Abstract
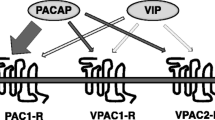
The effects of a vasoactive intestinal peptide (VIP) receptor antagonist (VIPhyb) on human glioblastoma cells were characterized. Pituitary adenylate cyclase activating polypeptide (125I-PACAP-27) bound with high affinity to U87, U118, and U373 cells. Specific 125I-PACAP-27 binding to U87 cells was inhibited, with high affinity, by PACAP but not VIP or VIPhyb (IC50=10, 1500, and 500 nM, respectively). By reverse transcriptasepolymerase chain reaction (RT-PCR), a major 305bp band was observed indicative of PAC1 receptors. PACAP-27 caused cAMP elevation and the increase in cAMP caused by PACAP-27, was inhibited by the VIPhyb. Also, PACAP-27 caused cytosolic Ca2+ elevation in Fura-2AM loaded U87 cells and the VIPhyb inhibited this increase. Using the MTT growth assay, the VIPhyb was shown to inhibit glioblastoma growth in a concentration-dependent manner. Using a clonogenic assay in vitro, 10 µM VIPhyb significantly inhibited proliferation of U87, U118, and U373 cells. In vivo, 0.4 µg/kg VIPhyb inhibited U87 xenograft proliferation in nude mice. These results suggest that the VIPhyb antagonizes PAC1 receptors on glioblastoma cells and inhibits their proliferation.
Similar content being viewed by others
References
Arimura A. (1992) Pituitary adenylate cyclase activating polypeptide (PACAP): discovery and current status of research. Reg. Peptides 37, 287–303.
Ashur-Fabian O., Giladi E., Brenneman D. E., and Gozes I. (1997) Identification of VIP/PACAP receptors on astrocytes using antisense oligodeoxynucleotides. J. Mol. Neurosci. 9, 211–222.
Bayraktar T., Staiger J. F., Acsady L., Cozzari C., Freund T. F., and Ailles, K. (1997) Co-localization of vasoactive intestinal polypeptide, gamma-aminobutyric acid and choline acetyltransferase in neocortical interneurons of the adult rat. Brain Res. 757, 209–217.
Bepler G. Zeymer U. Mahmoud S., Fiskum G., Palaszynski E., Totsch M., et al. (1988) Substance P analogues function as bombesin receptopr antagonists and inhibit small cell lung cancer clonal growth. Peptides 9, 1367–1372.
Brenneman D. E. and Eiden L. E. (1986) Vasoactive intestinal peptide and electrical activity influence neuronal survival. Proc. Natl. Acad. Sci. USA 83, 1159–1162.
Brenneman D. E. and Gozes I. (1996) A femtomolar-acting neuroprotective peptide. J. Clin. Invest. 97, 2299–2307.
Deschodt-Lanckman M., Robberecht P., and Christophe J. (1977) Characterization of VIP-sensitive adenylate cyclase activity in guinea pig brain. FEBS Lett. 83, 76–80.
DiCicco-Bloom E., Lu N., Pintar J. E., and Zhang J. (1998) The PACAP ligand/receptor system regulates cerebral cortical neurogenesis. Ann. NY Acad. Sci. 865, 274–289.
Duckles S. P. and Said S. I. (1982) Vasoactive intestinal peptide as a neurotransmitter in the cerebral circulation. Eur. J. Pharmacol. 78, 371–374.
Eden J. M., Hall M. D., Higginbottom M., Horwell D. C., Howson W., Hughes J., et al. (1996) PD165929-The first high affinity non-peptide neuromedin B (NMB) receptor selective antagonist. Bioorg. Med. Chem. Lett. 6, 2617–2623.
Etgen A. M. and Browning E. T. (1983) Activators of cyclic adenosine 3′-5′-monophosphate accumulation in rat hippocampal slices: action of vasoactive intestinal peptide (VIP). J. Neurosci. 3, 2487–2493.
Fishbein J. A., Coy D. H., Hocart S. J., Jiang N. Y., Mrozinski J. E. Jr., Mantey S. A., and Jensen R. T. (1994) A chimeric VIP-PACAP analogue but not VIP pseudopeptides function as VIP receptor antagonists. Peptides 15, 95–100.
Gottschall P. E., Tatsumo I., and Arimura A. (1994) Regulation of interleukin-6 (IL-6) secretion in primary cultured rat astrocytes: synergism of interleukin-1 (IL-1) and pituitary adenylate cyclase activating polypeptide (PACAP). Brain Res. 637, 197–203.
Gozes I., McCune S. K., Jacobson L., Warren D., Moody T. W., Fridkin M., and Brenneman D. E. (1991) An antagonist to vasoactive intestinal peptide affects cellular functions in the central nervous system. J. Pharm. Exp. Ther. 257, 959–966.
Granoth R., Fridkin M., Rubinraut S., and Gozes I. (2000a). VIP-derived sequences modified by N-terminal steryl moiety induces cell death: The human keratinocyte as a model. FEBS Lett. 475, 71–77.
Granoth R., Fridkin M., and Gozes I. (2000b). VIP and the potent analog, stearyl-Nle(17)VIP induce proliferation of keratinocytes. FEBS Lett. 475, 78–83.
Ishihara T., Shigemoto R., Mori K., Takahashi K., and Nagata S. (1992) Functional expression and tissue distribution of a novel receptor for vasoactive intestinal polypeptide. Neuron 8, 811–819.
Journot L., Villalba M., and Bockaert J. (1997) PACAP-38 protects cerebellar granule cells from apoptosis. Ann. NY Acad. Sci. 865, 100–110.
Levin V. A., Leibel S. A., and Gutin P. H. (1997) Neoplasms of the central nervous system, in Cancer Principles and Practice of Oncology (DeVita V. T. Jr., Hellman S., and Rosenberg S. A., eds.), Lippincott-Raven Publishers, Philadelphia New York, pp. 2022–2082.
Lilling G., Wollman Y., Goldstein M. N., Rubinraut S., Fridkin M., Brenneman D. E., and Gozes I. (1995) Inhibition of human neuroblastoma growth by a specific VIP antagonist. J. Mol. Neurosci. 5, 231–239.
Lutz E. M., Sheward W. J., West K. M., Morrow J. A., and Harmar A. J. (1993) The VIP2 receptor: molecular characterization of a cDNA encoding a novel receptor for vasoactive intestinal peptide. FEBS Lett. 334, 3–8.
Magistretti P. J., Morrison J. H., Shoemaker W. J., Sapin V., and Bloom F. E. (1981) Vasoactive intestinal polypeptide induces glycogenolysis in mouse cortical slices: a possible regulatory mechanism for the local control of energy metabolism. Proc. Natl. Acad. Sci. USA 78, 6535–6539.
Magistretti P. J. and Morrison J. H. (1988) Noradrenalineand vasoactive intestinal peptide containing neuronal systems in neocortex: Functional convergence with contrasting morphology. Neuroscience 24, 367–378.
Magistretti P. J., Cardinaux J. R., and Martin J. L. (1998) VIP and PACAP in the CNS: regulators of glial energy metabolism and modulators of glutamatergic signaling. Ann. NY Acad. Sci. USA 865, 213–225.
Mantey S. A., Weber H. C., Sainz E., Akeson M., Ryan R. R., Pradhan T. K., et al. (1997) Discovery of a high affinity radioligand for the human orphan receptor, bombesin receptor subtype 3, which demonstrates that it has a unique pharmacology compared with other mammalian bombesin receptors. J. Biol. Chem. 272, 26,062–26,071.
Maruno K. and Said S. I. (1993) Human small cell lung carcinoma inhibition by vasoactive intestinal peptide (VIP) and helodermin (Hd). Biomed. Res. 13, 373–375.
Moody T. W., Mahmoud S., Staley J. S., Naldini L., Cirillo D., South V., et al. (1989) Human glioblastoma cell lines have neuropeptide receptors for bombesin/GRP. J. Mol. Neurosci. 1, 235–242.
Moody T. W., Staley J., Zia F., Coy D. J., and Jensen R. T. (1992) Neuromedin B receptors are present on small cell lung cancer cells. J. Pharmacol. Exp. Therap. 263, 311–317.
Moody T. W., Zia F., Goldstein A., Naylor P., Sarin E., Brenneman D., et al. (1993a) VIP analogues inhibit small cell lung cancer growth. Biomed. Res. 13, 131–136.
Moody T. W., Zia F., Brenneman D., Fridkin M., Davidson A., and Gozes I. (1993b) A VIP antagonist inhibits the growth of non-small cell lung cancer. Proc. Natl. Acad. Sci. USA 90, 4345–4349.
Nielsen F.C., Gammertoft S., Westermark B., and Fahrenkrug J. (1990) High affinity receptors for vasoactive intestinal peptide on a human glioma cell line. Peptides 11, 1225–1231.
O’Dorisio M. S., Fleshman D. J., Qualman S. J., and O’Dorisio T. M. (1992) Vasoactive intestinal peptide: autocrine growth factor in neuroblastoma. Reg. Pept. 37, 213–226.
Pandol S. J., Dharmsathaphorn K., Schoeffield M. S., Vale W., and Rivier J. (1986) Vasoactive intestinal peptide receptor antagonist [4C1-D-Phe6, Leul7] VIP. Am. J. Physiol. 250 (4 Pt 1), G553-G557.
Pisegna J. R. and Wank S. A. (1993) Molecular cloning and functional expression of the pituitary adenylate cyclaseactivating polypeptide type I receptor. Proc. Natl. Acad. Sci. USA 90, 6345–6349.
Quick M., Iverson L. L., and Bloom S. R. (1978) Effect of vasoactive intestinal peptide (VIP) and other peptides on cAMP accumulation in rat brain. Biochem. Pharmacol. 27, 2209–2213.
Reubi J. C. (2000). In vitro evaluation of VIP/PACAP receptors in healthy and diseased human tissues: Clinical implication. New York Acad. Sci. 921, 1–25
Robberecht P., Woussen-Colle M. C., Vertongen P., DeNeef P., Hou X., Slamon I., and Brotchi J. (1994) Expression of pituitary adenylated cyclase activating polypeptide (PACAP) receptors in human glial cell tumors. Peptides 15, 661–665.
Said S. I. and Mutt V. (1970) Polypeptide with broad biological activity: Isolation from the small intestine. Science 69, 1217–1218.
Sethi T., Langdon S., Smyth J., and Rozengurt E. (1992) Growth of small cell lung cancer cells: Stimulation by multiple neuropeptides and inhibition by a broad spectrum antagonist in vitro and in vivo. Cancer Res. 52, 2737s-2742s.
Shaffer M. M., Carney D. N., Korman L. Y., Lebovic G. S., and Moody T. W. (1987) High affinity binding of VIP to human lung cancer cell lines. Peptides 8, 1101–1106.
Spengler D., Waeber C., Pantaloni C., Holsboer F., Bockaert J., Seeburg P. H., and Journot L. (1993) Differential signal transduction by five splice variants of the PACAP receptor.
Vertongen P., Camby I., Darro F., Kiss R., and Robberecht P. (1996) VIP and pituitary adenylate cyclase activating polypeptide (PACAP) have an antiproliferative effect on the T98G human glioblastoma cell line through interaction with VIP2 receptors. Neuropeptides 30, 492–496.
Waelbroeck M., Robberecht P., Coy D. H., Camus J. C., De Neef P., and Christophe J. (1985) Interaction of growth hormone-releasing factor (GRF) and 14 GRF analogs with vasoactive intestinal peptide (VIP) receptors of rat pancreas. Discovery of (N-Ac-Tyr1, D-Phe2)-GRF (1-29)-NH2 as a VIP antagonist. Endocrinology 116, 2643–2649.
Woll P. J. and Rozengurt E. (1988) D-Arg1, D-Phe5, D-Trp7,9, Leu11 substance P, a potent bombesin antagonist in murine Swiss 3T3 cells, inhibit the growth of human small cell lung cancer. Proc. Natl. Acad. Sci. USA 85, 1859–1863.
Wollman Y., Liling G., Goldstein M. N., Fridkin M., and Gozes I. (1993) Vasoactive intestinal peptide: A growth promoter in neuroblastoma cells. Brain Res. 624, 339–341.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, A., Walters, J., Gozes, Y. et al. A vasoactive intestinal peptide antagonist inhibits the growth of glioblastoma cells. J Mol Neurosci 17, 331–339 (2001). https://doi.org/10.1385/JMN:17:3:331
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:17:3:331