Abstract
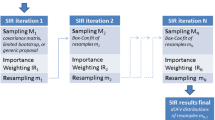
Efficient power calculation methods have previously been suggested for Wald test-based inference in mixed-effects models but the only available alternative for Likelihood ratio test-based hypothesis testing has been to perform computer-intensive multiple simulations and re-estimations. The proposed Monte Carlo Mapped Power (MCMP) method is based on the use of the difference in individual objective function values (ΔiOFV) derived from a large dataset simulated from a full model and subsequently re-estimated with the full and reduced models. The ΔiOFV is sampled and summed (∑ΔiOFVs) for each study at each sample size of interest to study, and the percentage of ∑ΔiOFVs greater than the significance criterion is taken as the power. The power versus sample size relationship established via the MCMP method was compared to traditional assessment of model-based power for six different pharmacokinetic and pharmacodynamic models and designs. In each case, 1,000 simulated datasets were analysed with the full and reduced models. There was concordance in power between the traditional and MCMP methods such that for 90% power, the difference in required sample size was in most investigated cases less than 10%. The MCMP method was able to provide relevant power information for a representative pharmacometric model at less than 1% of the run-time of an SSE. The suggested MCMP method provides a fast and accurate prediction of the power and sample size relationship.





Similar content being viewed by others
References
Sheiner LB. Learning versus confirming in clinical drug development. Clin Pharmacol Ther. 1997;61(3):275–91.
Statistical guide for clinical pharmacology & therapeutics. Clin Pharmacol Ther. 2010;88(2):150–152.
Yuh L, Beal S, Davidian M, Harrison F, Hester A, Kowalski K, et al. Population pharmacokinetic/pharmacodynamic methodology and applications: a bibliography. Biometrics. 1994;50(2):566–75.
Zhang L, Sinha V, Forgue ST, Callies S, Ni L, Peck R, et al. Model-based drug development: the road to quantitative pharmacology. J Pharmacokinet Pharmacodyn. 2006;33(3):369–93.
Lalonde RL, Kowalski KG, Hutmacher MM, Ewy W, Nichols DJ, Milligan PA, et al. Model-based drug development. Clin Pharmacol Ther. 2007;82(1):21–32.
Karlsson KE. Benefits of pharmacometric model-based design and analysis of clinical trials. Uppsala: Acta Universitatis Upsaliensis; 2010.
Jonsson EN, Sheiner LB. More efficient clinical trials through use of scientific model-based statistical tests. Clin Pharmacol Ther. 2002;72(6):603–14.
Charles P, Giraudeau B, Dechartres A, Baron G, Ravaud P. Reporting of sample size calculation in randomised controlled trials: review. BMJ. 2009;338:b1732.
Rochon J. Application of GEE procedures for sample size calculations in repeated measures experiments. Stat Med. 1998;17(14):1643–58.
Bloch DA. Sample size requirements and the cost of a randomized clinical trial with repeated measurements. Stat Med. 1986;5(6):663–7.
Dahmen G, Rochon J, Konig IR, Ziegler A. Sample size calculations for controlled clinical trials using generalized estimating equations (GEE). Methods Inf Med. 2004;43(5):451–6.
Ogungbenro K, Aarons L, Graham G. Sample size calculations based on generalized estimating equations for population pharmacokinetic experiments. J Biopharm Stat. 2006;16(2):135–50.
Kang D, Schwartz JB, Verotta D. Sample size computations for PK/PD population models. J Pharmacokinet Pharmacodyn. 2005;32(5–6):685–701.
Retout S, Comets E, Samson A, Mentre F. Design in nonlinear mixed effects models: optimization using the Fedorov–Wynn algorithm and power of the Wald test for binary covariates. Stat Med. 2007;26(28):5162–79.
Ogungbenro K, Aarons L. Sample size/power calculations for repeated ordinal measurements in population pharmacodynamic experiments. J Pharmacokinet Pharmacodyn. Feb;37(1):67-83.
Ogungbenro K, Aarons L. Sample size/power calculations for population pharmacodynamic experiments involving repeated-count measurements. J Biopharm Stat. 2010;20(5):1026–42.
Wald A. Tests of statistical hypotheses concerning several parameters when the number of observations is large. T Am Math Soc. 1943;54(1–3):426–82.
Beal SL. Sample size determination for confidence intervals on the population mean and on the difference between two population means. Biometrics. 1989;45(3):969–77.
White DB, Walawander CA, Liu DY, Grasela TH. Evaluation of hypothesis testing for comparing two populations using NONMEM analysis. J Pharmacokinet Biopharm. 1992;20(3):295–313.
Ogungbenro K, Aarons L. How many subjects are necessary for population pharmacokinetic experiments? Confidence interval approach. Eur J Clin Pharmacol. 2008;64(7):705–13.
Kowalski KG, Hutmacher MM. Design evaluation for a population pharmacokinetic study using clinical trial simulations: a case study. Stat Med. 2001;20(1):75–91.
Lee PI. Design and power of a population pharmacokinetic study. Pharm Res. 2001;18(1):75–82.
Ette EI, Sun H, Ludden TM. Balanced designs in longitudinal population pharmacokinetic studies. J Clin Pharmacol. 1998;38(5):417–23.
Wahlby U, Bouw MR, Jonsson EN, Karlsson MO. Assessment of type I error rates for the statistical sub-model in NONMEM. J Pharmacokinet Pharmacodyn. 2002;29(3):251–69.
Wahlby U, Jonsson EN, Karlsson MO. Assessment of actual significance levels for covariate effects in NONMEM. J Pharmacokinet Pharmacodyn. 2001;28(3):231–52.
Beal SL, Sheiner LB, Boeckmann AJ, Bauer RJ. NONMEM User's guides. Ellicot City: MD: Icon Development Solutions; 1989–2010.
Lindbom L, Pihlgren P, Jonsson EN. PsN-Toolkit—a collection of computer intensive statistical methods for non-linear mixed effect modeling using NONMEM. Comput Methods Programs Biomed. 2005;79(3):241–57.
Hamren B, Bjork E, Sunzel M, Karlsson M. Models for plasma glucose, HbA1c, and hemoglobin interrelationships in patients with type 2 diabetes following tesaglitazar treatment. Clin Pharmacol Ther. 2008;84(2):228–35.
Ding AA, Wu H. Assessing antiviral potency of anti-HIV therapies in vivo by comparing viral decay rates in viral dynamic models. Biostatistics. 2001;2(1):13–29.
Hornestam B, Jerling M, Karlsson MO, Held P. Intravenously administered digoxin in patients with acute atrial fibrillation: a population pharmacokinetic/pharmacodynamic analysis based on the Digitalis in Acute Atrial Fibrillation trial. Eur J Clin Pharmacol. 2003;58(11):747–55.
Hennig S, Friberg L, Karlsson M. Characterizing time to conversion to sinus rhythm under digoxin and placebo in acute atrial fibrillation. PAGE 18 (2009) Abstr 1504 [www.page-meeting.org/?abstract=1504]; 2009.
Mentre F, Mallet A, Baccar D. Optimal design in random-effects regression models. Biometrika. 1997;84(2):429–42.
Foracchia M, Hooker A, Vicini P, Ruggeri A. POPED, a software for optimal experiment design in population kinetics. Comput Methods Programs Biomed. 2004;74(1):29–46.
Retout S, Duffull S, Mentre F. Development and implementation of the population Fisher information matrix for the evaluation of population pharmacokinetic designs. Comput Methods Programs Biomed. 2001;65(2):141–51.
Atkinson A, Donev AN. Optimum experimental designs. Oxford: Clarendon; 1992.
Bazzoli C, Retout S, Mentre F. Fisher information matrix for nonlinear mixed effects multiple response models: evaluation of the appropriateness of the first order linearization using a pharmacokinetic/pharmacodynamic model. Stat Med. 2009;28(14):1940–56.
Samson A, Lavielle M, Mentre F. The SAEM algorithm for group comparison tests in longitudinal data analysis based on non-linear mixed-effects model. Stat Med. 2007;26(27):4860–75.
McGree JM, Eccleston JA, Duffull SB. Simultaneous versus sequential optimal design for pharmacokinetic-pharmacodynamic models with FO and FOCE considerations. J Pharmacokinet Pharmacodyn. 2009;36(2):101–23.
Lacroix BD, Lovern MR, Stockis A, Sargentini-Maier ML, Karlsson MO, Friberg LE. A pharmacodynamic Markov mixed-effects model for determining the effect of exposure to certolizumab pegol on the ACR20 score in patients with rheumatoid arthritis. Clin Pharmacol Ther. 2009;86(4):387–95.
Vong C, Bergstrand M, Karlsson M. Rapid sample size calculations for a defined likelihood ratio test based power in mixed effects models’. PAGE 19 (2010) Abstr 1863 [www.page-meeting.org/?abstract=1863]; 2010.
Karlsson KE, Grahnen A, Karlsson MO, Jonsson EN. Randomized exposure-controlled trials; impact of randomization and analysis strategies. Br J Clin Pharmacol. 2007;64(3):266-77.
Acknowledgement
We would like to acknowledge Kajsa Harling for computational assistance.
Conflict of interest
The authors declared no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vong, C., Bergstrand, M., Nyberg, J. et al. Rapid Sample Size Calculations for a Defined Likelihood Ratio Test-Based Power in Mixed-Effects Models. AAPS J 14, 176–186 (2012). https://doi.org/10.1208/s12248-012-9327-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-012-9327-8