis not only decreases HDL cholesterol concentrations, but also results in conformational and compositional changes of HDL, which decreases their anti-atherogenic potential.44
In addition to our finding of a decreasing HDL cholesterol concentration, increasing blood pressure towards a higher joint antibody level against A. actinomycetemcomitans, P. gingivalis, and HSV is also of interest. Low HDL cholesterol concentration, hypertension, central obesity, and glucose intolerance are characteristics of the metabolic syndrome.45 It has been reported previously that combined seropositivity to H. pylori and C. pneumoniae is associated not only with obesity but also with ageing and social class.46 Thus, further studies are needed to clarify whether periodontitis would be related to obesity or metabolic syndrome.
The present results suggest that the same individuals raise antibodies to both periodontal pathogens and HSV. Whether this is due to the exposure to these infectious agents or individual susceptibility to persistent infections is not known. Because high HSV antibodies are also associated with a long history of recurrent orofacial herpes, and because oral mucosal irritation may trigger recurrences, we cannot exclude the possibility that chronic periodontitis has been acting as a trigger of HSV recurrences and, consequently, an indirect inducer of the elevated HSV antibody levels. The independent association between low serum HDL cholesterol concentration and elevated systemic immune response to pathogens in two persistent infections commonly manifesting in the oral cavity prompts the need for further investigations on CVD risk in individuals with these characteristics.
The authors would like to thank Ms Tiina Karvonen for excellent technical assistance. This work was financially supported by the Academy of Finland (grants #77613, #75953 and #205987, #211129), by the Finnish Dental Association, by the Finnish Dental Society Apollonia, by the Paulo Foundation, and by the Finnish Cultural Foundation.
References
Epstein SE. The multiple mechanisms by which infection may contribute to atherosclerosis development and course.
Leinonen M, Saikku P. Evidence for infectious agents in cardiovascular disease and atherosclerosis.
Mattila KJ, Valtonen VV, Nieminen MS, Asikainen S. Role of infection as a risk factor for atherosclerosis, myocardial infarction and stroke.
Epstein SE, Zhou YF, Zhu J. Infection and atherosclerosis emerging mechanistic paradigms.
Li L, Messas E, Batista EL Jr, Levine RA, Amar S. Porphyromonas gingivalis infection accelerates the progression of atherosclerosis in heterozygous apolipoprotein E-deficient murine model.
Espinola-Klein C, Rupprecht HJ, Blankenberg S et al. Impact of infectious burden on progression of carotid atherosclerosis.
Pussinen PJ, Metso J, Malle E et al. The role of plasma phospholipid transfer protein (PLTP) in HDL remodeling in acute-phase patients.
Khovidhunkit W, Kim MS, Memon RA et al. Effects of infection and inflammation on lipid and lipoprotein metabolism: mechanisms and consequences to the host.
Sammalkorpi K, Valtonen V, Kerttula Y, Nikkilä E, Taskinen MR. Changes in serum lipoprotein pattern induced by acute infections.
Laurila A, Bloigu A, Näyhä S, Hassi J, Leinonen M, Saikku P. Chronic Chlamydia pneumoniae infection is associated with a serum lipid profile known to be a risk factor for atherosclerosis.
Fruchart JC, Duriez P. HDL and triglyceride as terapeutic targets.
Gordon DJ, Rifkind BM. High density lipoprotein – the clinical implications of recent studies.
Contreras A, Umeda M, Chen C, Bakker I, Morrison JL, Slots J. Relationship between herpesviruses and adult periodontitis and periodontopathic bacteria.
Slots J, Contreras A. Herpesviruses: a unifying causative factor in periodontitis?
Kamma JJ, Contreras A, Slots J. Herpes viruses and periodontopathic bacteria in early-onset periodontitis.
Scannapieco FA, Bush RB, Paju S. Associations between periodontal disease and risk for atherosclerosis, cardiovascular disease, and stroke. A systematic review.
DeStefano F, Merritt RK, Anda RF, Casper ML, Eaker ED. Trends in nonfatal coronary heart disease in the United States 1980 through 1989.
Beck J, Garcia R, Heiss G, Vokonas PS, Offenbacher S. Periodontal disease and cardiovascular disease.
Pussinen PJ, Jousilahti P, Alfthan G, Palosuo T, Asikainen S, Salomaa V. Antibodies to periodontal pathogens are associated with coronary heart disease.
Vartiainen E, Petäys T, Haahtela T, Jousilahti P, Pekkanen J. Allergic diseases, skin prick test responses and IgE levels in North Karelia, Finland, and the Republic of Karelia, Russia.
Laatikainen T, Alho H, Vartiainen E, Jousilahti P, Sillanaukee P, Puska P. Self-reported alcohol consumption and association to carbohydrate-deficient transferrin and gamma-glutamyltransferase in a random sample of the general population in the Republic of Karelia, Russia and in North Karelia, Finland.
Pussinen PJ, Vilkuna-Rautiainen T, Alfthan G, Mattila K, Asikainen S. Multiserotype enzyme-linked immunosorbent assay as a diagnostic aid for periodontitis in large-scale studies.
Ebersole JL, Cappelli D, Steffen MJ. Longitudinal dynamics of infection and serum antibody in A. actinomycetemcomitans periodontitis.
Lamster IB, Kaluszhner-Shapira M, Herrera-Abreu M, Sinha R, Crbic JT. Serum antibody response to Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis: implications to periodontal diagnosis.
Chen C, Ashimoto A. Variable serum immunoglobulin G immune response to genetically distinct Eikenella corrodens strains coexisting in the human oral cavity.
Vilkuna-Rautiainen T, Pussinen PJ, Mattila K et al. Antigenically diverse reference strains and autologous strains of Actinobacillus actinomycetemcomitans are equally efficient antigens in enzyme-linked immunosorbent assay analysis.
Hoffmeister A, Rothenbacher D, Bode G et al. Current infection with Helicobacter pylori but not seropositivity to Clamydia pneumoniae or cytomegalovirus is associated with an atherogenic, modified lipid profile.
Roivainen M, Viik-Kajander M, Palosuo T et al. Infections, inflammation, and the risk of coronary heart disease.
Slots J. Interactions between herpesviruses and bacteria in human periodontal disease. In: Brogden KA, Gutmiller JM (eds). Polymicrobial Diseases. Washington DC: ASM Press,
Lafferty WE. The changing epidemiology of HSV-1 and HSV-2 and implications for serological testing.
Linnavuori KH. History of recurrent mucocutaneous herpes correlates with relatively low interferon production by herpes simplex virus-exposed cultured monosytes.
Smith JS, Robinson JN. Age-specific prevalence of infection with Herpes simplex virus types 2 and 1: a global review.
Reshetnikov OV, Denisova DV, Zavyalova LG, Haiva VM, Granberg C. Helicobacter pylori seropositivity among adolescents in Novosibirsk, Russia: prevalence and associated factors.
Cutler CW, Shinedling EA, Nunn M et al. Association between periodontitis and hyperlipidemia: cause or effect?
Lösche W, Karapetow F, Pohl A, Pohl C, Kocher T. Plasma lipid and blood glucose levels in patients with destructive periodontal disease.
Katz J, Chaushu G, Sharabi Y. On the association between hypercholesterolemia, cardiovascular disease and severe periodontal disease.
Katz J, Flugelman MY, Goldberg A, Heft M. Association between periodontal pockets and elevated cholesterol and low density lipoprotein cholesterol levels.
Zhu J, Quyymi AA, Norman JE, Csako G, Epstein SE. Cytomegalovirus in the pathogenesis of atherosclerosis: the role of inflammation as reflected by elevated C-reactive protein levels.
Zhu J, Nieto FJ, Horne BD, Anderson JL, Muhlestein JB, Epstein SE. Prospective study of pathogen burden and risk of myocardial infarction and death.
Pussinen PJ, Nyyssönen K, Alfthan G, Salonen R, Laukkanen JA, Salonen JT. Serum antibody levels to Actinobacillus actinomycetemcomitans predict the risk for coronary heart disease.
Shi Y, Togunaga O. Herpesvirus (HSV-1, EBV and CMV) infections in atherosclerotic compared with non-atherosclerotic aortic tissue.
Pussinen PJ, Jauhiainen M, Vilkuna-Rautiainen T et al. Periodontitis decreases the antiatherogenic potency of high density lipoprotein.