-
PDF
- Split View
-
Views
-
Cite
Cite
Itziar Abete, Arne Astrup, J Alfredo Martínez, Inga Thorsdottir, Maria A Zulet, Obesity and the metabolic syndrome: role of different dietary macronutrient distribution patterns and specific nutritional components on weight loss and maintenance, Nutrition Reviews, Volume 68, Issue 4, 1 April 2010, Pages 214–231, https://doi.org/10.1111/j.1753-4887.2010.00280.x
Close - Share Icon Share
Abstract
Weight loss and subsequent body weight maintenance are difficult for obese individuals despite the wide variety of dietary regimens and approaches. A substantial body of scientific evidence has shown that by simply varying the macronutrient distribution and composition of dietary factors, weight losses of varying amounts, longer-term body weight maintenance periods, better appetite regulation, and changes in features of the metabolic syndrome can be achieved. At present, renewed efforts are underway to increase the protein content of weight-loss diets, simultaneously restrict fat consumption to no more than 30%, favor polyunsaturated fat, have carbohydrates account for between 40 and 50% of total energy intake, and promote the consumption of low-glycemic foods. The present article reviews the scientific evidence for the effects of several dietary manipulations and sustainable strategies for weight loss and body weight stability as well as for treating specific features of the metabolic syndrome.
INTRODUCTION
An excess amount of body fat not only leads to reduced quality of life and large healthcare-associated costs, it also increases the risk of death.1 Indeed, obesity has been related to a number of cardiovascular and metabolic disorders such as hypertension, type 2 diabetes, hyperinsulinemia, dyslipidemia, atherosclerosis, and even certain types of cancers.2,3 In addition to genetic predisposition, an inactive lifestyle and high caloric intake are the overriding causes of excessive weight gain.4,5 Thus, lifestyle changes affecting dietary habits and physical activity are essential to promote weight loss.6,–8 For decades, nutritional intervention studies have been focused on reducing dietary fat with no positive results over long-term periods.9,10 The carbohydrate content of diets was also reduced in some populations, but obesity prevalence continued to increase.11
In diets designed to prevent and treat obesity by manipulating energy content, macronutrient distribution was commonly set at 15% protein, <30% lipids, and 50–55% carbohydrates, with reductions in dietary fat and increases in fiber favored. Although this recommendation seemed to be effective for decreasing energy density and promoting weight loss in the short term, the low level of observed satiety it achieved decreased dietary adherence over longer periods.5,11 Monitoring of low-energy diets with these macronutrient proportions revealed that they were often not sustainable for long periods of time.12
Likewise, low-carbohydrate diets (low-CHO) have often been recommended as effective tools for weight loss over short-term periods, but their long-term effects have not been fully established.13 Moreover, the higher saturated fat intake of these diets may increase LDL-cholesterol levels14 and have other adverse effects.15 Other clinical intervention programs have studied different nutritional treatments in order to improve both weight loss and weight maintenance or associated metabolic related disturbances. These treatments include the following: varying macronutrient composition,13,–18 incorporating bioactive ingredients such as fiber19or flavonoids,20 manipulating the glycemic index (GI)21,22 on the omega-3 fatty acid profile,23 and manipulating the composition of minerals such as calcium24 and selenium.25 Studies with low-GI diets have shown a number of favorable effects such as rapid weight loss, better management of glucose and insulin levels, and reductions in triglyceride levels and blood pressure.5,–27
On the other hand, current evidence indicates that diets moderately high in protein and modestly restricted in carbohydrate and fat may have more beneficial effects on body weight homeostasis and associated metabolic parameters.28,–32 Likewise, the increase of dietary omega-3 fatty acids (FAs) may be an important factor in weight loss and weight maintenance approaches affecting satiety regulation.33
Nutritional intervention studies are presently focused on decreasing hunger and promoting satiety in an effort to improve adherence and make weight loss and weight maintenance easier.32,–34 Thus, diets designed to achieve weight loss via a moderately high protein content, low-GI carbohydrates, and specific omega-3 FAs may have beneficial effects on satiety and compliance contributing to weight loss and weight control, which may be an alternative to conventional diets.35,36
The current article reviews the effects of different weight-loss diets on body weight, metabolic markers, and body weight maintenance with a particular emphasis on moderate-protein diets (30–35% energy at the expense of carbohydrates), those with low-GI carbohydrates (within the 40% energy), and diets in which omega-3 FA intake is high at the expense of saturated FAs (within 30% of total fat).
WEIGHT-LOWERING APPROACHES
Ad libitum versus energy-restricted diets
The role of specific foods and nutrients in the prevention and treatment of obesity, as well as in associated complications, has become a major focus of concern in nutritional research.37,38 Dietary strategies to reduce fat mass have changed over time with variations seen in macronutrient composition, fiber content, and bioactive food ingredients; however, there is still an intense debate over which, if any, dietary regime might be the most beneficial for weight loss and long-term weight maintenance.39
Control of food intake is one of the most important factors putatively involved in the success of dietary treatment of obesity due to its relation with energy balance.33 The regulation of appetite as well as feelings of hunger and satiety is a determinant factor affecting adherence to a weight-loss program. Thus, nutritional programs have modified the type of energy restriction to improve compliance over long-term periods. Several decades ago most dietary strategies were energy restricted, but a number of currently available weight-loss diets are designed to be ad libitum. Energy restriction has consistently been proven to produce weight loss and to have beneficial health effects. However, most individuals are unable to sustain weight losses induced by reducing energy intake, potentially because of increased hunger levels9 and energy expenditure adaptations.40 Moreover, the dietary monotony and lack of variability make compliance difficult during extended periods.
Conversely, ad libitum programs with variety in the macronutrient composition reduce the patient's feeling of being restricted to a diet, which could improve their adherence and weight loss. Ad libitum low-fat diets, low-CHO diets, and high-protein diets have all been proposed as tools for facilitating weight lowering and stability thereafter.22,41 Indeed, a few long-term trials have demonstrated better results in weight loss and weight maintenance with ad libitum programs. Some studies based on low-CHO intake strategies, lasting up to 12 months and performed in obese subjects with no energy restriction, resulted in more weight loss than a conventional energy-restricted diet.42,43 The CARMEN trial also investigated the long-term effect of ad libitum, low-fat, high-CHO diets containing simple or complex carbohydrates on body weight in comparison with a control diet group with normal macronutrient intake. After 6 months, they found it was favorable to replace dietary fat with carbohydrates (simple or complex) for the purpose of body weight regulation, since those patients following the low-fat high-CHO (simple or complex) diets registered greater losses of weight and fat mass than patients in the control group.44,45
Thus, adherence to slimming programs and maintenance of achieved body weight reduction are challenging aspects of weight-loss strategies.46 Different nutritional intervention studies considering both energy-restricted and ad libitum approaches have been reported to differentially affect thermogenesis, energy expenditure, satiety, hormonal balance, and other factors that would not only reduce energy intake, but improve compliance, weight loss, and weight maintenance.11
Meal replacements
The adoption of healthy dietary patterns to facilitate weight loss, cardiovascular disease risk reduction, and weight management is often really difficult in an obesogenic environment in which palatable, inexpensive, and high-fat foods are easily available. Thus, alternative dietary strategies are being studied to facilitate adherence to healthy dietary habits. To this end, a number of investigations have suggested that meal-replacement products may represent an important strategy for combating the worldwide epidemic of obesity due to their simplicity and convenience.11,–49 Meal replacements may simplify the weight-loss regimens by replacing one or two meals a day with a product of defined nutrient and calorie content. Compared with the simple provision of food plans, a weight-loss program based on meal replacements for one or two meals per day has been shown to improve compliance with a calorie-restricted diet as well as weight management in overweight and obese individuals.47,–50 The composition of the meal replacement is also of interest. In this sense, moderate- or high-protein meal replacements may increase feelings of satiety and may favor the reduction of energy intake in subsequent meals.51 Accordingly, one nutritional intervention study showed that a relatively high-protein meal (25% proteins; 45% carbohydrates; 30% fats) was more satiating and had a higher thermogenic effect than a lower protein meal (10% proteins; 60% carbohydrates; 30% fats).52 Several health professionals currently recommend the use of meal-replacement products, and these products are used increasingly in weight-loss studies Meal-replacement products may prove very useful for facilitating compliance with weight-reduction programs and weight management, at least in the short term.
Macronutrient distribution
Low-calorie diets can achieve acute weight loss, but often the slimming result is not maintained long term.53 A number of nutritional approaches and diets with different proportions of lipids, proteins, and carbohydrates are currently being investigated. There is intense controversy about the types of diets and macronutrient distributions that are most effective for treating overweight or obesity. Many trials investigating the macronutrient composition of diets have been carried out for years to improve both the weight loss and the weight maintenance process. One recent nutritional intervention study compared the effect of four weight-loss diets with different compositions of fat, protein, and carbohydrates.54 The investigators assessed the change in body weight after 2 years, comparing diets with low fat versus high fat, average protein versus high protein, and the highest and lowest carbohydrate content. At 6 months, participants had lost 7% of their initial body weight with no group differences; however, all groups began to regain weight after 12 months. After 2 years, differences in weight loss were not apparent among the dietary groups. The authors also concluded there was no major macronutrient influence on weight loss. Nevertheless, compliance with dietary goals (whether high protein or low fat) was associated with improved weight loss.54,55
Low-fat diets. An alternative to calorie restriction is to decrease the fat content of the diet. In this context, a multicenter intervention trial investigated whether a hypoenergetic low-fat diet was superior to a hypoenergetic moderate high-fat diet for the treatment of obesity. After a 10-week weight-loss period, the low-fat diet produced similar mean weight loss to the high-fat diet, but it resulted in more subjects losing >10% of their initial body weight and fewer dropouts.56 However, the long-term effects of fat-restricted diets on weight loss have not been established. In a literature review by Summerbell et al.12 the authors investigated low-fat diets for obesity and concluded that fat-restricted diets are no better than calorie-restricted diets for achieving stable weight loss in overweight or obese people.
Another recent nutritional intervention trial compared the effects of three ad libitum diets on weight maintenance after an initial weight loss of ≥8%. The diets had different dietary contents, including carbohydrates and types of fat (monounsaturated fatty acid [MUFA] diet with 35–45% fat and >20% MUFA content; low-fat diet with 20–30% fat; or control diet with 35% fat), but the protein content was similar (10–20%). After 6 months, dietary composition had no major effect on preventing weight regain; however, both the MUFA diet and the low-fat diet resulted in less body fat regain than the control diet.57
Overall, the evidence supports that low-fat diets are an optimal choice for the prevention of weight gain and obesity. A reduction in the percentage of energy consumed as fat is positively associated with weight loss. Indeed, a 10% reduction in dietary fat is predicted to produce a 4–5 kg weight loss in an individual with a BMI of 30 kg/m2.58 However, future research needs to focus on improving and maintaining adherence to the treatments used to promote weight loss and maintenance.
Low-carbohydrate diets. Low-CHO/high-fat diets have been compared with low-fat energy-restricted diets. This nutritional strategy becomes an alternative to a low-fat diet for producing weight loss in short periods of time. Several reports indicate low-CHO diets can be favored for their proven effectiveness in producing rapid weight losses and beneficial metabolic changes.13,59 However, the lack of long-term studies and reports of some undesirable effects, such as increased levels of ketone bodies, high losses of body water, headache, constipation, and, especially, difficulties maintaining weight loss after the diet, make it difficult to recommend these diets as a healthy option for weight loss. Nevertheless, a recent intervention study evaluated the effect of a low-CHO high-fat diet for 1 year. The reported results showed that a low-CHO diet was effective for weight loss, did not induce deleterious metabolic effects, and did not increase risk for cardiovascular disease.13 Another nutritional study showed that a low-CHO ketogenic diet produced greater weight loss than a low-GI diet and led to greater improvements in glycemic control independent of weight reduction.59 However, most dietary studies have a relatively low duration (less than 1 year) and focus on weight reduction efficacy. Considerably less information is available regarding the potential clinical impact of very-low-CHO diets after weight has been stabilized. Indeed, results presented in one recent study suggest that during weight maintenance, following a high-fat low-CHO diet (Atkins diet) was associated with higher total cholesterol and LDL-cholesterol, and the high saturated fat intake was inversely correlated with endothelial function.60 It has also been suggested that very-low-CHO diets are not compatible with a healthy and palatable diet.11
On the other hand, a diet with moderate protein content (30%) at the expense of carbohydrates (40%) achieves a higher satiating effect than conventional energy-restricted diets and induces spontaneous weight loss.11 In this context, Skov et al.31 showed that diets with a fat content of 30% of calories produced greater weight loss when high in protein (25% of calories; −9.4 kg) than when normal in protein (12% of calories; −5.9 kg) after 6 months of treatment. Moreover, after 1 year of follow up, the protein diet reduced visceral fat to a greater degree than found from the loss of fat.31
A number of studies have suggested that protein is the most important macronutrient mediating satiety and leads to increased weight loss with retention of lean body mass.32,61 Thus, the satiating effect of moderate-protein diets could be a key factor in body weight loss programs and in body weight stability.
Moderate-protein-content (30%) diets. The popularity of moderately high-protein diets with an emphasis on some degree of carbohydrate restriction is increasing. Controlled energy intake in combination with a moderately elevated protein intake may represent an effective and practical weight-loss strategy.28
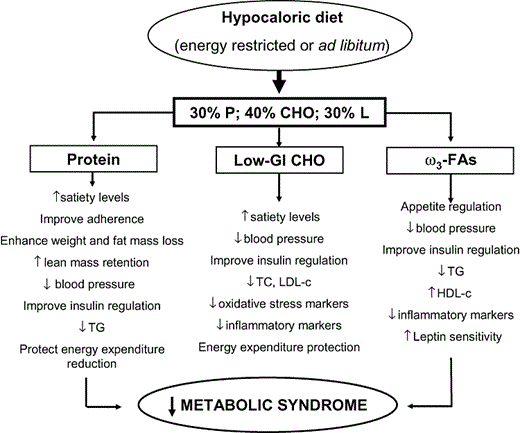
Potentially beneficial effects associated with moderate-protein diets have been reported (Figure 1) since protein has been observed to increase satiety to a greater extent than carbohydrate and fat, which may facilitate a reduction in energy consumption under ad libitum dietary conditions.31 It has also been shown62 that dietary protein has the highest and most prolonged thermic effect of separate macronutrients (20–30%), followed by carbohydrate (5–15%) and fat (0–3%). Indeed, higher-protein diets are not only associated with increased thermogenesis, they also influence satiety and enhance energy expenditure.34 Finally, for the maintenance of fat-free mass, in some individuals, a moderately high-protein diet may provide a stimulatory effect on muscle protein anabolism, favoring the retention of lean muscle mass while improving the metabolic profile.34
Metabolic changes that could be achieved with an energy-restricted or ad libitum diet combining moderate protein content with low glycemic index carbohydrates and high omega-3 fatty acids intake.
Several intervention studies have been conducted with diets relatively high in protein and the results showed positive effects and better results pertaining to weight loss and weight maintenance than normal-protein diets. Thus, with identical fat intake, a diet with a higher protein intake exchanged with carbohydrate, compared to an adequate protein intake, increases 24-h satiety, thermogenesis, sleeping energy expenditure, protein balance, and fat oxidation.63 Another recent multicenter weight-loss trial compared changes in body weight and composition after short-term weight loss (4 months), which was followed by a weight maintenance follow-up period (8 months) using a moderate-protein diet (30% of energy from protein, 40% carbohydrates, and 30% fat) or a conventional high-CHO diet (15% protein, 55% carbohydrates, and 30% fat); the results showed that the 30% protein diet was more effective for fat mass loss and body composition improvement during the initial period of weight loss and during long-term maintenance.64 In addition to the beneficial effects of the moderate-protein diet for changes in body composition, more participants completed the study in the moderate-protein group (64%) than in the high-CHO group (45%). These findings demonstrate there was greater compliance with a moderate-protein diet for long-term weight management than with the high-CHO diets often advocated for weight loss.65
Reduction of fat mass and maintenance of lean mass are critical factors for long-term health status and the prevention of weight regain.64 Evidence is accumulating that the recommended daily allowance for protein is inadequate to maintain muscle mass in adults during energy restriction for weight loss.66 The current recommendation for protein represents the minimum protein needs for healthy young adults with adequate energy intakes.67,68 During weight lowering, energy restriction increases the amount of protein required to maintain muscle mass.66 Thus, moderate-protein diets (30–35%) could be one of the most indicated strategies for weight loss and weight management, with the caveat that they should not be used by subjects with renal diseases.
Energy deficit is the key factor for weight loss, but the macronutrient composition influences changes in body composition and long-term compliance. In this context, more studies are needed to establish higher protein recommendations during periods of energy restriction in order to improve weight loss and long-term weight maintenance.
Other nutritional factors
Glycemic index. The glycemic index (GI) of foods is considered an important dietary factor that can influence body weight control.5 Although there are different conclusions for the efficacy of low-GI diets in body weight loss, appetite suppression, and reduction of metabolic rate, it is also known that many starchy foods, refined grain products, and potatoes produce high glycemic responses that elicit a sequence of hormonal changes that alter fuel partitioning and cause overeating.69 Studies in animal models have shown that diets based on high-GI starches promote weight gain, visceral adiposity, and higher activities of lipogenic enzymes than do counterpart diets based on low-GI foods.70,–72 On the contrary, diets based on low-GI foods may enhance weight control because they promote satiety, minimize postprandial insulin secretion, and maintain insulin sensitivity.17,–75 Several intervention studies have been performed in humans in which energy-restricted diets based on low-GI foods produced greater weight loss than comparable diets based on high-GI foods.21,–76
Data from clinical trials suggest that low-GI diets, based on high amounts of fruits, vegetables, legumes, and whole grains, are better than conventional diets for weight loss,26,77 even though some smaller trials may have been underpowered to detect the weight loss produced by the low-GI diet.78 Low-GI diets are also characterized by high fiber content79 and have been shown to delay hunger and decrease subsequent energy intake compared with high-GI diets.80 A recent dietary intervention trial showed that the patients who followed a low-GI diet during an 8-week energy-restricted period registered higher weight loss (7% of the initial body weight) than those included in a conventional diet (5% of the initial body weight) and the reduction in body weight was directly associated with fiber intake.21 Moreover, in studies comparing ad libitum low-GI diets to conventional energy-restricted low-fat diets, participants fared as well or better on the low-GI diet, even though they could eat as much as they desired.45,80 Thus, studies evaluating the effect of eating a high- or low-GI meal showed that voluntary energy intake after a high-GI meal was greater than after a low-GI meal.81
In addition to the higher satiety effect, benefits related to body composition, and improved insulin regulation, it has been shown that low-GI diets also produce favorable metabolic changes. In one study, patients following a low-GI diet lost less lean mass and showed a reduced decrease in resting metabolic rate, which could help improve weight loss as well as weight management.21 Indeed, the amount of weight regained 1 year after the end of the weight-loss program was lower in the low-GI-diet group. These findings are in accordance with previous studies, which also suggested that a low-GI-based diet may improve weight loss and weight management, minimizing negative metabolic adaptations during a period of energy restriction.26,27 However, controversy remains about the use of low-GI diets, since evidence from intervention studies using a low-GI approach for weight loss have produced inconsistent results, especially in longer-term studies. In intervention studies involving patients with type 2 diabetes, consumption of a low-GI diet resulted in lower hemoglobin A1c concentrations in shorter-term studies. However, as reported in a recent review of the topic, other evidence exists that adds to the controversy regarding the effectiveness of consuming low-GI diets for glycemic control and weight reduction.82 Moreover, there are limitations to how well the use of GI tables can predict the GI composition of a diet, leaving the applicability of the GI in the context of mixed meals and diets debatable.9,83
Fatty acids. In much of the current dietary advice “good” fats such as MUFAs and omega-3 FAs are emphasized, while saturated and trans fats are restricted. Olive oil, almonds, avocados, and nuts, for example, are considered important sources of MUFAs. Several studies have evaluated the effects of these fats on weight loss and weight management. Recently, Sloth et al.84 compared three different weight-maintenance diets (a moderate-fat 35–45% of energy from fat diet with high MUFA content; a low-fat diet with 20–30% energy from fat, or a control diet with 35% energy from fat) on appetite, glucose, and fat metabolism for 6 months after a weight-loss period. These authors did not find differences in appetite, energy intake, or body weight after the weight-maintenance period, but the MUFA and low-fat diets were equally effective at managing glucose metabolism, while the control diet was associated with decreased insulin sensitivity. Another recent study compared the effectiveness of energy-restricted high-MUFA and high-CHO diets on anthropometric and metabolic parameters specifically in diabetic individuals for 1 year; the results suggested that high-MUFA diets can be healthy alternatives to conventional lower-fat diets.85 Keogh et al.86 studied the effects of a low-CHO diet with a high monounsaturated fat content (50% fat; 20% protein; 30% carbohydrates) in relation to a relative high-protein, moderate-fat (30% protein; 30% fat; 40% carbohydrates) diet. However, limited subject compliance with the dietary patterns did not allow a long-term assessment of both diets. Due et al.57 compared the effect of a diet providing a moderate amount of fat (35–45% of energy) and >20% of fat as MUFAs with a low-fat diet (20–30% of energy) and a control diet (30% of energy as fat) on weight management during a 6-month controlled dietary intervention after an initial weight loss of 8%. Protein constituted 10–20% of energy in all three diets. At the end of the study the researchers found that dietary composition had no major effects on preventing weight regain. Both the low-fat and the high-MUFA diets produced less body fat regain than did the control diet. The high-MUFA diet was more beneficial for decreasing fasting insulin levels and improving insulin resistance as well as the LDL-cholesterol/HDL-cholesterol ratio.
Intake of fish and fish oil is considered an important part of a healthy diet, since they are rich sources of omega-3 FAs. Regular consumption of fish or fish oils is known to have pleiotropic effects, including effects against inflammation, platelet aggregation, hypertension, and hyperlipidemia.87,–89 These beneficial effects may be mediated through several distinct mechanisms, including alterations in cell membrane composition and function, gene expression modulation, or eicosanoid production.90 In addition to these beneficial effects, it was suggested that the inclusion of fish in dietary strategies could improve weight reduction. In this sense, one study showed that the substitution of fish oil for visible fats in a control diet (52% carbohydrates, 16% protein, 32% fat) reduced body fat mass and stimulated lipid oxidation in healthy adults.91 Also, a recent nutritional intervention program showed that the patients who participated in a weight-reduction regimen (–30% energy restriction) with fish consumption (150 g of cod, 5 days/week) for 8 weeks showed greater weight loss in comparison with the control group.92 However, the most important effects of omega-3 FAs provided as dietary fish have been described as improvements in cardiometabolic changes,23 inflammation,93 and oxidative stress.94 In addition, omega-3 FAs from fish consumption have been considered as nutritional factors that may influence appetite and satiety perception,33,95 two very important factors during an energy-restriction or weight-maintenance program. A nutritional intervention study showed that omega-3 FAs provided as dietary fish were able to modulate postprandial satiety in overweight and obese volunteers during weight loss.33
One observational study suggested that diet-derived omega-3 FAs lower leptin levels independent of body fat.96 High leptin levels have been related with features of the metabolic syndrome as well as with the presence of leptin resistance in obesity.97 Since leptin is involved in the regulation of energy expenditure and energy intake, a decrease in plasma leptin levels may involve an improvement in the control of its functions, favoring greater dietary intake control as well as better weight maintenance. Conversely, other authors suggest the process of weight loss may produce a state of leptin insufficiency, which may produce deleterious effects on energy metabolism that may hinder weight loss maintenance. In this sense, the administration of a low dose of leptin was found to be effective for reversing the energy expenditure decrease to pre-weight-loss levels, which could be a valid strategy for preventing weight regain.98
Overall, Mori et al.99 have shown that weight loss in combination with a daily serving of fish had additive effects on blood pressure reduction, improved heart rate, serum lipids, and glucose and insulin metabolism, leading to greater benefits than those achieved with a weight-loss intervention alone. Later, they also found that dietary fish enhanced the effects of weight loss on serum leptin levels.97 In agreement with this finding, a clinical trial that evaluated the effects of fish consumption during a period of energy restriction found a specific leptin and insulin decrease that was independent of body fat mass reduction.23 This study also suggested an improvement in the control of leptin and insulin functions, which could involve better body weight regulation after a nutritional intervention.
The health benefits of consuming fatty fish, fish oils, or foods rich in omega-3 FAs by persons with cardiovascular disease have led to recommendations to increase fish consumption.100,101 However, little fish is consumed in many Western societies and an increase in the consumption of omega-3 FAs would involve major dietary changes. In this sense, supplements as well as enriched foods may be considered. Thus, fatty fish, fish oils, omega-3 FA-rich foods, and omega-3 FA supplements could be included in weight loss and weight maintenance programs as well as be incorporated into the dietary habits of healthy subjects.
Micronutrients or other dietary components. Modest weight loss is possible for most subjects; however, very few achieve stable management of the body weight lost. Most studies of individuals who participated in a weight-lowering program show that the subjects regain their initial body weight, indicating that individuals did not change their eating behavior adequately.
A large number of new interventions are being designed in order to improve weight lowering and long-term weight maintenance. Because of this trend, the micronutrient composition of weight-loss diets is currently considered an important factor to consider in addition to the more traditional emphasis on macronutrient distribution. Indeed, several putative bioactive food ingredients, such as minerals and other dietary or herbal components could affect satiety, thermogenesis, energy balance, and other factors that would improve adherence to slimming programs, reduce energy intake, and favor weight loss and weight maintenance. Thus, a number of nutritional intervention studies with diets supplemented or enriched with different components have shown a variety of results regarding weight loss as well as weight maintenance.
Dietary calcium appears to play a pivotal role in the regulation of energy metabolism and obesity risk.102,–104 Zemel et al.105 observed that those patients in the highest quartile of adiposity were negatively associated with calcium and dairy product intake. A more recent nutritional intervention trial also demonstrated that higher low-fat dairy intake among overweight type-2 diabetic patients on isocaloric-restricted regimens enhances the weight-loss process.106 The proposed mechanisms are primarily mediated by circulating calcitriol.107 The increased calcitriol produced in response to low-calcium diets stimulates adipocyte Ca2+ influx and, consequently, promotes adiposity, while higher calcium diets inhibit lipogenesis, stimulate lipolysis, lipid oxidation and thermogenesis, and inhibit diet-induced obesity in mice.107 Moreover, a published meta-analysis concluded that dietary calcium has the potential to increase fecal fat excretion, which could be relevant for preventing weight (re)gain.108 However, some investigators did not find dietary calcium enrichment to have beneficial effects during a weight-loss process.109,110 Thus, the effect of calcium on weight loss continues to be unclear, indicating that more long-term studies are required in this field.
Other dietary micronutrients that have been used in the treatment of obesity and some metabolic disorders are potassium and magnesium. Both minerals are main components of the DASH diet, which has been designed as an anti-hypertensive treatment.111 This diet basically consists of high fruit, vegetable, and low-fat dairy content and is designed to be lower in total fat, saturated fat, and cholesterol and abundant in nutrients such as magnesium, calcium, and protein. Some data suggest that higher potassium and magnesium intake could favor decreases in blood pressure.111 Thus, high levels of potassium, magnesium, and calcium seem to have a beneficial effect on the weight loss process as well as on metabolic diseases. Indeed, some nutritional intervention studies have found the DASH diet to be more effective for weight loss and metabolic variables than other conventional diets,112,113 but the responsible mechanism remains to be elucidated.
Long-term weight maintenance is a key factor in the effective treatment of obesity. In this regard, interest in natural herbal supplements is growing rapidly. Human studies have shown that caffeine and green tea catechins may improve weight loss as well as weight maintenance through their effects on thermogenesis and fat oxidation.114,–116 One recent study investigated the role of a green tea-caffeine mixture on weight maintenance after a period of body weight loss and found weight stability to be improved in comparison with patients that did not eat the mixture.117
Overall, more and longer nutritional intervention strategies should be developed to investigate the efficacy of more micronutrient supplements or natural micronutrient-enriched foods for improving weight loss and facilitating weight maintenance.
Metabolic syndrome treatment
Metabolic syndrome (MetS) is defined by a group of risk factors such as visceral obesity, insulin resistance, elevated blood pressure, and dyslipidemia, associated with subsequent development of type 2 diabetes and cardiovascular disease.118 Despite the complexity of its origins, there is general agreement that the rising prevalence of MetS is largely due to the increasing incidence of obesity.119 Abdominal obesity or visceral fat are closely associated with insulin resistance and a low-grade pro-inflammatory state,120 which are considered primary factors in the development of MetS.121
There is general agreement that lifestyle changes focused on weight reduction should comprise the first-line approach to treating patients with MetS.122 Currently, however, there is no single diet recommended for these patients. General recommendations include a total fat intake of 25–35% of daily caloric consumption with an emphasis on unsaturated fat, saturated fat <7% of total calories, trans fatty acid intake <1% of total calories and <200 mg of dietary cholesterol, an intake of carbohydrates oscillating from 50% to 60% of total calories, and 15% of total calories as protein.123,124 However, some authors have emphasized that dietary treatment should be different depending on the presence of MetS components. For example, dietary treatment could be individualized on the basis of the specific metabolic disorders developed in each patient.26,36
Nutritional factors currently proposed for the treatment of obesity, cardiovascular diseases, and MetS are all those previously mentioned, i.e., high-protein low-CHO diets, low-GI carbohydrates, and regular fish consumption in order to assure an adequate omega-3 FA intake. Diets with low-GI and high-fiber content induce greater weight loss than conventional diets, decrease fasting glucose and insulin levels, reduce total and LDL cholesterol, and improve blood pressure.5,123 Several trials have already shown the benefits of dietary consumption of omega-3 FAs in MetS and obesity patients.90,–126 Fish intake and omega-3 FAs have been recommended as an integral component of the treatment of patients with MetS, since its consumption has been related with several metabolic benefits, including blood pressure reduction,97,–128 decreased plasma levels of triglycerides,129 improved HDL-cholesterol levels,130 and reduced inflammation.131 Moreover, several studies have confirmed that omega-3 FA consumption during energy reduction does not promote weight loss in comparison with a conventional diet; however, it does induce positive effects on insulin resistance, independently from changes in body weight,23,132 and it reduces triglyceride concentrations in a dose-dependent way89 as well as circulating inflammatory cytokines.90,133
Low-fat diets, on the other hand, may effectively reduce plasma LDL-cholesterol levels; however, if they promote higher carbohydrate consumption (intake of sugar and highly refined starches), this could have adverse effects on the metabolic risk profile.134,135 Indeed, there are reported negative effects of high-CHO diets on triglyceride levels as well as on insulin resistance.136 In the same way, very-low-CHO diets may induce rapid weight losses and some beneficial metabolic changes, such as insulin level reduction over short-term periods. However, these diets in which saturated and trans fats as well as high-fat meats and products are not controlled could produce adverse metabolic effects, especially in susceptible patients with MetS.11 A recent study assessed the effect of a very-low-CHO diet on endothelial function and reported that the cardiovascular risk was increased in the first days following a very-low-CHO diet.137
Diets prescribing an increased dietary protein intake have also been evaluated for use in the treatment of MetS.138 In conjunction with facilitating weight loss, diets in which carbohydrates are replaced by protein usually result in improvements in body composition and in plasma lipids and lipoproteins. High-protein low-CHO diets have been found to have positive effects on reducing risk factors for heart disease, including reducing serum triacylglycerol, increasing HDL cholesterol, increasing LDL particle size, and reducing blood pressure. These diets appear to be particularly useful for individuals exhibiting the atherogenic dyslipidemia of MetS.139 Several studies have shown that increasing dietary protein content (30%) and decreasing carbohydrates (40%) may favor the reduction of fat mass, particularly abdominal fat mass.140 Decreasing the dietary intake of carbohydrates and replacing it with either fat or protein has been shown to reduce triacylglycerides and increase HDL cholesterol even under weight-stable conditions.141,142 However, substitution with protein may be even more beneficial than fat for eliciting lipid changes141,–143 and improving insulin action.16,143 McAuley et al.142 provide evidence that the primary means of lowering triacylglycerides is reducing carbohydrate intake; they further suggest that increasing dietary fat as an approach to lowering dietary carbohydrates may not be as efficacious as increasing protein. Following extensive research, consensus was achieved that reducing dietary carbohydrates is the critical approach to treating or managing the manifestations of MetS.144
Diets with a high-MUFA content can positively affect insulin levels, as shown by several authors,57,–95 and have consequently been suggested for the treatment of obesity and MetS. Diets containing foods with antioxidant properties have also been studied to investigate the potential association between obesity and oxidative stress.145 Several studies have found different effects on weight loss and cardiometabolic changes when antioxidant-enriched foods such as legumes, fruits, and vegetables are consumed during a weight-reduction period.146,–148 Inclusion of legumes in a weight-lowering program has been shown to increase the intake of isoflavones, saponins, a variety of micronutrients, and phytochemicals.149 Flavonoids occurring normally in the diet exhibit a variety of beneficial effects on health. In addition to reducing total and LDL cholesterol, as well as triglyceride levels, intake of these plant products by obese subjects has been related with higher plasma antioxidant status and greater reductions in oxidative stress.145
The anti-inflammatory properties of flavonoids have also been studied.20 Recent investigations have shown that some flavonoids are modulators of proinflammatory gene expression, leading to attenuation of the inflammatory response.150,151 Thus, the utility of flavonoids as therapeutic agents in the treatment of inflammatory diseases is also under consideration.20 Likewise, antioxidant minerals are dietary factors of interest for the nutritional management of MetS.19 Particular attention is paid to the development of selenium-enriched food and nutritional products,152 since this element acts as a cofactor of enzymes, which protect against oxidative stress and are also involved in the regulation of the inflammatory response.153 Moreover, selenium-enriched diets have been associated with lower body mass index. In this context, vitamin C is also considered of nutritional value. In addition to its antioxidant properties, which may induce improvements in oxidative stress,154 an observational study examined the relationship between plasma vitamin C and adiposity, showing that plasma vitamin C was inversely related to BMI, percentage of body fat, and waist circumference in both women and men. However, in a nutritional intervention trial, supplementation of the diet with vitamin C did not improve weight loss in comparison with the placebo group.155 Thus, the effects of ascorbic acid during weight loss remain inconclusive.
Overall, the ideal diet should combine all the dietary factors thought to influence cardiometabolic risk. A diet reflecting a lower content of refined carbohydrates, a high content of fiber and low-GI carbohydrates, antioxidants and phytochemicals from vegetables, fruits, and legumes, a moderate fat content (mostly unsaturated and omega-3 FAs), and a moderate-to-high content of vegetable and fish proteins,156 should be appropriate for the treatment of MetS patients. These dietary patterns may exert positive effects on almost all components of MetS as well as other conditions associated with it, including inflammation, insulin resistance, and endothelial dysfunction.119,–158 The PREDIMED study evaluated the effect of the traditional Mediterranean diet on the features of MetS and on cardiovascular disease (CVD) prevention. The Mediterranean diet employed in the study was based on high intake of cereals, vegetables, fruits, and olive oil, moderate intake of fish and alcohol, mostly wine, and low intake of dairy products, meats, and sweets. The results of this trial showed that among older participants at high risk for developing CVD, those who consumed a non-energy-restricted, traditional, Mediterranean-style diet supplemented with one daily serving of mixed nuts for 1 year, showed an overall reduction in the prevalence of MetS compared with participants given advice on following a low-fat diet.159 These positive findings are supported by those of a prospective cohort study conducted in 13,380 Spanish university graduates who were followed up for a median of 4.4 years; the results indicated that adherence to a Mediterranean diet is associated with reduced risk of diabetes.160
CONCLUSION
As shown in this review, there are many dietary strategies focused on macronutrient distribution or micronutrient and food-enriched manipulation for the treatment of obesity and features of MetS features (Table 1). However, the real challenge is to find the appropriate approach for maintaining body weight loss and preventing subsequent relapse, while simultaneously reducing cardiovascular risk factors.
Changes on weight loss as well as biochemical and metabolic variables in some selected nutritional intervention studies differing in macronutrient distribution or dietary factors composition.
| Reference . | Study data . | Diets . | Weight loss and blood pressure . | Glucose and lipid metabolism . | Inflammation and/or oxidative stress markers . |
|---|---|---|---|---|---|
| Abete et al. (2008)21 | n = 32 healthy obese men and women 8 weeks | Higher-GI (15%P; 30%L; 55%CHO) Lower-GI (15%P; 30%L; 55%CHO) | −5.3%, ↓SBP, ↓DBP −7.7%, ↓SBP, ↓DBP | ↑insulin, ↑HOMA, ↓TC, ↓LDL-c, ↑TG, ↓HDL-c ↓Insulin, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | Not reported |
| Abete et al. (2009)162 | n = 19 healthy obese men 8 weeks | Control (15%P; 30%L; 55%CHO) Moderate-P (30%P; 30%L; 40%CHO) | −5.5%, ↓SBP, ↓DBP −8.3%, ↓SBP, ↓DBP | =insulin, =HOMA, ↓TC, ↓LDL-c, =TG, ↓HDL-c ↓insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | Not reported |
| Abete et al. (2008)23 | n = 32 healthy obese men and women 8 weeks | Control (15%P; 30%L; 55%CHO) Fatty fish (15%P; 30%L; 55%CHO) + 3 fatty fish meals/week. | −5.3%, ↓SBP, ↓DBP −5.5%, ↓SBP, ↓DBP | ↑Insulin, =HOMA, =TC, =LDL-c, =TG, =HDL-c ↓Insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↑HDL-c | =leptin ↓leptin |
| Agus et al. (2000)26 | n = 10 moderately overweight young men Two 9-day periods separated by a washout period of 2–6 weeks | Low-GI (43%CHO; 27%P; 30%L) High-GI (67%CHO; 15%P; 18%L) | −3.6 kg −3.2 kg Low-GI diet increased hunger levels | ↓Insulin, REE decreased to a lesser extent | ↓leptin |
| Backes et al. (2008)16 | n = 23 overweight insulin-resistant women 3 months | Low-CHO (40% CHO) | −5.0 kg; ↓DBP | ↓Glucose, ↓TG | Not reported |
| Brehm et al. (2005)163 | n = 50 healthy obese women 4 months | Low-CHO (ad libitum) Low-fat (15%P; 30%L; 55%CHO) | −9.7±0.7 kg −6.1±0.9 kg | No differences between diets on glucose, insulin, and HOMA. Both diets: ↓TC, ↓LDL-c, and ↓ TG ↑ HDL-c with low-CHO | Not reported |
| Brinkworth et al. (2009)14 | n = 118 men and women with abdominal obesity and at least one additional metabolic syndrome risk factor 1 year | LC (4%CHO; 35%P; 61%L) LF (46%CHO; 24%P; 30%L) | −14.5 kg; ↓SBP; ↓DBP −11.5 k; ↓SBP; ↓DBP | Both diets: ↓glucose, ↓insulin, ↓HOMA-IR LC: ↓TG, ↑LDL-c, ↑HDL-c, ↑Apo-B | ↓CRP independently of diet composition |
| Buscemi et al. (2009)137 | n = 20 healthy obese women 2 months | Atkin (30%P; 65%L; 5%CHO) Mediterranean (20%P; 25%L; 55%CHO) | −7.6 kg;↓SBP; ↓DBP −4.9 kg | Both diets: ↓insulin, ↓HOMA, ↓TC, ↓LDL-c, =HDL-c, =TG | Both diets =IL-6, =TNF-α, =adiponectin, =8-iso-PGF2α, |
| Claessens et al. (2009)37 | n = 48 healthy obese 6-week weight loss + 12-week maintenance | High-CHO (ad libitum) >55%CHO; 30%L High-P (ad libitum) >25%P; 30%L | +1.1 kg −1.1 kg; ↓SBP; ↓DBP | Both diets: =insulin, =HOMA, ↑TC, ↑LDL-c and ↑HDL-c Only high-P: ↓TG | Both diets ↓adiponectin, ↑leptin |
| Clifton et al. (2008)10 | n = 79 healthy overweight / obese women 64 weeks (12-week weight loss + 52-week follow-up) | HP (34%P; 20%L; 46%CHO) HC (64%CHO; 20%L; 17%P) <10% of saturated fat in both diets | −4.6 kg −4.4 kg | ↓Glucose, ↓insulin, ↓LDL-c, ↓TG, ↑HDL-c with no differences between diets | Both diets: ↓CRP, ↓homocysteine |
| Crujeiras et al. (2007)145 | n = 32 healthy obese men/women 8 weeks | Control (15%P; 30%L; 55%CHO) Legume (15%P; 30%L; 55%CHO) (4 legume meals/week) | −5.3%; ↓SBP; ↓DBP −7.7%; ↓SBP; ↓DBP | ↑Insulin, ↑HOMA, ↓TC, ↓LDL-c, ↑TG, ↓HDL-c ↓Insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | =ox-LDL,=MDA, =8-iso-PGF2α ↓ox-LDL, ↓MDA, ↓8-iso-PGF2α, ↑AOP |
| Dumesnil et al. (2001)134 | n = 12 healthy obese men two 6-day experimental periods with a 2-week washout period | Low-fat-high-CHO-high-P (30%P; 30%L; 40%CHO) ad libitum AHA (15%P; 30%L; 55%CHO) ad libitum | −2.3 kg; Higher satiety level No change in body weight Low-satiety level | ↓Insulin, =TC, =LDL-c, ↓TG, =HDL-c =insulin, =TC, =LDL-c, ↑TG, ↓HDL-c | Not reported |
| Das et al. (2007)46 | n = 34 healthy overweight adults 12 months | High-glycemic load (20%P; 20%L; 60%CHO) Low-glycemic load (30%P; 30%L; 40%CHO) | −8.0% −7.8% | Both diets: ↓insulin, ↓TC, ↓LDL-c, ↓TG, and ↑HDL-c | Not reported |
| Foster et al. (2003)42 | n = 63 obese men and women 1 year Poor adherence in both diets | Low-CHO high-P high-L (ad libitum) High-CHO (60%CHO; 25%L; 15%P) | −7.3%; =SBP;↓DBP −4.5%; =SBP;↓DBP | ↑HDL-c; ↓TG Both diets: ↓insulin; =TC; =LDL-c | Not reported |
| Gardner et al. (2007)164 | n = 77 n = 79 n = 79 n = 76 Obese women 1 year of intervention | Atkins Zone (30%P; 30%L; 40%CHO) Learn (15%P; 30%L; 55%CHO) Ornish (very-high-CHO) All diets were ad libitum | −4.7 kg, ↓SBP; ↓DBP −1.6 kg, ↓SBP; ↓DBP −2.2 kg, ↓SBP; ↓DBP −2.6 kg, ↓SBP; =DBP | ↓Insulin, =LDL-c, ↑HDL-c, ↓TG ↓Insulin, =LDL-c, ↑HDL-c, =TG ↓Insulin, =LDL-c, ↑HDL-c, ↓TG =Insulin, ↓LDL-c, ↓HDL-c, ↓TG | Not reported |
| Layman et al. (2009)64 | n = 130 4-months No differences in compliance between groups | PRO-group (30%P; 30%L; 40%CHO) CHO-group (15%P; 30%L; 55%CHO) | BW no differences between groups PRO-group greater ↓fat mass | PRO-d: ↓TG, ↑HDL-c CHO-d: ↓TC, ↓LDL-c, ↓HDL-c | Not reported |
| Noakes et al. (2005)135 | n = 100 healthy obese women 12 weeks | High-P (34%P; 20%L; 46%CHO) High-CHO (17%P; 20%L; 64% CHO) Both diets ≤10% saturated fat | −7.6 kg −6.9 kg | High-P: ↓TG, ↑B-12 Both diets: ↓glucose, ↓insulin, ↓LDL-c, ↓HDL-c, ↑folate, ↑B-6 | Both diets =homocysteine, ↓CRP |
| O'Brien et al. (2005)165 | n = 42 3 months Obese women | Low-fat (15%P; 30%L; 55%CHO) Very-low-CHO (ad libitum) | −4.3 ± 3.5 kg; ↓SBP; ↓DBP −7.6 ± 3.2 kg; ↓SBP; ↓DBP | Both diets: ↓insulin, ↓HOMA-IR; ↓TC and ↓LDL-c. Only very-low-CHO ↓TG | Both diets ↓CRP; ↓SAA |
| Petersen et al. (2006)56 | n = 771 healthy obese adults 10 weeks | Low-fat (20–25%L; 60–65%CHO; 15%P) High-fat (40–45%%L; 60–65%CHO; 15%P) | −6.9 kg −6.6 kg | ↓TC, ↓LDL-c, ↓TG, ↓HDL-c (changes were higher in this group) ↓TC, ↓LDL-c, ↓TG, ↓HDL-c Both groups:↓insulin | Not reported |
| Raben et al. (2002)44 | n = 398 obese subjects 6 months | Low-fat high-CHO high-GI (ad libitum) Low-fat high-CHO low-GI (ad libitum) | −1.7 kg −2.6 kg | Both diets improved glucose, insulin, and fasting blood lipids | Not reported |
| Seshadri et al. (2004)166 | n = 78 severe obese subjects 31 were diabetics, 36 had metabolic syndrome 49 with hypertension medication and 35 with lipid-lowering medication 6 months | Low-CHO (<30 g CHO; ad libitum) Conventional (≤30%L) energy restricted | −8.5 kg −3.5 kg | ↓insulin; ↓TG Both diets: =TC; =LDL-c; =HDL-c | ↓CRP |
| Skov et al. (1999)31 | n = 65 healthy overweight and obese men and women 6 months | High-P (25%P; 30%L; 45%CHO) High-CHO (12%P; 30%L; 58%CHO) | −8.9 kg −5.1 kg | ↓TG; ↓FFA | Not reported |
| Sloth et al. (2009)84 | n = 131 healthy overweight and obese subjects 6 months weight maintenance period | After losing 8% of the initial body weight, participants were instructed to follow ad libitum diets: MUFA, moderate fat (35–45%L; >20%MUFA) Low-F (20–30%L) Control (35%L; >15% SFA) | There was a significant weight increase with no differences between groups | Low-L and MUFA groups had lower insulin concentrations HbA1c increased in the control group There were no group differences in GLP-1; GLP-2 | Not reported |
| Stern et al. (2004)43 | n = 132 severe obese subjects 83% had diabetes or metabolic syndrome 1 year | Low-CHO (<30 g CHO; ad libitum) Conventional (≤30%L) energy restricted | −5.1 kg −3.1 kg Both: =SBP; =DBP | ↓TG; =HDL-c; ↓HBA1C ↓HDL-c; =HBA1C Both: =glucose; =insulin, =TC; =LDL-c | Not reported |
| Reference . | Study data . | Diets . | Weight loss and blood pressure . | Glucose and lipid metabolism . | Inflammation and/or oxidative stress markers . |
|---|---|---|---|---|---|
| Abete et al. (2008)21 | n = 32 healthy obese men and women 8 weeks | Higher-GI (15%P; 30%L; 55%CHO) Lower-GI (15%P; 30%L; 55%CHO) | −5.3%, ↓SBP, ↓DBP −7.7%, ↓SBP, ↓DBP | ↑insulin, ↑HOMA, ↓TC, ↓LDL-c, ↑TG, ↓HDL-c ↓Insulin, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | Not reported |
| Abete et al. (2009)162 | n = 19 healthy obese men 8 weeks | Control (15%P; 30%L; 55%CHO) Moderate-P (30%P; 30%L; 40%CHO) | −5.5%, ↓SBP, ↓DBP −8.3%, ↓SBP, ↓DBP | =insulin, =HOMA, ↓TC, ↓LDL-c, =TG, ↓HDL-c ↓insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | Not reported |
| Abete et al. (2008)23 | n = 32 healthy obese men and women 8 weeks | Control (15%P; 30%L; 55%CHO) Fatty fish (15%P; 30%L; 55%CHO) + 3 fatty fish meals/week. | −5.3%, ↓SBP, ↓DBP −5.5%, ↓SBP, ↓DBP | ↑Insulin, =HOMA, =TC, =LDL-c, =TG, =HDL-c ↓Insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↑HDL-c | =leptin ↓leptin |
| Agus et al. (2000)26 | n = 10 moderately overweight young men Two 9-day periods separated by a washout period of 2–6 weeks | Low-GI (43%CHO; 27%P; 30%L) High-GI (67%CHO; 15%P; 18%L) | −3.6 kg −3.2 kg Low-GI diet increased hunger levels | ↓Insulin, REE decreased to a lesser extent | ↓leptin |
| Backes et al. (2008)16 | n = 23 overweight insulin-resistant women 3 months | Low-CHO (40% CHO) | −5.0 kg; ↓DBP | ↓Glucose, ↓TG | Not reported |
| Brehm et al. (2005)163 | n = 50 healthy obese women 4 months | Low-CHO (ad libitum) Low-fat (15%P; 30%L; 55%CHO) | −9.7±0.7 kg −6.1±0.9 kg | No differences between diets on glucose, insulin, and HOMA. Both diets: ↓TC, ↓LDL-c, and ↓ TG ↑ HDL-c with low-CHO | Not reported |
| Brinkworth et al. (2009)14 | n = 118 men and women with abdominal obesity and at least one additional metabolic syndrome risk factor 1 year | LC (4%CHO; 35%P; 61%L) LF (46%CHO; 24%P; 30%L) | −14.5 kg; ↓SBP; ↓DBP −11.5 k; ↓SBP; ↓DBP | Both diets: ↓glucose, ↓insulin, ↓HOMA-IR LC: ↓TG, ↑LDL-c, ↑HDL-c, ↑Apo-B | ↓CRP independently of diet composition |
| Buscemi et al. (2009)137 | n = 20 healthy obese women 2 months | Atkin (30%P; 65%L; 5%CHO) Mediterranean (20%P; 25%L; 55%CHO) | −7.6 kg;↓SBP; ↓DBP −4.9 kg | Both diets: ↓insulin, ↓HOMA, ↓TC, ↓LDL-c, =HDL-c, =TG | Both diets =IL-6, =TNF-α, =adiponectin, =8-iso-PGF2α, |
| Claessens et al. (2009)37 | n = 48 healthy obese 6-week weight loss + 12-week maintenance | High-CHO (ad libitum) >55%CHO; 30%L High-P (ad libitum) >25%P; 30%L | +1.1 kg −1.1 kg; ↓SBP; ↓DBP | Both diets: =insulin, =HOMA, ↑TC, ↑LDL-c and ↑HDL-c Only high-P: ↓TG | Both diets ↓adiponectin, ↑leptin |
| Clifton et al. (2008)10 | n = 79 healthy overweight / obese women 64 weeks (12-week weight loss + 52-week follow-up) | HP (34%P; 20%L; 46%CHO) HC (64%CHO; 20%L; 17%P) <10% of saturated fat in both diets | −4.6 kg −4.4 kg | ↓Glucose, ↓insulin, ↓LDL-c, ↓TG, ↑HDL-c with no differences between diets | Both diets: ↓CRP, ↓homocysteine |
| Crujeiras et al. (2007)145 | n = 32 healthy obese men/women 8 weeks | Control (15%P; 30%L; 55%CHO) Legume (15%P; 30%L; 55%CHO) (4 legume meals/week) | −5.3%; ↓SBP; ↓DBP −7.7%; ↓SBP; ↓DBP | ↑Insulin, ↑HOMA, ↓TC, ↓LDL-c, ↑TG, ↓HDL-c ↓Insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | =ox-LDL,=MDA, =8-iso-PGF2α ↓ox-LDL, ↓MDA, ↓8-iso-PGF2α, ↑AOP |
| Dumesnil et al. (2001)134 | n = 12 healthy obese men two 6-day experimental periods with a 2-week washout period | Low-fat-high-CHO-high-P (30%P; 30%L; 40%CHO) ad libitum AHA (15%P; 30%L; 55%CHO) ad libitum | −2.3 kg; Higher satiety level No change in body weight Low-satiety level | ↓Insulin, =TC, =LDL-c, ↓TG, =HDL-c =insulin, =TC, =LDL-c, ↑TG, ↓HDL-c | Not reported |
| Das et al. (2007)46 | n = 34 healthy overweight adults 12 months | High-glycemic load (20%P; 20%L; 60%CHO) Low-glycemic load (30%P; 30%L; 40%CHO) | −8.0% −7.8% | Both diets: ↓insulin, ↓TC, ↓LDL-c, ↓TG, and ↑HDL-c | Not reported |
| Foster et al. (2003)42 | n = 63 obese men and women 1 year Poor adherence in both diets | Low-CHO high-P high-L (ad libitum) High-CHO (60%CHO; 25%L; 15%P) | −7.3%; =SBP;↓DBP −4.5%; =SBP;↓DBP | ↑HDL-c; ↓TG Both diets: ↓insulin; =TC; =LDL-c | Not reported |
| Gardner et al. (2007)164 | n = 77 n = 79 n = 79 n = 76 Obese women 1 year of intervention | Atkins Zone (30%P; 30%L; 40%CHO) Learn (15%P; 30%L; 55%CHO) Ornish (very-high-CHO) All diets were ad libitum | −4.7 kg, ↓SBP; ↓DBP −1.6 kg, ↓SBP; ↓DBP −2.2 kg, ↓SBP; ↓DBP −2.6 kg, ↓SBP; =DBP | ↓Insulin, =LDL-c, ↑HDL-c, ↓TG ↓Insulin, =LDL-c, ↑HDL-c, =TG ↓Insulin, =LDL-c, ↑HDL-c, ↓TG =Insulin, ↓LDL-c, ↓HDL-c, ↓TG | Not reported |
| Layman et al. (2009)64 | n = 130 4-months No differences in compliance between groups | PRO-group (30%P; 30%L; 40%CHO) CHO-group (15%P; 30%L; 55%CHO) | BW no differences between groups PRO-group greater ↓fat mass | PRO-d: ↓TG, ↑HDL-c CHO-d: ↓TC, ↓LDL-c, ↓HDL-c | Not reported |
| Noakes et al. (2005)135 | n = 100 healthy obese women 12 weeks | High-P (34%P; 20%L; 46%CHO) High-CHO (17%P; 20%L; 64% CHO) Both diets ≤10% saturated fat | −7.6 kg −6.9 kg | High-P: ↓TG, ↑B-12 Both diets: ↓glucose, ↓insulin, ↓LDL-c, ↓HDL-c, ↑folate, ↑B-6 | Both diets =homocysteine, ↓CRP |
| O'Brien et al. (2005)165 | n = 42 3 months Obese women | Low-fat (15%P; 30%L; 55%CHO) Very-low-CHO (ad libitum) | −4.3 ± 3.5 kg; ↓SBP; ↓DBP −7.6 ± 3.2 kg; ↓SBP; ↓DBP | Both diets: ↓insulin, ↓HOMA-IR; ↓TC and ↓LDL-c. Only very-low-CHO ↓TG | Both diets ↓CRP; ↓SAA |
| Petersen et al. (2006)56 | n = 771 healthy obese adults 10 weeks | Low-fat (20–25%L; 60–65%CHO; 15%P) High-fat (40–45%%L; 60–65%CHO; 15%P) | −6.9 kg −6.6 kg | ↓TC, ↓LDL-c, ↓TG, ↓HDL-c (changes were higher in this group) ↓TC, ↓LDL-c, ↓TG, ↓HDL-c Both groups:↓insulin | Not reported |
| Raben et al. (2002)44 | n = 398 obese subjects 6 months | Low-fat high-CHO high-GI (ad libitum) Low-fat high-CHO low-GI (ad libitum) | −1.7 kg −2.6 kg | Both diets improved glucose, insulin, and fasting blood lipids | Not reported |
| Seshadri et al. (2004)166 | n = 78 severe obese subjects 31 were diabetics, 36 had metabolic syndrome 49 with hypertension medication and 35 with lipid-lowering medication 6 months | Low-CHO (<30 g CHO; ad libitum) Conventional (≤30%L) energy restricted | −8.5 kg −3.5 kg | ↓insulin; ↓TG Both diets: =TC; =LDL-c; =HDL-c | ↓CRP |
| Skov et al. (1999)31 | n = 65 healthy overweight and obese men and women 6 months | High-P (25%P; 30%L; 45%CHO) High-CHO (12%P; 30%L; 58%CHO) | −8.9 kg −5.1 kg | ↓TG; ↓FFA | Not reported |
| Sloth et al. (2009)84 | n = 131 healthy overweight and obese subjects 6 months weight maintenance period | After losing 8% of the initial body weight, participants were instructed to follow ad libitum diets: MUFA, moderate fat (35–45%L; >20%MUFA) Low-F (20–30%L) Control (35%L; >15% SFA) | There was a significant weight increase with no differences between groups | Low-L and MUFA groups had lower insulin concentrations HbA1c increased in the control group There were no group differences in GLP-1; GLP-2 | Not reported |
| Stern et al. (2004)43 | n = 132 severe obese subjects 83% had diabetes or metabolic syndrome 1 year | Low-CHO (<30 g CHO; ad libitum) Conventional (≤30%L) energy restricted | −5.1 kg −3.1 kg Both: =SBP; =DBP | ↓TG; =HDL-c; ↓HBA1C ↓HDL-c; =HBA1C Both: =glucose; =insulin, =TC; =LDL-c | Not reported |
Abbreviations: 8-iso-PGF2α, 8-isoprostane F2α; AHA, American Heart Association; AOP, total plasma antioxidant power; BW, body weight; CHO, carbohydrates; CRP, C-reactive protein; FFA, free fatty acids; FM, fat mass; HBA1C, hemoglobin A1C; L, lipids; MDA, malondialdehyde; MUFA, monounsaturated fatty acids; ox-LDL, oxidized-LDL.
Changes on weight loss as well as biochemical and metabolic variables in some selected nutritional intervention studies differing in macronutrient distribution or dietary factors composition.
| Reference . | Study data . | Diets . | Weight loss and blood pressure . | Glucose and lipid metabolism . | Inflammation and/or oxidative stress markers . |
|---|---|---|---|---|---|
| Abete et al. (2008)21 | n = 32 healthy obese men and women 8 weeks | Higher-GI (15%P; 30%L; 55%CHO) Lower-GI (15%P; 30%L; 55%CHO) | −5.3%, ↓SBP, ↓DBP −7.7%, ↓SBP, ↓DBP | ↑insulin, ↑HOMA, ↓TC, ↓LDL-c, ↑TG, ↓HDL-c ↓Insulin, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | Not reported |
| Abete et al. (2009)162 | n = 19 healthy obese men 8 weeks | Control (15%P; 30%L; 55%CHO) Moderate-P (30%P; 30%L; 40%CHO) | −5.5%, ↓SBP, ↓DBP −8.3%, ↓SBP, ↓DBP | =insulin, =HOMA, ↓TC, ↓LDL-c, =TG, ↓HDL-c ↓insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | Not reported |
| Abete et al. (2008)23 | n = 32 healthy obese men and women 8 weeks | Control (15%P; 30%L; 55%CHO) Fatty fish (15%P; 30%L; 55%CHO) + 3 fatty fish meals/week. | −5.3%, ↓SBP, ↓DBP −5.5%, ↓SBP, ↓DBP | ↑Insulin, =HOMA, =TC, =LDL-c, =TG, =HDL-c ↓Insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↑HDL-c | =leptin ↓leptin |
| Agus et al. (2000)26 | n = 10 moderately overweight young men Two 9-day periods separated by a washout period of 2–6 weeks | Low-GI (43%CHO; 27%P; 30%L) High-GI (67%CHO; 15%P; 18%L) | −3.6 kg −3.2 kg Low-GI diet increased hunger levels | ↓Insulin, REE decreased to a lesser extent | ↓leptin |
| Backes et al. (2008)16 | n = 23 overweight insulin-resistant women 3 months | Low-CHO (40% CHO) | −5.0 kg; ↓DBP | ↓Glucose, ↓TG | Not reported |
| Brehm et al. (2005)163 | n = 50 healthy obese women 4 months | Low-CHO (ad libitum) Low-fat (15%P; 30%L; 55%CHO) | −9.7±0.7 kg −6.1±0.9 kg | No differences between diets on glucose, insulin, and HOMA. Both diets: ↓TC, ↓LDL-c, and ↓ TG ↑ HDL-c with low-CHO | Not reported |
| Brinkworth et al. (2009)14 | n = 118 men and women with abdominal obesity and at least one additional metabolic syndrome risk factor 1 year | LC (4%CHO; 35%P; 61%L) LF (46%CHO; 24%P; 30%L) | −14.5 kg; ↓SBP; ↓DBP −11.5 k; ↓SBP; ↓DBP | Both diets: ↓glucose, ↓insulin, ↓HOMA-IR LC: ↓TG, ↑LDL-c, ↑HDL-c, ↑Apo-B | ↓CRP independently of diet composition |
| Buscemi et al. (2009)137 | n = 20 healthy obese women 2 months | Atkin (30%P; 65%L; 5%CHO) Mediterranean (20%P; 25%L; 55%CHO) | −7.6 kg;↓SBP; ↓DBP −4.9 kg | Both diets: ↓insulin, ↓HOMA, ↓TC, ↓LDL-c, =HDL-c, =TG | Both diets =IL-6, =TNF-α, =adiponectin, =8-iso-PGF2α, |
| Claessens et al. (2009)37 | n = 48 healthy obese 6-week weight loss + 12-week maintenance | High-CHO (ad libitum) >55%CHO; 30%L High-P (ad libitum) >25%P; 30%L | +1.1 kg −1.1 kg; ↓SBP; ↓DBP | Both diets: =insulin, =HOMA, ↑TC, ↑LDL-c and ↑HDL-c Only high-P: ↓TG | Both diets ↓adiponectin, ↑leptin |
| Clifton et al. (2008)10 | n = 79 healthy overweight / obese women 64 weeks (12-week weight loss + 52-week follow-up) | HP (34%P; 20%L; 46%CHO) HC (64%CHO; 20%L; 17%P) <10% of saturated fat in both diets | −4.6 kg −4.4 kg | ↓Glucose, ↓insulin, ↓LDL-c, ↓TG, ↑HDL-c with no differences between diets | Both diets: ↓CRP, ↓homocysteine |
| Crujeiras et al. (2007)145 | n = 32 healthy obese men/women 8 weeks | Control (15%P; 30%L; 55%CHO) Legume (15%P; 30%L; 55%CHO) (4 legume meals/week) | −5.3%; ↓SBP; ↓DBP −7.7%; ↓SBP; ↓DBP | ↑Insulin, ↑HOMA, ↓TC, ↓LDL-c, ↑TG, ↓HDL-c ↓Insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | =ox-LDL,=MDA, =8-iso-PGF2α ↓ox-LDL, ↓MDA, ↓8-iso-PGF2α, ↑AOP |
| Dumesnil et al. (2001)134 | n = 12 healthy obese men two 6-day experimental periods with a 2-week washout period | Low-fat-high-CHO-high-P (30%P; 30%L; 40%CHO) ad libitum AHA (15%P; 30%L; 55%CHO) ad libitum | −2.3 kg; Higher satiety level No change in body weight Low-satiety level | ↓Insulin, =TC, =LDL-c, ↓TG, =HDL-c =insulin, =TC, =LDL-c, ↑TG, ↓HDL-c | Not reported |
| Das et al. (2007)46 | n = 34 healthy overweight adults 12 months | High-glycemic load (20%P; 20%L; 60%CHO) Low-glycemic load (30%P; 30%L; 40%CHO) | −8.0% −7.8% | Both diets: ↓insulin, ↓TC, ↓LDL-c, ↓TG, and ↑HDL-c | Not reported |
| Foster et al. (2003)42 | n = 63 obese men and women 1 year Poor adherence in both diets | Low-CHO high-P high-L (ad libitum) High-CHO (60%CHO; 25%L; 15%P) | −7.3%; =SBP;↓DBP −4.5%; =SBP;↓DBP | ↑HDL-c; ↓TG Both diets: ↓insulin; =TC; =LDL-c | Not reported |
| Gardner et al. (2007)164 | n = 77 n = 79 n = 79 n = 76 Obese women 1 year of intervention | Atkins Zone (30%P; 30%L; 40%CHO) Learn (15%P; 30%L; 55%CHO) Ornish (very-high-CHO) All diets were ad libitum | −4.7 kg, ↓SBP; ↓DBP −1.6 kg, ↓SBP; ↓DBP −2.2 kg, ↓SBP; ↓DBP −2.6 kg, ↓SBP; =DBP | ↓Insulin, =LDL-c, ↑HDL-c, ↓TG ↓Insulin, =LDL-c, ↑HDL-c, =TG ↓Insulin, =LDL-c, ↑HDL-c, ↓TG =Insulin, ↓LDL-c, ↓HDL-c, ↓TG | Not reported |
| Layman et al. (2009)64 | n = 130 4-months No differences in compliance between groups | PRO-group (30%P; 30%L; 40%CHO) CHO-group (15%P; 30%L; 55%CHO) | BW no differences between groups PRO-group greater ↓fat mass | PRO-d: ↓TG, ↑HDL-c CHO-d: ↓TC, ↓LDL-c, ↓HDL-c | Not reported |
| Noakes et al. (2005)135 | n = 100 healthy obese women 12 weeks | High-P (34%P; 20%L; 46%CHO) High-CHO (17%P; 20%L; 64% CHO) Both diets ≤10% saturated fat | −7.6 kg −6.9 kg | High-P: ↓TG, ↑B-12 Both diets: ↓glucose, ↓insulin, ↓LDL-c, ↓HDL-c, ↑folate, ↑B-6 | Both diets =homocysteine, ↓CRP |
| O'Brien et al. (2005)165 | n = 42 3 months Obese women | Low-fat (15%P; 30%L; 55%CHO) Very-low-CHO (ad libitum) | −4.3 ± 3.5 kg; ↓SBP; ↓DBP −7.6 ± 3.2 kg; ↓SBP; ↓DBP | Both diets: ↓insulin, ↓HOMA-IR; ↓TC and ↓LDL-c. Only very-low-CHO ↓TG | Both diets ↓CRP; ↓SAA |
| Petersen et al. (2006)56 | n = 771 healthy obese adults 10 weeks | Low-fat (20–25%L; 60–65%CHO; 15%P) High-fat (40–45%%L; 60–65%CHO; 15%P) | −6.9 kg −6.6 kg | ↓TC, ↓LDL-c, ↓TG, ↓HDL-c (changes were higher in this group) ↓TC, ↓LDL-c, ↓TG, ↓HDL-c Both groups:↓insulin | Not reported |
| Raben et al. (2002)44 | n = 398 obese subjects 6 months | Low-fat high-CHO high-GI (ad libitum) Low-fat high-CHO low-GI (ad libitum) | −1.7 kg −2.6 kg | Both diets improved glucose, insulin, and fasting blood lipids | Not reported |
| Seshadri et al. (2004)166 | n = 78 severe obese subjects 31 were diabetics, 36 had metabolic syndrome 49 with hypertension medication and 35 with lipid-lowering medication 6 months | Low-CHO (<30 g CHO; ad libitum) Conventional (≤30%L) energy restricted | −8.5 kg −3.5 kg | ↓insulin; ↓TG Both diets: =TC; =LDL-c; =HDL-c | ↓CRP |
| Skov et al. (1999)31 | n = 65 healthy overweight and obese men and women 6 months | High-P (25%P; 30%L; 45%CHO) High-CHO (12%P; 30%L; 58%CHO) | −8.9 kg −5.1 kg | ↓TG; ↓FFA | Not reported |
| Sloth et al. (2009)84 | n = 131 healthy overweight and obese subjects 6 months weight maintenance period | After losing 8% of the initial body weight, participants were instructed to follow ad libitum diets: MUFA, moderate fat (35–45%L; >20%MUFA) Low-F (20–30%L) Control (35%L; >15% SFA) | There was a significant weight increase with no differences between groups | Low-L and MUFA groups had lower insulin concentrations HbA1c increased in the control group There were no group differences in GLP-1; GLP-2 | Not reported |
| Stern et al. (2004)43 | n = 132 severe obese subjects 83% had diabetes or metabolic syndrome 1 year | Low-CHO (<30 g CHO; ad libitum) Conventional (≤30%L) energy restricted | −5.1 kg −3.1 kg Both: =SBP; =DBP | ↓TG; =HDL-c; ↓HBA1C ↓HDL-c; =HBA1C Both: =glucose; =insulin, =TC; =LDL-c | Not reported |
| Reference . | Study data . | Diets . | Weight loss and blood pressure . | Glucose and lipid metabolism . | Inflammation and/or oxidative stress markers . |
|---|---|---|---|---|---|
| Abete et al. (2008)21 | n = 32 healthy obese men and women 8 weeks | Higher-GI (15%P; 30%L; 55%CHO) Lower-GI (15%P; 30%L; 55%CHO) | −5.3%, ↓SBP, ↓DBP −7.7%, ↓SBP, ↓DBP | ↑insulin, ↑HOMA, ↓TC, ↓LDL-c, ↑TG, ↓HDL-c ↓Insulin, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | Not reported |
| Abete et al. (2009)162 | n = 19 healthy obese men 8 weeks | Control (15%P; 30%L; 55%CHO) Moderate-P (30%P; 30%L; 40%CHO) | −5.5%, ↓SBP, ↓DBP −8.3%, ↓SBP, ↓DBP | =insulin, =HOMA, ↓TC, ↓LDL-c, =TG, ↓HDL-c ↓insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | Not reported |
| Abete et al. (2008)23 | n = 32 healthy obese men and women 8 weeks | Control (15%P; 30%L; 55%CHO) Fatty fish (15%P; 30%L; 55%CHO) + 3 fatty fish meals/week. | −5.3%, ↓SBP, ↓DBP −5.5%, ↓SBP, ↓DBP | ↑Insulin, =HOMA, =TC, =LDL-c, =TG, =HDL-c ↓Insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↑HDL-c | =leptin ↓leptin |
| Agus et al. (2000)26 | n = 10 moderately overweight young men Two 9-day periods separated by a washout period of 2–6 weeks | Low-GI (43%CHO; 27%P; 30%L) High-GI (67%CHO; 15%P; 18%L) | −3.6 kg −3.2 kg Low-GI diet increased hunger levels | ↓Insulin, REE decreased to a lesser extent | ↓leptin |
| Backes et al. (2008)16 | n = 23 overweight insulin-resistant women 3 months | Low-CHO (40% CHO) | −5.0 kg; ↓DBP | ↓Glucose, ↓TG | Not reported |
| Brehm et al. (2005)163 | n = 50 healthy obese women 4 months | Low-CHO (ad libitum) Low-fat (15%P; 30%L; 55%CHO) | −9.7±0.7 kg −6.1±0.9 kg | No differences between diets on glucose, insulin, and HOMA. Both diets: ↓TC, ↓LDL-c, and ↓ TG ↑ HDL-c with low-CHO | Not reported |
| Brinkworth et al. (2009)14 | n = 118 men and women with abdominal obesity and at least one additional metabolic syndrome risk factor 1 year | LC (4%CHO; 35%P; 61%L) LF (46%CHO; 24%P; 30%L) | −14.5 kg; ↓SBP; ↓DBP −11.5 k; ↓SBP; ↓DBP | Both diets: ↓glucose, ↓insulin, ↓HOMA-IR LC: ↓TG, ↑LDL-c, ↑HDL-c, ↑Apo-B | ↓CRP independently of diet composition |
| Buscemi et al. (2009)137 | n = 20 healthy obese women 2 months | Atkin (30%P; 65%L; 5%CHO) Mediterranean (20%P; 25%L; 55%CHO) | −7.6 kg;↓SBP; ↓DBP −4.9 kg | Both diets: ↓insulin, ↓HOMA, ↓TC, ↓LDL-c, =HDL-c, =TG | Both diets =IL-6, =TNF-α, =adiponectin, =8-iso-PGF2α, |
| Claessens et al. (2009)37 | n = 48 healthy obese 6-week weight loss + 12-week maintenance | High-CHO (ad libitum) >55%CHO; 30%L High-P (ad libitum) >25%P; 30%L | +1.1 kg −1.1 kg; ↓SBP; ↓DBP | Both diets: =insulin, =HOMA, ↑TC, ↑LDL-c and ↑HDL-c Only high-P: ↓TG | Both diets ↓adiponectin, ↑leptin |
| Clifton et al. (2008)10 | n = 79 healthy overweight / obese women 64 weeks (12-week weight loss + 52-week follow-up) | HP (34%P; 20%L; 46%CHO) HC (64%CHO; 20%L; 17%P) <10% of saturated fat in both diets | −4.6 kg −4.4 kg | ↓Glucose, ↓insulin, ↓LDL-c, ↓TG, ↑HDL-c with no differences between diets | Both diets: ↓CRP, ↓homocysteine |
| Crujeiras et al. (2007)145 | n = 32 healthy obese men/women 8 weeks | Control (15%P; 30%L; 55%CHO) Legume (15%P; 30%L; 55%CHO) (4 legume meals/week) | −5.3%; ↓SBP; ↓DBP −7.7%; ↓SBP; ↓DBP | ↑Insulin, ↑HOMA, ↓TC, ↓LDL-c, ↑TG, ↓HDL-c ↓Insulin, ↓HOMA, ↓TC, ↓LDL-c, ↓TG, ↓HDL-c | =ox-LDL,=MDA, =8-iso-PGF2α ↓ox-LDL, ↓MDA, ↓8-iso-PGF2α, ↑AOP |
| Dumesnil et al. (2001)134 | n = 12 healthy obese men two 6-day experimental periods with a 2-week washout period | Low-fat-high-CHO-high-P (30%P; 30%L; 40%CHO) ad libitum AHA (15%P; 30%L; 55%CHO) ad libitum | −2.3 kg; Higher satiety level No change in body weight Low-satiety level | ↓Insulin, =TC, =LDL-c, ↓TG, =HDL-c =insulin, =TC, =LDL-c, ↑TG, ↓HDL-c | Not reported |
| Das et al. (2007)46 | n = 34 healthy overweight adults 12 months | High-glycemic load (20%P; 20%L; 60%CHO) Low-glycemic load (30%P; 30%L; 40%CHO) | −8.0% −7.8% | Both diets: ↓insulin, ↓TC, ↓LDL-c, ↓TG, and ↑HDL-c | Not reported |
| Foster et al. (2003)42 | n = 63 obese men and women 1 year Poor adherence in both diets | Low-CHO high-P high-L (ad libitum) High-CHO (60%CHO; 25%L; 15%P) | −7.3%; =SBP;↓DBP −4.5%; =SBP;↓DBP | ↑HDL-c; ↓TG Both diets: ↓insulin; =TC; =LDL-c | Not reported |
| Gardner et al. (2007)164 | n = 77 n = 79 n = 79 n = 76 Obese women 1 year of intervention | Atkins Zone (30%P; 30%L; 40%CHO) Learn (15%P; 30%L; 55%CHO) Ornish (very-high-CHO) All diets were ad libitum | −4.7 kg, ↓SBP; ↓DBP −1.6 kg, ↓SBP; ↓DBP −2.2 kg, ↓SBP; ↓DBP −2.6 kg, ↓SBP; =DBP | ↓Insulin, =LDL-c, ↑HDL-c, ↓TG ↓Insulin, =LDL-c, ↑HDL-c, =TG ↓Insulin, =LDL-c, ↑HDL-c, ↓TG =Insulin, ↓LDL-c, ↓HDL-c, ↓TG | Not reported |
| Layman et al. (2009)64 | n = 130 4-months No differences in compliance between groups | PRO-group (30%P; 30%L; 40%CHO) CHO-group (15%P; 30%L; 55%CHO) | BW no differences between groups PRO-group greater ↓fat mass | PRO-d: ↓TG, ↑HDL-c CHO-d: ↓TC, ↓LDL-c, ↓HDL-c | Not reported |
| Noakes et al. (2005)135 | n = 100 healthy obese women 12 weeks | High-P (34%P; 20%L; 46%CHO) High-CHO (17%P; 20%L; 64% CHO) Both diets ≤10% saturated fat | −7.6 kg −6.9 kg | High-P: ↓TG, ↑B-12 Both diets: ↓glucose, ↓insulin, ↓LDL-c, ↓HDL-c, ↑folate, ↑B-6 | Both diets =homocysteine, ↓CRP |
| O'Brien et al. (2005)165 | n = 42 3 months Obese women | Low-fat (15%P; 30%L; 55%CHO) Very-low-CHO (ad libitum) | −4.3 ± 3.5 kg; ↓SBP; ↓DBP −7.6 ± 3.2 kg; ↓SBP; ↓DBP | Both diets: ↓insulin, ↓HOMA-IR; ↓TC and ↓LDL-c. Only very-low-CHO ↓TG | Both diets ↓CRP; ↓SAA |
| Petersen et al. (2006)56 | n = 771 healthy obese adults 10 weeks | Low-fat (20–25%L; 60–65%CHO; 15%P) High-fat (40–45%%L; 60–65%CHO; 15%P) | −6.9 kg −6.6 kg | ↓TC, ↓LDL-c, ↓TG, ↓HDL-c (changes were higher in this group) ↓TC, ↓LDL-c, ↓TG, ↓HDL-c Both groups:↓insulin | Not reported |
| Raben et al. (2002)44 | n = 398 obese subjects 6 months | Low-fat high-CHO high-GI (ad libitum) Low-fat high-CHO low-GI (ad libitum) | −1.7 kg −2.6 kg | Both diets improved glucose, insulin, and fasting blood lipids | Not reported |
| Seshadri et al. (2004)166 | n = 78 severe obese subjects 31 were diabetics, 36 had metabolic syndrome 49 with hypertension medication and 35 with lipid-lowering medication 6 months | Low-CHO (<30 g CHO; ad libitum) Conventional (≤30%L) energy restricted | −8.5 kg −3.5 kg | ↓insulin; ↓TG Both diets: =TC; =LDL-c; =HDL-c | ↓CRP |
| Skov et al. (1999)31 | n = 65 healthy overweight and obese men and women 6 months | High-P (25%P; 30%L; 45%CHO) High-CHO (12%P; 30%L; 58%CHO) | −8.9 kg −5.1 kg | ↓TG; ↓FFA | Not reported |
| Sloth et al. (2009)84 | n = 131 healthy overweight and obese subjects 6 months weight maintenance period | After losing 8% of the initial body weight, participants were instructed to follow ad libitum diets: MUFA, moderate fat (35–45%L; >20%MUFA) Low-F (20–30%L) Control (35%L; >15% SFA) | There was a significant weight increase with no differences between groups | Low-L and MUFA groups had lower insulin concentrations HbA1c increased in the control group There were no group differences in GLP-1; GLP-2 | Not reported |
| Stern et al. (2004)43 | n = 132 severe obese subjects 83% had diabetes or metabolic syndrome 1 year | Low-CHO (<30 g CHO; ad libitum) Conventional (≤30%L) energy restricted | −5.1 kg −3.1 kg Both: =SBP; =DBP | ↓TG; =HDL-c; ↓HBA1C ↓HDL-c; =HBA1C Both: =glucose; =insulin, =TC; =LDL-c | Not reported |
Abbreviations: 8-iso-PGF2α, 8-isoprostane F2α; AHA, American Heart Association; AOP, total plasma antioxidant power; BW, body weight; CHO, carbohydrates; CRP, C-reactive protein; FFA, free fatty acids; FM, fat mass; HBA1C, hemoglobin A1C; L, lipids; MDA, malondialdehyde; MUFA, monounsaturated fatty acids; ox-LDL, oxidized-LDL.
The ideal diet should be personalized to each patient and should include those dietary factors that imbue healthy and satiating dietary habits that are beneficial not only for effective weight loss, but are also good for body-weight maintenance as well as for reducing cardiovascular risk. Thus, a currently available diet may be one that has a moderate protein content (30%), high monounsaturated and omega-3 FAs, low-GI carbohydrates (40%), and includes adequate quantities of fiber, isoflavones, calcium, and antioxidant minerals. Since adherence to healthy dietary patterns can be difficult, meal replacement and dietary supplements should be considered as effective strategies for weight loss, weight maintenance, and treatment of MetS. Several factors such as genetics, physical activity, psychopathological conditions, obesity type, gender, age, or yo-yo cycles may influence the outcome of any dietary intervention.161 They should be taken into account as much as possible when dietary advice is prescribed for body weight management.
Declaration of interest
Some authors (IT and JAM) have been directly involved in the SEAFOODplus project (FOOD-CT-2004-506359), while others (AA and JAM) have been involved in the DIOGENES project (FOOD-CT-2005-513946).
REFERENCES