Abstract
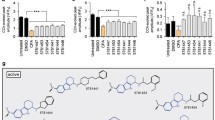
We screened for drugs that specifically interact with the 5′-untranslated region of the mRNA encoding the Alzheimer’s amyloid precursor protein (APP). Our goal was to use newly discovered APP 5′ UTR directed compounds to limit amyloid-β (Aβ)-peptide output in cell culture systems. The APP 5′ UTR folds into a stable RNA secondary structure (Gibbs free energy: ΔG=−54.9 kcal/mol) and is an important regulator of the amount of APP translated in response to IL-1 (Nilsson et al., 1998; Rogers et al., 1999) and iron (Rogers et al., 2002). Seventeen drug “hits” were identified from a library of 1,200 FDA preapproved drugs (Rogers et al., 2002). Six of the original 17 compounds were validated for their capacity to suppress reporter gene expression in stable neuroblastoma transfectants expressing the dicistronic reporter construct shown in Fig. 2. These six leads suppressed APP 5′ UTR driven luciferase translation while causing no effect on the translation of dicistronic GFP gene translated from a viral IRES (negative control to ensure specificity during drug screens). In this report, we show that paroxetine (serotonin reuptake blocker) and dimercaptopropanol (Hg chelator) exerted significant effects on APP expression (steady-state levels of APP), whereas Azithromycin altered APP processing. None of these three compounds altered APLP-1 expression. In the future, we will identify further novel compounds that influence Aβ levels, either via translation inhibition or by changing the activity of proteins coupled between APP translation and APP processing.
Similar content being viewed by others

References
Arjona A., Pooler A., Lee R., and Wurtman R. (2002) Effect of a 5-HT(2C) serotonin agonist, dexnorfenfluramine, on amyloid precursor protein metabolism in guinea pigs. Brain Res. 951, 135.
Buxbaum J., Oishi M., Chen H. I., Pinkas-Kramarski R., Jaffe E. A., Gandy S. E., and Geengard P. (1992) Cholinergic agonists and interleukin-1 regulate processing and secretion of the Alzheimer βA4 amyloid precursor. Proc. Natl. Acad. Sci. USA 89, 10,075–10,078.
Cao X. and Sudhof T. C. (2001) A transcriptively active complex of APP with Fe65 and histone acetyltransferase Tip60. Science 293, 115–120.
Cherny R., Atwood C. S., Xilinas M. E., Gray D. N., Jones W. D., McLean C. A., et al. (2001) Treatment with copper-zinc chelator markedly and rapidly inhibits beta-amyloid accumulation in Alzheimer’s disease transgenic mice. Neuron 30, 641–642.
Chishti M. A., Yang D. S., Janus C., Phinney A. L., Horne P., Pearson J., et al. (2001) Early-onset amyloid deposition and cognitive deficits in transgenic mice expressing a double mutant form of amyloid precursor protein 695. J. Biol. Chem. 276, 21,562–21,570.
Crapper McLachlan D. R., Dalton A. J., Kruck T. P. A., Bell M. Y., Smith W. L., Kalow, W., and Andrews, D. F. Intramuscular desferrioxamine in patients with Alzheimer’s disease. Lancet 337, 1304–1308.
Feng Y., Zhang F., Lokey L. K., Chastain J. L., Lakkis L., Eberhart D., and Warren S. T. (1995) Translational suppression by trinucleotide repeat expansion at FMR1. Science 268, 731–734.
Fisher A., Michaelson D. M., Brandeis R., Haring R., Chapman S., and Pittel Z. (2000) M1 muscarinic agonists as potential disease-modifying agents in Alzheimer’s disease. Rationale and perspectives. Ann. NY Acad. Sci. 920, 315–320.
Horowitz B. Z. and Mirkin D. B. (2001) Lead poisoning and chelation in a mother-neonate pair. J. Toxicol. Clin. Toxicol. 39, 727–731.
Janus C., Pearson J., McLaurin J., Mathews P. M., Jiang Y., Schmidt S. D., et al. (2000) A beta peptide immunization reduces behavioural impairment and plaques in a model of Alzheimer’s disease. Nature 408, 979–982.
Khan W.A., Sahaa D., Rahmana A., Salama M., Bogaertsa J., and Bennish M. (2002) Comparison of single-dose azithromycin and 12-dose, 3-day erythromycin for childhood cholera: a randomised, double-blind trial. Lancet 360(9347), 1722–1727.
Lim G. P., Yang F., Chu T., Chen P., Beech W., Teter B., et al. (2000) Ibuprofen suppresses plaque pathology and inflammation in a mouse model for Alzheimer’s disease. J. Neurosci. 20, 5709–5714.
Ly B. T., Williams S. R., and Clark R. F. (2002) Mercuric oxide poisoning treated with whole-bowel irrigation and chelation therapy. Ann. Emerg. Med. 39, 312–315.
Moechars D. G. M., Kuiperi C., Laenen I., and Van Leuven F. (1998) Aggressive behaviour in transgenic mice expressing APP is alleviated by serotonergic drugs. Neuroreport 9, 3561–3564.
Nilsson L., Rogers J., and Potter H. (1998) The essential role of inflammation and induced gene expression in the pathogenic pathway of Alzheimer’s disease. Front. Biosci. 3, 436–446.
Poehlsgaard J. and Douthwaite S. (2002) The macrolide binding site on the bacterial ribosome. Curr. Drug Targets Infect. Disord. 2(1), 67–78.
Rogers J. T., Randall J. D., Leiter L. M., McPhee J., Cahill C. M., Zhan S. S., Potter H., and Nilsson L. N. (1999) Translation of the Alzheimer amyloid precursor protein mRNA is up-regulated by interleukin-1 through 5′-untranslated region sequences. J. Biol. Chem. 274, 6421–6431.
Rogers J. T, Randall J. D., Cahill C. M., Eder P. S., Huang X., Gunshin H., Leiter L., et al. (2002a) An ironresponsive element type II in the 5′ untranslated region of the Alzheimer’s amyloid precursor protein transcript. J. Biol. Chem. 277, 45,518–45,528.
Rogers J. T., Randall J. D., Eder P. S., Huang X., Bush A. I., Tanzi R. E., Venti A., et al. (2002b) Alzheimer’s disease drug discovery targeted to the APP mRNA 5′ untranslated region. J. Mol. Neurosci. 19, 77–82.
Shaw K. T., Utsuki T., Rogers J., Yu Q. S., Sambamurti K., et al. (2001) Phenserine regulates translation of beta-amyloid precursor protein mRNA by a putative interleukin-1 responsive element, a target for drug development. Proc. Natl. Acad. Sci. U.S.A. 98, 7605–7610.
Svitkin Y. V., Pause A., Haghighat A., Pyronnet S., Witherell G., Belsham G. J., and Sonenberg N. (2001) The requirement for eukaryotic initiation factor 4A(elF4A) in translation is in direct proportion to the degree of mRNA 5′ secondary structure. RNA 7, 382–394.
von Koch C. S., Zheng H., Chen H., Trumbauer M., Thinakaran G., van der Ploeg L. H., et al. (1997) Generation of APLP2 KO mice and early postnatal lethality in APLP2/APP double KO mice. Neurobiol. Aging 18, 661–669.
Wasco W., Gurubhagavatula S., Paradis M. D., Romano D. M., Sisodia S. S., Hyman B. T., et al. (1993) Isolation and characterization of APLP2 encoding a homologue of the Alzheimer’s associated amyloid beta protein precursor. Nat. Genet. 5, 95–100.
Weggen S., Eriksen J. L., Das P., Sagi S. A., Wang R., Pietrzik C. U., et al. (2001) A subset of NSAIDs lower amyloidogenic Abeta42 independently of cyclooxygenase activity. Nature 414, 212–216.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Payton, S., Cahill, C.M., Randall, J.D. et al. Drug discovery targeted to the Alzheimer’s APP mRNA 5′-untranslated region. J Mol Neurosci 20, 267–275 (2003). https://doi.org/10.1385/JMN:20:3:267
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:20:3:267