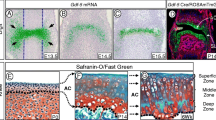
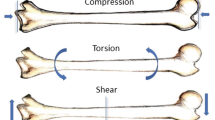
Abstract
Two major contributions to the development of articular cartilage are growth factors and mechanical loading. Growth factors have long been used to modulate the secretion of certain molecules from different cells. The TGF-β superfamily, specifically the BMPs, CDMPs, OPs, and GDFs, have a dramatic effect on the development of bone and cartilage tissue. These growth factors help produce an extracellular matrix that can withstand extreme loading conditions in the body. In addition to growth factors, it is known that mechanical forces stimulate the synthesis of extracellular proteins in vitro and in vivo and can affect the tissue's overall structure. Load-bearing tissue, such as articular cartilage, will atrophy in the absence of mechanical forces, and this observation has caused researchers to incorporate mechanical stimulation into the tissue engineering process. This article focuses on the importance of mechanical forces in tissue engineering of articular cartilage and the growth factors that help stimulate the formation of load-bearing tissue. © 2003 Biomedical Engineering Society.
PAC2003: 8780Rb, 8719Rr, 8715La, 8718La
Similar content being viewed by others
References
Alfredson, H., and R. Lorentzon. Superior results with continuous passive motion compared to active motion after periosteal transplantation. A retrospective study of human patella cartilage defect treatment. Knee Surg. Sports Traumatol. Arthrosc7:232–238, 1999.
Arevalo-Silva, C. A., Y. Cao, Y. Weng, M. Vacanti, A. Rodriguez, C. A. Vacanti, and R. D. Eavey. The effect of fibroblast growth factor and transforming growth factor-beta on porcine chondrocytes and tissue-engineered autologous elastic cartilage. Tissue Eng.7:81–88, 2001.
Athanasiou, K. A., A. Agarwal, and F. J. Dzida. Comparative study of the intrinsic mechanical properties of the human acetabular and femoral head cartilage. J. Orthop. Res.12:340–349, 1994.
Athanasiou, K. A. and G. Constantinides. 1995, Board of Regents, University of Texas System: USA Patent No. 5,433,215.
Athanasiou, K. A., G. Constantinides, and D. R. Lanctot. 1996, Board of Regents, University of Texas System: USA Patent No. 5,503,162.
Athanasiou, K. A., G. Constantinides, and D. R. Lanctot.1997, Board of Regents, University of Texas System: USA Patent No. 5,673,708.
Athanasiou, K. A., D. Korvick, and R. C. Schenck. Biodegradable implants for the treatment of osteochondral defects in a goat model. Tissue Eng.3:363–373, 1997.
Athanasiou, K. A., M. P. Rosenwasser, J. A. Buckwalter, M. Olmstead, and V. C. Mow. Biomechanical modeling of repair articular cartilage: Effects of passive motion on osteochondral defects in monkey knee joints. Tissue Eng.4:185–195, 1998.
Athanasiou, K. A., A. R. Shah, R. J. Hernandez, and R. G. LeBaron. Basic science of articular cartilage repair. Clin. Sports Med.20:223–247, 2001.
Bashir, A., M. L. Gray, J. Hartke, and D. Burstein. Nondestructive imaging of human cartilage glycosaminoglycan concentration by MRI. Magn. Reson. Med.41:857–865, 1999.
Below, S., S. P. Arnoczky, J. Dodds, C. Kooima, and N. Walter. The split-line pattern of the distal femur: A consideration in the orientation of autologous cartilage grafts. Arthroscopy18:613–617, 2002.
Blunk, T., A. L. Sieminski, K. J. Gooch, D. L. Courter, A. P. Hollander, A. M. Nahir, R. Langer, G. Vunjak-Novakovic, and L. E. Freed. Differential effects of growth factors on tissue-engineered cartilage. Tissue Eng.8:73–84, 2002.
Bonassar, L. J., A. J. Grodzinsky, E. H. Frank, S. G. Davila, N. R. Bhaktav, and S. B. Trippel. The effect of dynamic compression on the response of articular cartilage to insulin-like growth factor-I. J. Orthop. Res.19:11–17, 2001.
Chaipinyo, K., B. W. Oakes, and M. P. van Damme. Effects of growth factors on cell proliferation and matrix synthesis of low-density, primary bovine chondrocytes cultured in collagen I gels. J. Orthop. Res.20:1070–1078, 2002.
Darling, E. M., and K. A. Athanasiou. Articular cartilage bioreactors and bioprocesses. Tissue Eng.9:9–26, 2003.
Darling, E. M., and K. A. Athanasiou. Bioactive scaffold design for articular cartilage engineering. In: Biomedical Technology and Devices Handbook. Boca Raton, FL: CRC Press(in press).
Davisson, T., S. Kunig, A. Chen, R. Sah, and A. Ratcliffe. Static and dynamic compression modulate matrix metabolism in tissue engineered cartilage. J. Orthop. Res.20:842–848, 2002.
Davisson, T., R. L. Sah, and A. Ratcliffe. Perfusion increases cell content and matrix synthesis in chondrocyte three-dimensional cultures. Tissue Eng.8:807–816, 2002.
Drexler, W., D. Stamper, C. Jesser, X. Li, C. Pitris, K. Saunders, S. Martin, M. B. Lodge, J. G. Fujimoto, and M. E. Brezinski. Correlation of collagen organization with polarization sensitive imaging of in vitro cartilage: implications for osteoarthritis. J. Rheumatol.28:1311–1318, 2001.
Dunkelman, N. S., M. P. Zimber, R. G. LeBaron, R. Pavelec, M. Kwan, and A. F. Purchio. Cartilage production by rabbit articular chondrocytes on polyglycolic acid scaffolds in a closed bioreactor system. Biotechnol. Bioeng.46:299–305, 1995.
Erlacher, L., C. K. Ng, R. Ullrich, S. Krieger, and F. P. Luyten. Presence of cartilage-derived morphogenetic proteins in articular cartilage and enhancement of matrix replacement. Arthritis Rheum.41:263–273, 1998.
Forslund, C., and P. Aspenberg. CDMP-2 induces bone or tendon-like tissue depending on mechanical stimulation. J. Orthop. Res.20:1170–1174, 2002.
Frenkel, S. R., P. B. Saadeh, B. J. Mehrara, G. S. Chin, D. S. Steinbrech, B. Brent, G. K. Gittes, and M. T. Longaker. Transforming growth factor beta superfamily members: Role in cartilage modeling. Plast. Reconstr. Surg.105:980–990, 2000.
Gooch, K. J., T. Blunk, D. L. Courter, A. L. Sieminski, P. M. Bursac, G. Vunjak-Novakovic, and L. E. Freed. IGF-I and mechanical environment interact to modulate engineered cartilage development. Biochem. Biophys. Res. Commun.286:909–915, 2001.
Gooch, K. J., T. Blunk, D. L. Courter, A. L. Sieminski, G. Vunjak-Novakovic, and L. E. Freed. Bone morphogenetic proteins-2,-12, and-13 modulate development of engineered cartilage. Tissue Eng.8:591–601, 2002.
Grimaud, E., D. Heymann, and F. Redini. Recent advances in TGF-beta effects on chondrocyte metabolism. Potential therapeutic roles of TGF-beta in cartilage disorders. Cytokine Growth Factor Rev.13:241–257, 2002.
Gruber, R., C. Mayer, K. Bobacz, M. T. Krauth, W. Graninger, F. P. Luyten, and L. Erlacher. Effects of cartilage-derived morphogenetic proteins and osteogenic protein-1 on osteochondrogenic differentiation of periosteum-derived cells. Endocrinology142:2087–2094, 2001.
Guerne, P. A., A. Sublet, and M. Lotz. Growth factor responsiveness of human articular chondrocytes: distinct profiles in primary chondrocytes, subcultured chondrocytes, and fibroblasts. J. Cell Physiol.158:476–484, 1994.
Hodge, W. A., K. L. Carlson, R. S. Fijan, R. G. Burgess, P. O. Riley, W. H. Harris, and R. W. Mann. Contact pressures from an instrumented hip endoprosthesis. J. Bone Jt. Surg., Am. Vol.71:1378–1386, 1989.
Hu, J. C. Y., and K. A. Athanasiou. The role of mechanical forces in tissue engineering of articular cartilage, In: Functional Tissue Engineering. New York: Springer (in press).
Ikenoue, T., M. C. Trindade, M. S. Lee, E. Y. Lin, D. J. Schurman, S. B. Goodman, and R. L. Smith. Mechanoregulation of human articular chondrocyte aggrecan and type II collagen expression by intermittent hydrostatic pressure. J. Orthop. Res.21:110–116, 2003.
Kaps, C., C. Bramlage, H. Smolian, A. Haisch, U. Ungethum, G. R. Burmester, M. Sittinger, G. Gross, and T. Haupl. Bone morphogenetic proteins promote cartilage differentiation and protect engineered artificial cartilage from fibroblast invasion and destruction. Arthritis Rheum.46:149–162, 2002.
Kim, H. K., R. G. Kerr, T. F. Cruz, and R. B. Salter. Effects of continuous passive motion and immobilization on synovitis and cartilage degradation in antigen induced arthritis. J. Rheumatol.22:1714–1721, 1995.
Koay, E. J., A. C. Shieh, and K. A. Athanasiou. Creep indentation of single cells. J. Biomech. Eng.125:334–341, 2003.
Laasanen, M. S., J. Toyras, J. Hirvonen, S. Saarakkala, R. K. Korhonen, M. T. Nieminen, I. Kiviranta, and J. S. Jurvelin. Novel mechano-acoustic technique and instrument for diagnosis of cartilage degeneration. Physiol. Meas23:491–503, 2002.
Laurent, D., J. Wasvary, J. Yin, M. Rudin, T. C. Pellas, and E. O'Byrne. Quantitative and qualitative assessment of articular cartilage in the goat knee with magnetization transfer imaging. Magn. Reson. Imaging19:1279–1286, 2001.
Lee, M. S., T. Ikenoue, M. C. Trindade, N. Wong, S. B. Goodman, D. J. Schurman, and R. L. Smith. Protective effects of intermittent hydrostatic pressure on osteoarthritic chondrocytes activated by bacterial endotoxin. J. Orthop. Res.21:117–122, 2003.
Luyten, F. P., Y. M. Yu, M. Yanagishita, S. Vukicevic, R. G. Hammonds, and A. H. Reddi. Natural bovine osteogenin and recombinant human bone morphogenetic protein-2B are equipotent in the maintenance of proteoglycans in bovine articular cartilage explant cultures. J. Biol. Chem.267:3691–3695, 1992.
Lyyra, T., I. Kiviranta, U. Vaatainen, H. J. Helminen, and J. S. Jurvelin. characterization of indentation stiffness of articular cartilage in the normal human knee. J. Biomed. Mater. Res.48:482–487, 1999.
Madry, H., R. Padera, J. Seidel, R. Langer, L. E. Freed, S. B. Trippel, and G. Vunjak-Novakovic. Gene transfer of a human insulin-like growth factor I cDNA enhances tissue engineering of cartilage. Hum. Gene Ther.13:1621–1630, 2002.
Mattioli-Belmonte, M., A. Gigante, R. A. Muzzarelli, R. Politano, A. De Benedittis, N. Specchia, A. Buffa, G. Biagini, and F. Greco. N,N-dicarboxymethyl chitosan as delivery agent for bone morphogenetic protein in the repair of articular cartilage. Med. Biol. Eng. Comput.37:130–134, 1999.
Mauck, R. L., M. A. Soltz, C. C. Wang, D. D. Wong, P. H. Chao, W. B. Valhmu, C. T. Hung, and G. A. Ateshian. Functional tissue engineering of articular cartilage through dynamic loading of chondrocyte-seeded agarose gels. J. Biomech. Eng.122:252–260, 2000.
Miura, Y., J. Parvizi, J. S. Fitzsimmons, and S. W. O'Driscoll. Brief exposure to high-dose transforming growth factor-betal enhances periosteal chondrogenesis: A preliminary report. J. Bone Jt. Surg., Am. Vol.84–A:793–799, 2002.
Mizuno, S., T. Tateishi, T. Ushida, and J. Glowacki. Hydrostatic fluid pressure enhances matrix synthesis and accumulation by bovine chondrocytes in three-dimensional culture. J. Cell Physiol.193:319–327, 2002.
Mow, V. C., E. L. Flatow, and G. A. Ateshian. Biomechanics, In: Orthopaedic Basic Science: Biology and Biomechanics of the Musculoskeletal System, edited by J. A. Buckwalter, T. A. Einhorn, and S. R. Simon. American Academy of Orthopaedic Surgeons, 2000, pp. 140–142.
Niederauer, G. G., G. M. Niederauer, L. C. J. Cullen, K. A. Athanasiou, J. B. Thomas, and M. Q. Niederauer.Trans. BMES.
Niederauer, G. G., D. R. Schmidt, J. C. DeLee, K. A. Athanasiou, J. B. Thomas, H. M. Aberman, T. M. Simon, D. W. Jackson, G. M. Niederauer, and M. Q. Niederauer. Trans. ICRS.
Nieminen, M. T., J. Rieppo, J. Silvennoinen, J. Toyras, J. M. Hakumaki, M. M. Hyttinen, H. J. Helminen, and J. S. Jurvelin. Spatial assessment of articular cartilage proteoglycans with Gd-DTPA-enhanced T1 imaging. Magn. Reson. Med.48:640–648, 2002.
O'Connor, W. J., T. Botti, S. N. Khan, and J. M. Lane. The use of growth factors in cartilage repair. Orthop. Clin. North Am.31:399–410, 2000.
Pacifici, M., E. Koyama, M. Iwamoto, and C. Gentili. Development of articular cartilage: what do we know about it and how may it occur?Connect. Tissue Res.41:175–184, 2000.
Paul, P. K., E. O'Byrne, V. Blancuzzi, D. Wilson, D. Gunson, F. L. Douglas, J. Z. Wang, and R. S. Mezrich. Magnetic resonance imaging reflects cartilage proteoglycan degradation in the rabbit knee. Skeletal Radiol.20:31–36, 1991.
Pecina, M., M. Jelic, S. Martinovic, M. Haspl, and S. Vukicevic. Articular cartilage repair: The role of bone morphogenetic proteins. Int. Orthop.26:131–136, 2002.
Pei, M., L. A. Solchaga, J. Seidel, L. Zeng, G. Vunjak-Novakovic, A. I. Caplan, and L. E. Freed. Bioreactors mediate the effectiveness of tissue engineering scaffolds. FASEB J.16:1691–1694, 2002.
Perka, C., U. Arnold, R. S. Spitzer, and K. Lindenhayn. The use of fibrin beads for tissue engineering and subsequential transplantation. Tissue Eng.7:359–361, 2001.
Reddi, A. H.Role of morphogenetic proteins in skeletal tissue engineering and regeneration. Nat. Biotechnol.16:247–252, 1998.
Schinagl, R. M., M. K. Ting, J. H. Price, and R. L. Sah. Video microscopy to quantitate the inhomogeneous equilibrium strain within articular cartilage during confined compression. Ann. Biomed. Eng.24:500–512, 1996.
Shieh, A. C., and K. A. Athanasiou. Principles of cell mechanics for cartilage tissue engineering. Ann. Biomed. Eng.31:1–11, 2003.
Shin, D., and K. Athanasiou. Cytoindentation for obtaining cell biomechanical properties. J. Orthop. Res.17:880–890, 1999.
Smith, R. L., B. S. Donlon, M. K. Gupta, M. Mohtai, P. Das, D. R. Carter, J. Cooke, G. Gibbons, N. Hutchinson, and D. J. Schurman. Effects of fluid-induced shear on articular chondrocyte morphology and metabolism. J. Orthop. Res.13:824–831, 1995.
Sporn, M. B., A. B. Roberts, L. M. Wakefield, and R. K. Assoian. Transforming growth factor-beta: Biological function and chemical structure. Science233:532–534, 1986.
Suh, J. K., I. Youn, and F. H. Fu. An calibration of an ultrasound transducer: A potential application for an ultrasonic indentation test of articular cartilage. J. Biomech.34:1347–1353, 2001.
Toyras, J., T. Lyyra-Laitinen, M. Niinimaki, R. Lindgren, M. T. Nieminen, I. Kiviranta, and J. S. Jurvelin. Estimation of the Young's modulus of articular cartilage using an arthroscopic indentation instrument and ultrasonic measurement of tissue thickness. J. Biomech.34:251–256, 2001.
Uchio, Y., M. Ochi, N. Adachi, K. Kawasaki, and J. Iwasa. Arthroscopic assessment of human cartilage stiffness of the femoral condyles and the patella with a new tactile sensor. Med. Eng. Phys.24:431–435, 2002.
Valcourt, U., J. Gouttenoire, A. Moustakas, D. Herbage, and F. Mallein-Gerin. Functions of transforming growth factor-beta family type I receptors and Smad proteins in the hypertrophic maturation and osteoblastic differentiation of chondrocytes. J. Biol. Chem.277:33545–33545, 2002.
van der Kraan, P., E. Vitters, and W. van den Berg. Differential effect of transforming growth factor beta on freshly isolated and cultured articular chondrocytes. J. Rheumatol.19:140–145, 1992.
Vanwanseele, B., F. Eckstein, H. Knecht, E. Stussi, and A. Spaepen. Knee cartilage of spinal cord-injured patients displays progressive thinning in the absence of normal joint loading and movement. Arthritis Rheum.46:2073–2078, 2002.
Verschure, P. J., L. A. Joosten, P. M. van der Kraan, and W. B. Van den Berg. Responsiveness of articular cartilage from normal and inflamed mouse knee joints to various growth factors. Ann. Rheum. Dis.53:455–460, 1994.
Vunjak-Novakovic, G., N. Searby, J. De Luis, and L. E. Freed. Microgravity studies of cells and tissues. Ann. N.Y. Acad. Sci.974:504–517, 2002.
Weisser, J., B. Rahfoth, A. Timmermann, T. Aigner, R. Brauer, and K. von der Mark. Role of growth factors in rabbit articular cartilage repair by chondrocytes in agarose. Osteoarthritis Cartilage9, Suppl. A:S48–54, 2001.
Ziegler, J., U. Mayr-Wohlfart, S. Kessler, D. Breitig, and K. P. Gunther. Adsorption and release properties of growth factors from biodegradable implants. J. Biomed. Mater. Res.59:422–428, 2002.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Darling, E.M., Athanasiou, K.A. Biomechanical Strategies for Articular Cartilage Regeneration. Annals of Biomedical Engineering 31, 1114–1124 (2003). https://doi.org/10.1114/1.1603752
Issue Date:
DOI: https://doi.org/10.1114/1.1603752