-
PDF
- Split View
-
Views
-
Cite
Cite
Kei Matsushita, Kiyotaka Nagahama, Yoshiaki Inayama, Katsumichi Fujimaki, Kouichi Tamura, Nobuhito Hirawa, Minoru Kihara, Yoshiyuki Toya, Machiko Yabana, Kensuke Joh, Satoshi Umemura, Lobular membranoproliferative glomerulonephritis with organized microtubular monoclonal immunoglobulin deposits associated with B cell small lymphocytic lymphoma, Nephrology Dialysis Transplantation, Volume 20, Issue 6, June 2005, Pages 1273–1274, https://doi.org/10.1093/ndt/gfh838
Close - Share Icon Share
Sir,
Fibrillary glomerulonephritis (FGN) and immunotactoid glomerulopathy (ITG) are well known glomerular diseases with Congo-red-negative deposits [1–4]. While FGN is characterized by randomly arranged microfibrils around 20 nm in diameter with no underlying systemic disorder, ITG is defined by orderly arranged microtubular deposits, usually >30 nm in diameter with a hollow core; patients with ITG tend to have underlying lymphoproliferative diseases.
Microtubular monoclonal immunoglobulin deposits with a diameter smaller than that commonly observed in ITG have been described in a few patients with lymphoproliferative disorders, and Touchard et al. coined the term ‘GOMMID’ for such glomerulonephritis with organized microtubular monoclonal immunoglobulin deposits [5,6]. We report here a patient with nephrotic syndrome due to GOMMID following B-cell malignant lymphoma associated with IgG-κ monoclonal gammopathy. Interestingly, although the patient developed AL amyloid deposition (κ type) in a cervical lymph node, no amyloid was recognized in the kidney.
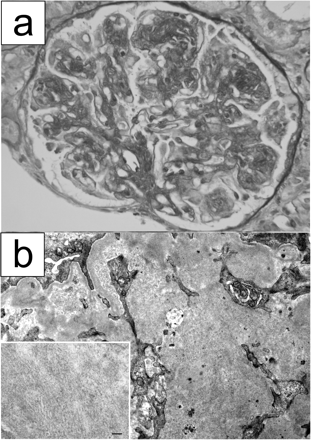
Case. A 46-year-old Japanese man presented with lymph node swelling and IgG-κ monoclonal gammopathy. The lymph node biopsy revealed small lymphocytic lymphoma cell infiltration with amyloid deposition in the vessel walls and interstitium. The lymphoma cells were positive for CD5, CD20 and CD23, and κ light chains were detected in the cytoplasm of plasma cell-differentiated lymphoma cells. Amyloid deposits were also positive for κ light chains. He developed nephrotic syndrome 5 months later from the lymph node biopsy. Serum immunoglobulin concentrations at renal biopsy were as follows: IgG was 1410 mg/dl, IgA was 93 mg/dl and IgM was 51 mg/dl. Although serum cryoglobulin was weakly positive, its type was not determined due to insufficient concentration. Renal biopsy was performed and light microscopic studies revealed membranoproliferative glomerulonephritis with lobular accentuation (Figure 1a). Congo red staining was negative in the renal biopsy specimens. Immunofluorescence microscopic studies demonstrated positive staining for IgG, C3 and κ light chain in the mesangium and glomerular capillaries, and IgG1 was dominantly positive among the IgG subclasses. Electron microscopy showed expanded mesangial areas containing orderly arranged microtubular deposits with an average external diameter of 15 nm (Figure 1b).
Pathological findings of renal biopsy specimens. (a) Light microscopy. A glomerulus showing membranoproliferative glomerulonephritis with lobular accentuation. Periodic acid–Schiff, original magnification ×400. (b) Electron microscopic examination. Marked mesangial widening is noted with microtubular deposits (original magnification ×10 000). Inset: high power view of microtubular deposits in the mesangial area. Bar = 100 nm (original magnification ×30 000).
Discussion. GOMMID was proposed to include the following criteria: (i) the presence of organized microtubular deposits (15 nm in width) in glomeruli without thrombi in the glomerular capillaries and/or renal arterioles; and (ii) monotypic immunoglobulin deposits, irrespective of the presence of detectable monoclonal immunoglobulin in patient serum and/or urine, type I cryoglobulinaemia or immunoproliferative disorder [5]. Our case satisfies these criteria and features both pathological and clinical aspects of GOMMID.
The relationship between malignant lymphoma and GOMMID is not fully understood. Touchard et al. [5] showed that leukaemia/lymphoma cells were the major source of glomerular immunoglobulin deposits from immunofluorescence and electron microscopic studies. In the present case, although we failed to observe lymphoma cells with electron microscopy, their role was strongly suggested by the correspondence of the isotype of monotypic immunoglobulin deposited in the glomeruli with that of the serum paraprotein. On the other hand, the different patterns of Congo red staining, positive in the lymph node and negative in the glomeruli, were remarkable in this case. It would be suggested that each immunoglobulin deposition was related to the production of amyloid- or non-amyloid-forming κ light chains derived from different plasma cell clones. Diversity of the tissue microenvironment might also explain such different patterns of immunoglobulin deposition [7].
Conflict of interest statement. None declared.
References
Korbet SM, Schwartz MM, Rosenberg BF, Sibley RK, Lewis EJ. Immunotactoid glomerulopathy.
Alpers CE. Immunotactoid (microtubular) glomerulopathy: an entity distinct from fibrillary glomerulonephritis?
Bridoux F, Hugue V, Coldefy O et al. Fibrillary glomerulonephritis and immunotactoid (microtubular) glomerulopathy are associated with distinct immunologic features.
Rosenstock JL, Markowitz GS, Valeri AM, Sacchi G, Appel GB, D’Agati VD. Fibrillary and immunotactoid glomerulonephritis: distinct entities with different clinical and pathologic features.
Touchard G, Bauwens M, Goujon JM, Aucouturier P, Patte D, Preud’homme JL. Glomerulonephritis with organized microtubular monoclonal immunoglobulin deposits.
Ronco PM. Paraneoplastic glomerulopathies: new insights into an old entity.
Author notes
1Department of Second Internal Medicine2 Department of Pathology3 Department of First Internal Medicine Yokohama City University School of Medicine Kanagawa4 Clinical Research Center Chiba-East National Hospital Chiba Japan Email: umemuras@med.yokohama-cu.ac.jp





Comments