-
PDF
- Split View
-
Views
-
Cite
Cite
Stefan Herget‐Rosenthal, Günther Marggraf, Frank Pietruck, Johannes Hüsing, Miriam Strupat, Thomas Philipp, Andreas Kribben, Procalcitonin for accurate detection of infection in haemodialysis, Nephrology Dialysis Transplantation, Volume 16, Issue 5, May 2001, Pages 975–979, https://doi.org/10.1093/ndt/16.5.975
Close - Share Icon Share
Abstract
Background. Infection results in considerable morbidity and mortality in haemodialysis patients. Diagnosis of infection can be difficult because currently applied laboratory parameters may be non‐specifically altered due to uraemia or haemodialysis (HD). This study investigated the diagnostic value and kinetics of serum procalcitonin (PCT), a low‐molecular‐weight protein, in patients receiving intermittent HD.
Methods. Sixty‐eight patients receiving intermittent HD for end‐stage renal disease (n=48) or acute renal failure (n=20) were prospectively studied, 47 treated with high‐flux and 21 with low‐flux membranes. Of 36 patients with severe infections or sepsis, 27 were treated with high‐flux and nine with low‐flux membranes. WBC, serum PCT and C‐reactive protein (CRP) concentrations were measured immediately before HD, and PCT repeatedly during the following 48 h.
Results. When determined immediately before HD, PCT demonstrated a sensitivity of 89%, a specificity of 81%, and positive and negative predictive values of 84 and 87%, indicating severe infection or sepsis. These levels were higher than the respective values for CRP (89, 48, 68 and 78%) and WBC (58, 75, 71 and 59%). After 4 h of HD with high‐flux membranes, PCT decreased significantly to 83±25% and did not return to predialysis concentrations before 48 h. This decrease in serum PCT resulted in markedly reduced sensitivity (65%) and negative predictive value (54%). In contrast, no marked change in PCT concentration occurred during or after HD with low‐flux membranes.
Conclusion. Serum PCT is an accurate indicator of severe infection and sepsis in patients receiving intermittent HD. High‐flux membranes substantially decrease PCT. When utilizing high flux membranes, serum PCT concentrations should be determined prior to the start of HD.
Introduction
Infections account for considerable morbidity and mortality in patients requiring haemodialysis (HD) [1,2]. Currently applied laboratory parameters of inflammation can be misleading in these patients because of uraemia or HD. Some of these parameters may be non‐specifically decreased, such as white blood count (WBC) [3,4] and others may be non‐specifically increased such as erythrocyte sedimentation rate [5], C‐reactive protein (CRP) [6,7], or other acute‐phase proteins [8].
Serum procalcitonin (PCT) is a precursor of calcitonin and has been reported to increase in patients with normal renal function during sepsis and severe bacterial or fungal infections [9–11]. Unlike other laboratory parameters, PCT does not increase, or only very moderately increases, in viral and localized infections or other non‐infectious inflammatory processes [10,12–14]. PCT may aid in differentiating sepsis from systemic inflammatory response syndrome [9,10,15] and serve as a monitor for the activity of infection [10,16]. An increase in serum PCT is detectable 4 h after endotoxaemia. Serum PCT peaks 6 h after endotoxaemia and has a serum half‐life of 25–30 h [17,18]. Recent findings indicate that PCT may be a cytokine‐like mediator of inflammation [18,19]. Mononuclear cells express PCT [20], but its precise pathophysiological role has not been determined yet.
Considering its characteristics, serum PCT may serve as an indicator of severe infections and sepsis in HD patients. However, elimination of serum PCT, a low‐molecular‐weight protein of 13 kDa [21], by HD with high‐flux membranes may limit its usefulness as an indicator of infection. On the contrary, synthesis of PCT could be induced during HD as has been demonstrated for some cytokines [22,23]. In both circumstances, HD would alter serum PCT concentrations independently of the presence, nature, or activity of infection.
In this study the diagnostic value of serum PCT as an indicator of severe infection and sepsis was evaluated in patients receiving intermittent HD for end‐stage renal disease (ESRD) or acute renal failure and was compared to that of CRP and WBC. We also evaluated serum PCT concentrations prior to, during, and in the 48 h after the start of HD with high‐ and low‐flux membranes to determine the effect of high‐flux membranes on serum PCT concentrations.
Subjects and methods
Patients
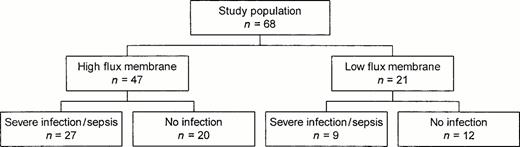
Sixty‐eight hospitalized patients were prospectively studied who required intermittent HD for ESRD (n=48) or acute renal failure (n=20). The cohort consisted of 47 patients dialysed with high‐flux and 21 patients with low‐flux membranes as indicated in Figure 1. The study period covered 48 h from the start of the HD procedure. Patients requiring HD before the 48 h end‐point of the study were excluded. Since surgery may increase PCT concentration [24], patients having surgery in the 5 days previous to or during the study period were also excluded. Informed consent was obtained from all patients prior to enrolment.
Age, gender, duration of HD procedure, type of dialysis membrane, mean blood flow and ultrafiltration rate, weight before and after HD, and type of infection were recorded for each patient. Severe infections were defined as previously published according to clinical and radiological criteria and in every case confirmed by positive blood cultures or the respective microbiological proof of pathogenic micro‐organisms [25,26]. Sepsis was defined as microbiologically confirmed infection in addition to at least two of the following criteria: WBC >12.0×109/l, temperature >38° or <36°C, heart rate >90 beats/min, respiratory rate >20 breaths/min or PaCO2 <4.3 kPa [27].
Methods
Intermittent haemodialysis was performed with high‐ or low‐flux polysulphone membranes (F60 or F8, Fresenius Medical Care, Bad Homburg, Germany). Membranes had not been previously used and the dialysate buffer was bicarbonate. Dialysate flow was kept constant at 500 ml/min for all patients.
Blood samples were obtained just prior to the start of dialysis (T0), and 2, 4, 12, 24 and 48 h after the start of HD. Blood samples 2 and 4 h after the start of HD were drawn from the line leading to the dialyser. Blood samples at all other time points were drawn from peripheral veins or central venous lines. Serum PCT concentrations were measured by immunoluminometric assay (Brahms Diagnostica, Berlin, Germany). PCT concentrations were corrected for HD induced fluid loss according to the formula of Bergstroem and Wehle [28]. Since severe infections and sepsis are unlikely at PCT levels below 1.5 ng/ml in patients with normal renal function [10,16], the concentration of 1.5 ng PCT/ml was chosen as cut‐off value indicating severe infection or sepsis. CRP and WBC were determined from the blood sample at T0. CRP was measured by immunonephelometric technique (DadeBehring, Marburg, Germany). Cut‐off values of greater than 5.0 mg/dl for CRP and 12×l09/l for WBC were adopted from previous reports in which they showed good accuracy indicating severe infections and sepsis [27,29,30].
Statistical analysis
Sensitivity, specificity, predictive values, and accuracy of PCT, CRP, and WBC were calculated from data of the entire cohort at T0 and additionally from data of the subgroup of patients dialysed with high‐flux membranes prior to, during, and at different time points after HD. Data are presented as median values with interquartile values in parenthesis (lower quartile; upper quartile), except when stated differently. Serum PCT concentrations are expressed either as absolute values or as percent of the T0 value. All testing was performed with the non‐parametric Wilcoxon's rank‐sum test comparing differences between groups. We used a nested hypotheses approach to test for statistically significant differences of PCT concentrations at various time points. As we assumed that the lowest PCT concentrations would be measured 4 h after the start of haemodialysis, testing was performed in the following order: PCT concentrations at T0vs PCT concentrations 4 h after the start of HD, PCT concentrations at T0vs PCT concentrations 2 h after the start of HD, PCT concentrations at T0vs PCT concentrations 12 h the after start of HD, PCT concentrations at T0vs PCT concentrations 24 h after the start of HD, and PCT concentrations at T0vs PCT concentrations 48 h after the start of HD. Testing was not continued if the previous test did not reach significance. A P value of less than 0.05 was considered to be statistically significant. All analyses were performed with SAS, version 6.12 (SAS Institute, Cary, NC, USA).
Results
Twelve of the 68 patients studied were diagnosed with sepsis (8 patients with high‐flux membranes/4 patients with low flux membranes) and 24 patients with the following severe infections: nine with pneumonia (6/3), eight with endocarditis (7/1), three with pyelonephritis (2/1), two with invasive enterocolitis (2/0), and two with other infections (2/0). Characteristics of patients dialysed with high‐ or low‐flux membranes, including factors determining the clearance by HD, did not markedly differ, as shown in Table 1.
Patients with severe infections and sepsis had higher values for CRP and WBC than patients without infections: 11.2 mg/dl (7.1; 19.0) vs 4.7 mg/dl (2.2; 10.8), P<0.01 and 12.2×109/l (8.3; 14.1) vs 6.7×l09/l (4.5; 11.6).
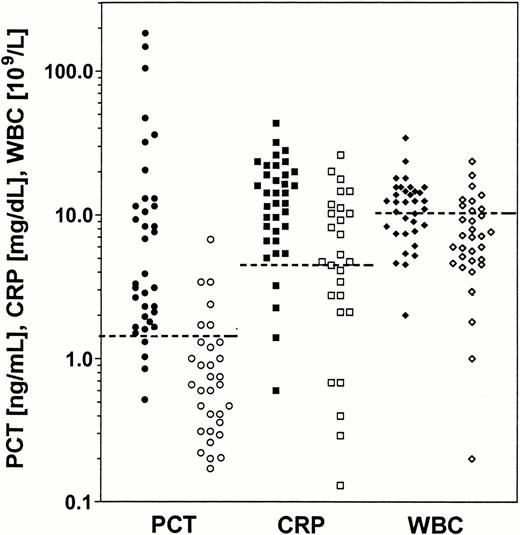
Serum PCT concentration of patients without infection was 0.8 ng/ml (0.3; 1.4) prior to the start of HD. Patients with severe infections and sepsis showed significantly higher PCT values prior to the start of HD (5.9 ng/ml (2.1; 11.5), P<0.01). Patients with and without infection were well discriminated by the PCT cut‐off value of 1.5 ng/ml, as shown in Figure 2. Thirty‐two of 36 patients were correctly classified by serum PCT concentrations greater than 1.5 ng/ml as having severe infections or sepsis. Discrimination with PCT was superior to that with CRP or WBC count at the respective cut‐off values (Figure 2). Serum PCT concentrations prior to the start of HD were sensitive, accurate, and predictive of severe infections and sepsis and superior to CRP or WBC in this respect (Table 2).
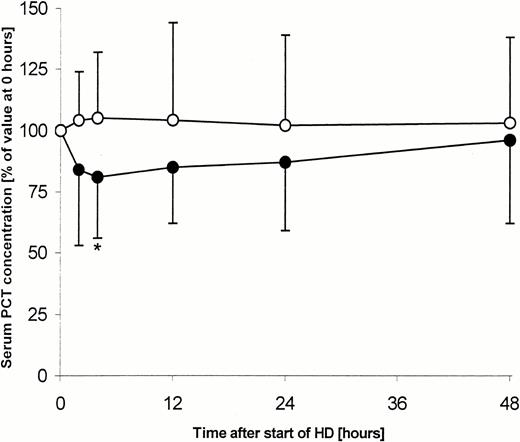
The time course of serum PCT concentrations over the 48‐h study period is illustrated in Figure 3. In all patients dialysed with high‐flux membranes, serum PCT concentrations were 2.69 ng/ml (0.98; 9.14) prior to the start of HD (T0). After 4 h of HD treatment, PCT concentrations decreased to 83±25% of the PCT concentrations prior to the start of HD (P<0.05) and increased only slowly thereafter. Forty‐eight hours after HD, prior to the next HD treatment, serum PCT concentrations had returned to T0 values. In all patients dialysed with low‐flux membranes, serum PCT concentrations were 1.62 ng/ml (0.47; 7.6) prior to the start of HD and did not decrease during the study period. Although we observed a small rise of PCT concentrations in these patients, this change was not significant.
In the subgroup of patients treated with high‐flux membranes sensitivity, negative predictive value and accuracy markedly decreased during and in the 24 h after HD. Sensitivity, negative predictive value, and accuracy of this subgroup reached their lowest values 4 h after the start of HD, with sensitivity falling from 91 to 65%, negative predictive value from 83 to 54%, and accuracy from 84 to 69%. In keeping with serum PCT concentrations, sensitivity, negative predictive value, and accuracy returned to baseline values 48 h after HD, with values of 87, 76 and 82% respectively. Specificity and positive predictive value of PCT remained unchanged for patients with high‐flux membranes during the study period.
Serum PCT concentration, serum CRP concentration, and WBC immediately prior to the start of HD in patients with severe infections and sepsis and in patients without infection. PCT, CRP and WBC values for patients with severe infections and sepsis are indicated by filled symbols (•, ▪, ◊), PCT, CRP and WBC values for patients without infection by open symbols (○, □, ⋄). The dashed lines indicate the cut‐off values for PCT of 1.5 ng/ml, for CRP of 5.0 mg/dl, and for WBC of 12.0×109/l.
Serum PCT concentration just prior to the start of HD (0), during HD (2 and 4 h) and in the 48 h after the start of HD (12, 24 and 48 h). Serum PCT of patients treated with high‐flux (•) or low‐flux (○) membranes is expressed as percentage of the respective PCT concentration (mean±SD) prior to the start of HD (*P<0.05).
Characteristics of patients dialysed with high‐flux and low‐flux membranes
| High‐flux membrane (n=47) | Low‐flux membrane (n=21) | |
| Age (years) | 42 (35; 57)a | 47 (33; 66)a |
| Female/male (n) | 21/26 | 8/13 |
| Duration of HD (h) | 4.5 (4; 5)a | 4.5 (4; 5)a |
| Blood flow rate (ml/min) | 230 (200; 240)a | 220 (200; 240)a |
| Ultrafiltration rate (ml/min) | 600 (500; 700)a | 600 (500; 700)a |
| High‐flux membrane (n=47) | Low‐flux membrane (n=21) | |
| Age (years) | 42 (35; 57)a | 47 (33; 66)a |
| Female/male (n) | 21/26 | 8/13 |
| Duration of HD (h) | 4.5 (4; 5)a | 4.5 (4; 5)a |
| Blood flow rate (ml/min) | 230 (200; 240)a | 220 (200; 240)a |
| Ultrafiltration rate (ml/min) | 600 (500; 700)a | 600 (500; 700)a |
aData are presented as median value with interquartile values in parenthesis.
Characteristics of patients dialysed with high‐flux and low‐flux membranes
| High‐flux membrane (n=47) | Low‐flux membrane (n=21) | |
| Age (years) | 42 (35; 57)a | 47 (33; 66)a |
| Female/male (n) | 21/26 | 8/13 |
| Duration of HD (h) | 4.5 (4; 5)a | 4.5 (4; 5)a |
| Blood flow rate (ml/min) | 230 (200; 240)a | 220 (200; 240)a |
| Ultrafiltration rate (ml/min) | 600 (500; 700)a | 600 (500; 700)a |
| High‐flux membrane (n=47) | Low‐flux membrane (n=21) | |
| Age (years) | 42 (35; 57)a | 47 (33; 66)a |
| Female/male (n) | 21/26 | 8/13 |
| Duration of HD (h) | 4.5 (4; 5)a | 4.5 (4; 5)a |
| Blood flow rate (ml/min) | 230 (200; 240)a | 220 (200; 240)a |
| Ultrafiltration rate (ml/min) | 600 (500; 700)a | 600 (500; 700)a |
aData are presented as median value with interquartile values in parenthesis.
Diagnostic performance of serum PCT compared to CRP and WBC indicating severe infections and sepsis, all measured immediately prior to the start of HD in all patients dialysed with high‐flux and low‐flux membranes
| Sensitivity (%) | Specificity (%) | Positive predictive value (%) | Negative predictive value (%) | Accuracy (%) | |
| PCT | 89 | 81 | 84 | 87 | 85 |
| CRP | 89 | 48 | 68 | 78 | 71 |
| WBC | 58 | 75 | 71 | 59 | 67 |
| Sensitivity (%) | Specificity (%) | Positive predictive value (%) | Negative predictive value (%) | Accuracy (%) | |
| PCT | 89 | 81 | 84 | 87 | 85 |
| CRP | 89 | 48 | 68 | 78 | 71 |
| WBC | 58 | 75 | 71 | 59 | 67 |
Diagnostic performance of serum PCT compared to CRP and WBC indicating severe infections and sepsis, all measured immediately prior to the start of HD in all patients dialysed with high‐flux and low‐flux membranes
| Sensitivity (%) | Specificity (%) | Positive predictive value (%) | Negative predictive value (%) | Accuracy (%) | |
| PCT | 89 | 81 | 84 | 87 | 85 |
| CRP | 89 | 48 | 68 | 78 | 71 |
| WBC | 58 | 75 | 71 | 59 | 67 |
| Sensitivity (%) | Specificity (%) | Positive predictive value (%) | Negative predictive value (%) | Accuracy (%) | |
| PCT | 89 | 81 | 84 | 87 | 85 |
| CRP | 89 | 48 | 68 | 78 | 71 |
| WBC | 58 | 75 | 71 | 59 | 67 |
Discussion
Serum PCT is an accurate indicator of severe infections and sepsis in patients requiring intermittent HD for ESRD and acute renal failure as demonstrated by this study. Serum PCT concentrations exceeding 1.5 ng/ml accurately indicate HD patients with severe infections and sepsis and discriminate them well from patients without infection. In addition, our results suggest that serum PCT has a higher diagnostic value indicating severe infections and sepsis than CRP or WBC. Unlike CRP and WBC, which may be unreliable due to non‐specific, HD and uraemia‐induced alterations [3,4,6,7], PCT can considerably aid in the accurate detection of severe infections and sepsis and thus impact on morbidity and mortality in HD patients.
The accuracy of PCT and its superior performance compared to CRP and WBC in HD patients indicating severe infections and sepsis are consistent with previous publications in patients with normal renal function [9–11,14–16]. In addition, PCT can distinguish between patients with and without severe infections following cardiac surgery with cardiopulmonary bypass, unlike CRP [31]. In this clinical circumstance even patients without infections demonstrate high acute‐phase responses, with markedly elevated CRP concentrations but only minor elevations of serum PCT. However, a transitory increase of PCT was also reported due to non‐infectious, post‐traumatic systemic inflammatory response syndrome and the increase correlated with severity of tissue injury [32]. In patients with normal renal function cut‐off values for serum PCT of 0.5 and 1.5 ng/ml have been proposed to discriminate between infection and non‐infection [9–12,16]. Our data indicate that a serum PCT concentration of 1.5 ng/ml is an appropriate cut‐off value in HD patients. A recent study on chronic haemodialysis patients supports this finding [33].
Our data show that high‐flux membranes significantly reduce serum PCT concentrations during and in the 24 h after the start of HD. Due to the long half‐life of PCT (25–30 h) [17,18], decreased synthesis or increased degradation of PCT were unlikely to cause or contribute significantly to the observed reduction of serum PCT. Neither mechanism should affect serum PCT concentrations earlier than 25 h after an initial PCT measurement. By that time, PCT had started to increase again from its nadir 4 h after the start of HD. Serum PCT is presumably eliminated by diffusive and convective transport across the dialysis membrane in parallel to β2‐microglobulin and other low‐molecular‐weight proteins [34]. In order to maximize sensitivity, accuracy, and predictive value, PCT should be measured only prior to the start of HD with high‐flux membranes, when PCT concentration is unaltered by HD. Since serum PCT concentrations from samples obtained after the start of HD with high‐flux membranes showed reduced accuracy, they should be interpreted with caution.
Serum PCT concentration remains unchanged in HD performed with low‐flux membranes. This is in parallel with other low‐molecular‐weight proteins, which are not eliminated by low‐flux membranes [34,35]. The fact that HD with low‐flux membranes does not result in a substantial increase of serum PCT may suggest that PCT synthesis is not induced by HD with polysulphone membranes, in contrast to cytokines such as tumour necrosis factor α, interleukin 1β and interleukin 6 [24,25].
In conclusion, serum PCT is an accurate indicator of severe infections and sepsis in patients receiving intermittent HD for ESRD and acute renal failure and is superior to CRP and WBC. Since HD with high‐flux membranes significantly decreases PCT concentration, serum PCT in patients treated with high‐flux membranes should be measured prior to the start of haemodialysis.
Correspondence and offprint requests to: Andreas Kribben MD, Abteilung für Nieren‐ und Hochdruckkrankheiten, Medizinische Klinik und Poliklinik, Universitätsklinikum Essen, D‐45122 Essen, Germany.
We thank Mrs Karin Kaiser for excellent measurement of serum PCT, Timothy A. Sutton, for helpful discussions, and the staff of the dialysis unit at the University Hospital Essen for their help and collaboration in the study. The kits for measurement of PCT were kindly provided by Brahms Diagnostica Corp. (Berlin, Germany).
References
Khan IN, Catto GRD. Long‐term complications of dialysis: infection.
Rinehart A, Collins AJ, Keane WF. Host defense and infectious complications in maintenance hemodialysis patients. In: Jacobs C, Kjellstrand CM, Koch KM, Winchester JF, eds.
Hakim RM, Lowrie EG. Hemodialysis‐associated neutropenia and hypoxemia: the effect of dialyzer membrane materials.
Hoenich NA, Johnston SRD, Woffindin C, Kerr DNS. Haemodialysis leucopenia: the role of membrane type and reuse.
Bathon J, Graves, Jens P, Hamrick R, Mayes M. The erythrocyte sedimentation rate in end‐stage renal failure.
Haubitz M, Schulze M, Koch KM. Increase of C‐reactive protein serum values following haemodialysis.
Owen WF, Lowrie EG. C‐reactive protein as an outcome predictor for maintenance hemodialysis patients.
Honkanen E, Groenhagen‐Riska C, Teppo AM, Maury CPJ, Meri S. Acute‐phase proteins during hemodialysis: correlations with serum interleukin‐1β levels and different dialysis membranes.
AI‐Nawas B, Krammer I, Shah PM. Procalcitonin in diagnosis of severe infections.
Assicot M, Gendrel D, Carsin H, Raymond J, Guilbaud J, Bohuon C. High serum procalcitonin concentrations in patients with sepsis and infection.
Rau B, Steinbach G, Gansauge F, Mayer JM, Grunert A, Beger HG. The potential role of procalcitonin and interleukin 8 in the prediction of infected necrosis in acute pancreatitis.
Eberhard OK, Haubitz M, Brunkhorst FM, Kliem V, Koch KM, Brunkhorst R. Usefulness of procalcitonin for differentiation between activity of systemic autoimmune disease and invasive bacterial infection.
Eberhard OK, Langefeld I, Kuse ER et al. Procalcitonin in the early phase after renal transplantation—will it add to diagnostic accuracy.
Monneret G, Doche C, Duran DV, Lepape A, Bienvenu J. Procalcitonin as a specific marker of bacterial infection in adults.
Whang KT, Steinwald PM, White JC et al. Serum calcitonin precursors in sepsis and systemic inflammation.
De Werra I, Jaccard C, Corradin SB et al. Cytokines, nitrite/ nitrate, soluble tumor necrosis factor receptors, and procalcitonin concentrations: comparisons in patients with septic shock, cardiogenic shock, and bacterial pneumonia.
Brunkhorst FM, Heinz U, Forycki ZF. Kinetics of procalcitonin in iatrogenic sepsis.
Dandona P, Nix D, Wilson MF et al. Procalcitonin increase after endotoxin injection in normal subjects.
Nylen ES, Whang KT, Snider RH, Steinwald PM, White JC, Becker KL. Mortality is increased by procalcitonin and decreased by an antiserum reactive to procalcitonin in experimental sepsis.
Oberhoffer M, Stonans I, Russwurm S et al. Procalcitonin expression in human peripheral blood mononuclear cells and its modulation by lipopolysaccharides and sepsis‐related cytokines in vitro.
Jacobs JW, Lund PK, Potts JT, Bell NH, Habener JF. Procalcitonin is a glycoprotein.
Herbelin A, Nguyen AT, Zingraff J, Urena P, Deschamps‐Latscha B. Influence of uremia and hemodialysis on circulating interleukin‐1 and tumor necrosis factor α.
Herbelin A, Urena P, Nguyen AT, Zingraff J, Deschamps‐Latscha B. Elevated circulating levels of interleukin‐6 in patients with chronic renal failure.
Meisner M, Tschaikowsky K, Hutzler A, Schick C, Schuttler J. Postoperative plasma concentrations of procalcitonin after different types of surgery.
Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings.
Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM. CDC definitions for nosocomial infections.
American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis.
Bergstroem J, Wehle B. No change in corrected β2‐microglobulin concentration after cuprophane haemodialysis.
Povoa P, Almeida E, Moreira P et al. C‐reactive protein as an indicator of sepsis.
Smith RP, Lipworth BJ, Cree IA, Spiers EM, Winter JH. C‐reactive protein. A clinical marker in community‐acquired pneumonia.
Rothenburger M, Markewitz A, Lenz A et al. Detection of acute phase response and infection. The role of procalcitonin and C‐reactive protein.
Mimoz O, Benoist JF, Edouard AR, Assicot M, Bohuon C, Samii K. Procalcitonin and C‐reactive protein during the early posttraumatic systemic inflammatory response syndrome.
Schmidt M, Burchardi C, Sitter T, Held E, Schiffl H. Procalcitonin in patients undergoing chronic hemodialysis.
Hoenich NA, Woffindin C, Matthews JNS, Goldfinch ME, Turnbull J. Clinical comparison of high‐flux cellulose acetate and synthetic membranes.





Comments