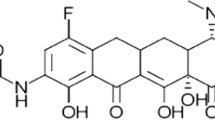
Abstract
Synopsis
The parenteral carbapenem mewpenem is relatively stable to inactivation by human renal dehydropeptidase (DHP-1) and does not require concomitant administration of a DHP-1 inhibitor such as cilastatin. It has a broad spectrum of antibacterial activity in vitro, the majority of Gram-negative, Gram-positive and anaerobic pathogens being highly susceptible to the drug.
Meropenem has shown clinical and bacteriological efficacy in the treatment of a wide range of serious infections in adults and children which is at least comparable with that of currently available treatment options. Its clinical and bacteriological efficacy is similar to that of imipenem/cilastatin, clindamycin plus tobramycin and cefotaxime plus metronidazole in the treatment of intra-abdominal infections; cefotaxime or ceftriaxone in the treatment of meningitis; imipenem/cilastatin, and ceftazidime with or without an aminoglycoside, in lower respiratory tract infections; and imipenem/cilastatin or ceftazidime in the treatment of urinary tract infections. Satisfactory clinical and bacteriological response rates have also been achieved in patients with skin and skin structure infections, obstetric and gynaecological infections or septicaemia, and in immunocompromised patients with febrile episodes. Preliminary findings also indicate efficacy in the treatment of respiratory tract infections in patients with cystic fibrosis.
The tolerability profile of mewpenem is generally similar to that of comparator agents, although it is associated with a lower incidence of adverse gastrointestinal effects (nausea and vomiting) than imipenem/cilastatin. Importantly, the incidence of seizures in patients with meningitis is not increased following administration of meropenem.
Thus, mewpenem is an effective broad spectrum antibacterial drug for the treatment of a wide range of infections including polymicrobial infections in both adults and children, with comparable efficacy to imipenem/cilastatin and various other treatment regimens. Mewpenem is likely to be of greatest value as empiric monotherapy in the treatment of serious infections or those caused by multiply-resistant pathogens. Further clinical experience is necessary, however, to ultimately define its place in therapy.
Antibacterial Activity
Meropenem causes bacterial cell death by binding covalently to penicillin binding proteins involved in cell wall synthesis. For a wide range of aerobic and anaerobic bacteria, the ratio of the minimum bactericidal activity (MBC) to minimum inhibitory concentration (MIC) was 1 or 2. Unlike other β-lactam antibacterial agents, carbapenems induce a postantibiotic effect against Gram-negative bacilli, and that produced by meropenem was often longer than that produced by imipenem in the same bacteria. The drug is generally more active in vitro than imipenem against Enterobacteriaceae, including nosocomial clinical isolates resistant to ceftazidime, cefotaxime, ceftriaxone, piperacillin and gentamicin, with over 90% of isolates generally inhibited by meropenem 0.008 to 0.5 mg/L. In most studies, the MIC of meropenem for 90% (MIC90) of Pseudomonas aeruginosa isolates was ≤4 mg/L. All tested strains of Haemophilus influenzae and Neisseria gonorrhoeae were susceptible to meropenem.
The drug is also active against Staphylococcus aureus, as well as S. epidermidis, S. saprophyticus and other coagulase-negative staphylococci, but is generally less active than imipenem against these Gram-positive organisms. Combinations of meropenem with several other antibacterial agents have shown a synergistic antibacterial activity against methicillin-resistant S. aureus. Streptococcus pyogenes, S. agalactiae and S. pneumoniae, including penicillin-resistant strains, are inhibited by low concentrations of meropenem, and most strains of Enterococcus faecalis are susceptible or moderately susceptible. E. faecium strains are resistant to meropenem.
Meropenem 0.06 to 4 mg/L inhibits the growth of virtually all tested strains of Bacteroides fragilis, B. fragilis group, and Fusobacterium spp., and is also active against non-B. fragilis group Bacteroides spp., Prevotella and Porphyromonas species. Meropenem is more active than imipenem against clinical isolates of Clostridium perfringens, C. difficile, Veillonella spp. and Peptococcus species. Carbapenems appear to be highly resistant to hydrolysis by all of the TEM and SHV β-lactamases (Richmond Sykes Class III), and meropenem appears to be stable to all β-lactamases belonging to Richmond Sykes Class I including those with an extended spectrum against third-generation cephalosporins. Altered penicillin binding proteins account for resistance among E. faecium and methicillin-resistant S. aureus. Impermeability resistance is a clinical problem with Gram-negative bacilli, but these organisms are rarely resistant to meropenem. A similar type of resistance mechanism occurs in P. aeruginosa, and meropenem-resistant strains lack the D2 outer membrane protein. There is considerable cross-resistance between monobactams, second- and third-generation cephalosporins and ureidopenicillins, but very little between these drugs and meropenem.
Pharmacokinetic Properties
Meropenem exhibits approximately linear kinetics, with plasma concentrations increasing with dose (reaching higher levels after a bolus injection than after a 30 minute infusion in the first hour). Other pharmacokinetic parameters are unchanged after bolus injection compared with infusion. The mean peak plasma concentration after a single 30 minute infusion of meropenem 1g ranges between 53.1 and 61.6 mg/L.
Meropenem is widely distributed into body tissues and fluids, including cerebrospinal fluid. The elimination half-life is approximately 1 hour after intravenous doses of 0.5 to 1g in healthy volunteers, and is increased in neonates, infants and patients with impaired renal function. Renal clearance occurs mainly via glomerular filtration but tubular secretion also plays a part. 54 to 79% of the original dose of meropenem is recovered unchanged in the urine, indicating stability of the molecule to the human renal dehydropeptidase enzyme (DHP-1); about 2% is recovered in the faeces, and the remainder appears to be excreted in the urine as the inactive metabolite.
The pharmacokinetic profile of meropenem is similar to that of imipenem administered as imipenem/cilastatin.
Clinical Efficacy
Meropenem has shown clinical efficacy at least equal to that of comparator agents in the treatment of a wide range of infections in adults and children in randomised, generally nonblinded, clinical trials.
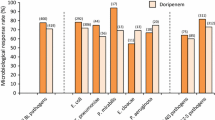
Intravenous meropenem 0.5 or 1g every 8 hours showed clinical and bacteriological efficacy similar to that of imipenem/cilastatin, clindamycin plus tobramycin and cefotaxime plus metronidazole in patients with intra-abdominal infections. Clinical responses (cure or improvement) were achieved in 91 to 100% of patients following treatment with meropenem, and the bacteriological response rate ranged between 84 and 95%.
Preliminary findings have shown the efficacy of intravenous meropenem 1g every 8 hours to be similar to that of ceftazidime 2g every 8 hours as initial empiric monotherapy for the treatment of febrile episodes in patients with neutropenia. 44% of febrile episodes responded to treatment with meropenem versus 41% with ceftazidime.
Intravenous meropenem 120 mg/kg/day (up to 6 g/day in adults and children weighing ≥ 50kg) has shown clinical and bacteriological efficacy similar to that of the cephalosporins cefotaxime and ceftriaxone in adults and children with bacterial meningitis caused predominantly by S. pneumoniae, N. meningitidis or H. influenzae. Clinical responses were achieved in 98% of meropenem recipients and the bacteriological eradication rate was 100% in one study. Comparative studies with imipenem/cilastatin are not available as this drug combination is not licensed for bacterial meningitis due to an associated increased incidence of seizures. Case studies have reported efficacy of meropenem in the treatment of cephalosporin-resistant pseudomonal infections; however, data on the drug’s clinical value against infections caused by other pathogens resistant to current treatment regimens are not available.
The clinical efficacy of intravenous or intramuscular meropenem 0.5 to 3 g/day was similar to that of imipenem/cilastatin and ceftazidime with or without an aminoglycoside in patients with lower respiratory tract infections. The clinical response rate in patients with nosocomially-acquired infections was 98% in one study, and ranged between 93 and 100% in patients with community-acquired infections. Bacteriological eradication rates were 88% and 91 to 100%, respectively. Clinical and bacteriological responses were achieved in 81 and.71% of patients with severe nosocomial infection and in 93% and 91 to 100% of patients with severe community-acquired infections. The clinical value of meropenem against infections in patients with cystic fibrosis remains to be established. However, studies in small numbers of patients have demonstrated efficacy in reducing pulmonary sepsis similar to that shown by ceftazidime.
Intravenous or intramuscular meropenem 0.5 to 1.5 g/day was also effective in the treatment of uncomplicated and complicated urinary tract infections, producing clinical responses in 79 to 97% of patients and bacteriological eradication in 56 to 92%. Randomised trials indicated an efficacy generally similar to that of imipenem/cilastatin or ceftazidime, although one study reported a significantly greater clinical response rate in meropenem compared with imipenem/cilastatin recipients (97 versus 90%; p < 0.05).
Clinical and bacteriological response rates in patients with skin and skin structure infections were 98 and 94%, respectively, (similar to those achieved with imipenem/cilastatin), and were 88 to 100% and 88 to 90% in patients with obstetric and gynaecological infections (similar to those achieved with combination therapy of intravenous clindamycin and gentamicin).
Preliminary studies have shown meropenem to be as effective as ceftazidime in the treatment of septicaemia (clinical response rate 92 versus 94%). The mean response rate to meropenem in clinical trials of the drug in children with a range of infections was 98% and bacteria were eradicated from between 89 and 97% of patients.
Tolerability
The tolerability profile of intravenous (as an infusion or bolus injection) or intramuscular meropenem 1.5 to 6 g/day is similar to that of other β-lactam antibacterials with which it has been compared. It is similar in children, adults and the elderly and is not altered in patients with renal impairment. Nausea and vomiting (in ≤3.6% of patients), diarrhoea (≤4.3%) and transient changes in hepatic biochemistry (increased ALT and AST in 7 and 5.6%, respectively) occur most often. However, nausea and vomiting and inflammation at the injection site appear to occur less frequently in patients receiving meropenem than in those receiving imipenem/cilastatin.
Seizures appear to occur at a similar rate in meropenem recipients to that seen in patients receiving other antibacterial drugs (0.38% in one large analysis). Unlike imipenem/cilastatin, however, meropenem has not induced seizures in patients with meningitis.
Dosage and Administration
The recommended intravenous dosage of meropenem in adults with normal renal function is 0.5 to 1g every 8 hours, and 1g every 8 hours in patients with neutropenia. Children should receive 10 to 20 mg/kg every 8 hours. The dosage is increased in patients with meningitis (adults: 2g every 8 hours; children: 40 mg/kg every 8 hours), and decreased in patients with renal impairment.
Meropenem may be administered by intravenous bolus injection over approximately 5 minutes or by intravenous infusion over 15 to 30 minutes.
Similar content being viewed by others
References
Hikida M, Kawashima K, Yoshida M, et al. Inactivation of new carbapenem antibiotics by dehydropeptidase-I from porcine and human renal cortex. J Antimicrob Chemother 1992 Aug; 30: 129–34
Fukasawa M, Sumita Y, Harabe ET, et al. Stability of meropenem and effect of 1 beta-methyl substitution on its stability in the presence of renal dehydropeptidase I. Antimicrob Agents Chemother 1992 Jul; 36: 1577–9
Sumita Y, Fukasawa M, Okuda T. Comparison of two carbapenems, SM-7338 and imipenem: affinities for penicillin-binding proteins and morphological changes. J Antibiot Tokyo 1990 Mar; 43: 314–20
Kitzis MD, Acar JF, Gutmann L. Antibacterial activity of meropenem against Gram-negative bacteria with a permeability defect against staphylococci. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 125–32
Sumita Y, Fukasawa M, Okuda T. Affinities of SM-7338 for penicillin-binding proteins and its release from these proteins in Staphylococcus aureus. Antimicrob Agents Chemother 1990 Mar; 34: 484–6
Pfaller MA, Jones RN, Washington JA, et al. Interpretive criteria and quality control guidelines for lomefloxacin and meropenem in susceptibility tests of Haemophilus influenzae using Haemophilus test medium. Diagn Microbiol Infect Dis 1992 Feb; 15: 145–50
National Committee for Clinical Laboratory Standards. Performance standards for antimicrobial susceptibility testing; Fourth Information Supplement. NCCLS Document M100-S4 (ISBN 1-56238-172-5). Pennsylvania: NCCLS, 771 E. Lancaster Avenue, Villonova., 1992
Nishino T, Tada E, Otsuki M, et al. In vitro and in vivo antibacterial activities of meropenem, a new carbapenem antibiotic [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 50–66
Harabe E, Kawai Y, Kanazawa K. In vitro and in vivo antibacterial activities of meropenem, a new carbapenem antibiotic. Drugs Exp Clin Res 1992; 18(2): 37–46
Hoban DJ, Jones RN, Yamane N, et al. In vitro activity of three carbapenem antibiotics. Comparative studies with biapenem (L-627), imipenem, and meropenem against aerobic pathogens isolated worldwide. Diagn Microbiol Infect Dis 1993 Nov–Dec; 17: 299–305
Inderlied CB, Lancero MG, Young LS. Bacteriostatic and bactericidal in-vitro activity of meropenem against clinical isolates, including Mycobacterium avium complex. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 85–99
Jones RN, Barry AL, Thornsberry C. In vitro studies of meropenem. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 9–29
King A, Boothman C, Phillips I. Comparative in-vitro activity of meropenem on clinical isolates from the United Kingdom. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 31–45
Sader HS, Jones RN. Antimicrobial activity of the new carbapenem biapenem compared to imipenem, meropenem and other broad-spectrum beta-lactam drugs. Eur J Clin Microbiol Infect Dis 1993 May; 12: 384–91
Sentochnik DE, Eliopoulos GM, Ferraro MJ, et al. Comparative in vitro activity of SM7338, a new carbapenem antimicrobial agent. Antimicrob Agents Chemother 1989 Aug; 33: 1232–6
Wiedemann B, Zuhlsdorf M. Antibacterial properties of meropenem towards clinical isolates, beta-lactamase producers and laboratory mutants. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 197–205
Bauernfeind A, Jungwirth R, Schweighart S. In-vitro activity of meropenem, imipenem, the penem HRE 664 and ceftazidime against clinical isolates from West Germany. J Anti-microb Chemother 1989 Sep; 24 Suppl. A: 73–84
Fukasawa M, Sumita Y, Tada E, et al. In vitro antibacterial activity of meropenem [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 74–89
Jones RN, Aldridge KE, Allen SD, et al. Multicenter in vitro evaluation of SM-7338, a new carbapenem. Antimicrob Agents Chemother 1989 Apr; 33: 562–5
Nakamura H, Fukui T, Katsu M, et al. Bacteriological and clinical studies on meropenem [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 312–25
Schito GC, Chezzi C, Ravizzola G, et al. In-vitro activity of meropenem against clinical isolates in a multicentre study in Italy. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 57–72
Sumita Y, Inoue M, Mitsuhashi S. In vitro antibacterial activity and beta-lactamase stability of the new carbapenem SM-7338. Eur J Clin Microbiol 1989 Oct; 8: 908–16
Edwards JR, Turner PJ, Wannop C, et al. In vitro antibacterial activity of SM-7338, a carbapenem antibiotic with stability to dehydropeptidase I. Antimicrob Agents Chemother 1989 Feb; 33: 215–22
Neu HC, Novelli A, Chin NX. In vitro activity and beta-lactamase stability of a new carbapenem, SM-7338. Antimicrob Agents Chemother 1989 Jul; 33: 1009–18
Visser MR, Hoepelman IM, Beumer H, et al. Comparative in vitro antibacterial activity of the new carbapenem meropenem (SM-7338). Eur J Clin Microbiol Infect Dis 1989 Dec; 8: 1061–4
Catchpole CR, Wise R, Thornber D. In vitro activity of L-627, a new carbapenem. Antimicrob Agents Chemother 1992 Sep; 36: 1928–34
Wise R, Andrews JM, Ashby JP, et al. In vitro activity of a catechol-substituted cephalosporin, GR69153. Antimicrob Agents Chemother 1991 Feb; 35: 329–34
Yeo S-F, Livermore DM. Comparative in-vitro activity of biapenem and other carbapenems against Haemophilus influenzae isolates with known resistance mechanisms to ampicillin. J Antimicrob Chemother 1994 Apr; 33: 861–5
Jorgensen JH, Maher LA, Howell AW. Activity of a new carbapenem antibiotic, meropenem, against Haemophilus influenzae strains with beta-lactamase- and non-enzyme-mediated resistance to ampicillin. Antimicrob Agents Chemother 1991 Mar; 35: 600–2
Clarke AM, Zemcov SJ. In-vitro activity of meropenem against clinical isolates obtained in Canada. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 47–55
Hayashi Y, Yamamoto K, Yamada Y, et al. Laboratory and clinical studies on meropenem [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 836–9
Ravizzola G, Pinsi G, Gonzales R, et al. Antibacterial activity of the new carbapenem meropenem (SM-7338) against clinical isolates. Eur J Clin Microbiol Infect Dis 1989 Dec; 8: 1053–61
Sarubbi F, Franzus B, Verghese A. Comparative activity of meropenem (SM-7338) against major respiratory pathogens and amikacin-resistant nosocomial isolates. Eur J Clin Microbiol Infect Dis 1992 Jan; 11: 65–8
Voutsinas D, Mavroudis T, Avlamis A, et al. In-vitro activity of meropenem, a new carbapenem, against multiresistant Pseudomonas aeruginosa compared with that of other antipseudomonal antimicrobials. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 143–7
Lewin C, Doherty C, Go van J. In vitro activities of meropenem, PD 127391, PD 131628, ceftazidime, chloramphenicol, cotrimoxazole, and ciprofloxacin against Pseudomonas cepacia. Antimicrob Agents Chemother 1993 Jan; 37: 123–5
Chanal C, Sirot M, Chanal M, et al. Comparative in-vitro activity of meropenem against clinical isolates including Enterobacteriaceae with expanded-spectrum beta-lactamases. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 133–41
Powell M, Seetulsingh P, Williams JD. In-vitro susceptibility of Haemophilus influenzae to meropenem compared with imipenem, five other beta-lactarns, chloramphenicol and ciprofloxacin. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 175–81
Jorgensen JH, Maher LA, Howell AW. Activity of meropenem against antibiotic-resistant or infrequently encountered Gram-negative bacilli. Antimicrob Agents Chemother 1991 Nov; 35: 2410–4
Kayser FH, Morenzoni G, Strassle A, et al. Activity of meropenem, against gram-positive bacteria. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 101–12
Linares J, Alonso T, Perez JL, et al. Decreased susceptibility of penicillin-resistant pneumococci to twenty-four beta-lactam antibiotics. J Antimicrob Chemother 1992 Sep; 30: 279–88
Edwards JR. Meropenem: a microbiological overview. J Antimicrob Chemother. In press
Nordmann P, Ronco E. In-vitro antimicrobial susceptibility of Rhodococcus equi. J Antimicrob Chemother 1992 Apr; 29: 383–93
Yazawa K, Mikami Y, Ohashi S, et al. In-vitro activity of new carbapenem antibiotics: comparative studies with meropenem, L-627 and imipenem against pathogenic Nocardia spp. J Antimicrob Chemother 1992 Feb; 29: 169–72
Watt B, Naden M. The growth-inhibitory properties of meropenem against anaerobes of clinical importance. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 119–24
Sheikh W, Pitkin DH, Nadler H. Antibacterial activity of meropenem and selected comparative agents against anaerobic bacteria at seven North American centers. Clin Infect Dis 1993 Jun; 16 Suppl. 4: 361–6
Garcia-Rodriguez JA, Garcia SJE, Trujillano I, et al. Meropenem: in-vitro activity and kinetics against organisms of the Bacteroides fragilis group. J Antimicrob Chemother 1991 May; 27: 599–606
Aldridge KE, Morice N, Schiro DD. In vitro activity of biapenem (L-627), a new carbapenem, against anaerobes. Antimicrob Agents Chemother 1994 Apr; 38: 889–93
Nord CE, Lindmark A, Persson I. Susceptibility of anaerobic bacteria to meropenem. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 113–7
Murray PR, Niles AC. In vitro activity of meropenem (SM-7338), imipenem, and five other antibiotics against anaerobic clinical isolates. Diagn Microbiol Infect Dis 1990 Jan–Feb; 13: 57–61
Appelbaum PC, Spangler SK, Jacobs MR. Susceptibilities of 394 Bacteroides fragilis, non-B. fragilis group Bacteroides species, and Fusobacterium species to newer antimicrobial agents. Antimicrob Agents Chemother 1991 Jun; 35: 1214–8
García-Rodríguez JA, García-Sánchez JE, Trujillano-Martín I, et al. L-627, a novel carbapenem: in-vitro activity against anaerobes. J Antimicrob Chemother 1994 Jan; 33: 183–6
Nord CE, Lindmark A, Persson I. In vitro activity of L-627 against anaerobic bacteria. Eur J Clin Microbiol Infect Dis 1992 Aug; 11: 757–60
Goldstein EJC, Citron DM, Cherubin CE, et al. Comparative susceptibility of the Bacteroides fragilis group species and other anaerobic bacteria to meropenem, imipenem, piperacillin, cefoxitin, ampicillin/sulbactam, clindamycin and metronidazole. J Antimicrob Chemother 1993 Mar; 31: 363–72
Ferrara A, Grassi G, Grassi FA, et al. Bactericidal activity of meropenem and interactions with other antibiotics. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 239–50
Eng RH, Padberg FT, Smith SM, et al. Bactericidal effects of antibiotics on slowly growing and nongrowing bacteria. Antimicrob Agents Chemother 1991 Sep; 35: 1824–8
Wu PJ, Livermore DM. Response of chemostat cultures of Pseudomonas aeruginosa to carbapenems and other beta-lactams. J Antimicrob Chemother 1990 Jun; 25: 891–902
Yourassowsky E, Van der Linden MP, Crokaert F. Antibacterial effects of meropenem and imipenem against Haemophilus influenzae. Chemotherapy (Basel) 1991 Jan–Feb; 37: 38–42
Yourassowsky E, Van der Linden MP, Lismont MJ, et al. Bactericidal activity of meropenem against Pseudomonas aeruginosa. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 169–74
Yourassowsky E, van der LMP, Crokaert F. Antibacterial effect of meropenem and imipenem on Proteus mirabilis. J Antimicrob Chemother 1990 Aug; 26: 185–92
Chen HY, Livermore DM. Comparative in-vitro activity of biapenem against enterobacteria with beta-lactamase-mediated antibiotic resistance. J Antimicrob Chemother 1994 Mar; 33: 453–64
Kitzis M-D, Liassine N, Ferré B, et al. In vitro activities of 15 oral β-lactams against Klebsiella pneumoniae harboring new extended-spectrum β-lactamases. Antimicrob Agents Chemother 1990; 34(9): 1783–6
Labia R, Morand A, Tiwari K, et al. Interactions of meropenem with beta-lactamases, including enzymes with extended-spectrum activity against third-generation cephalosporins. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 219–23
Sirot D. Extended-spectrum plasmid-mediated β-lactamases. J Antimicrob Chemother. In press
Cullmann W, Dick W. Heterogeneity of beta-lactamase production in Pseudomonas maltophilia, a nosocomial pathogen. Chemotherapy 1990; 36: 117–26
Sanders CC, Sanders WE Jr, Thomson KS, et al. Meropenem: activity against resistant gram-negative bacteria and interactions with beta-lactamases. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 187–96
Akova M, Bonfiglio G, Livermore DM. Susceptibility to beta-lactam antibiotics of mutant strains of Xanthomonas maltophilia with high- and low-level constitutive expression of L1 and L2 beta-lactamases. J Med Microbiol 1991 Oct; 35: 208–13
Edwards R, Brown J, Greenwood D. Comparative stability of meropenem against beta-lactamases from Bacteroides spp. Chemotherapy (Basel) 1993 Nov–Dec; 39: 390–3
Livermore DM, Yang Y. Comparative activity of meropenem against Pseudomonas aeruginosa strains with well-characterized resistance mechanisms. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 149–59
Bonfiglio G, Livermore DM. Effect of media composition on the susceptibility of Xanthomonas maltophilia to beta-lactam antibiotics. J Antimicrob Chemother 1991 Dec; 28: 837–42
Jones RN, Gardiner RV. Stability of SM-7338, a new carbapenem in mediums recommended for the susceptibility testing of anaerobic bacteria and gonococci. Diagn Microbiol Infect Dis 1989 May–Jun; 12: 271–3
Wise R, Ashby JP, Andrews JM. The antibacterial activity of meropenem in combination with gentamicin or vancomycin. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 233–8
Ito A, Kaminaga Y, Murano T, et al. In vitro studies of combinations of piperacillin and various antimicrobial agents on MRSA [in Japanese], Chemotherapy (Tokyo) 1993 Nov; 41: 1145–53
MacKenzie FM, Gould IM, Chapman DG, et al. Comparison of methodologies used in assessing the postantibiotic effect. J Antimicrob Chemother 1994 Aug; 34: 223–30
Nadler HL, Sheikh W. A comparison of the in vitro post-antibiotic effect of meropenem and imipenem versus selected enterobacteriaceae and other pathogens. Diagn Microbiol Infect Dis 1993 Jul; 17: 71–3
Tanio T, Fukasawa M. In vitro and in vivo postantibiotic effect of meropenem [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 103–7
Odenholt-Tornqvist I. Studies on the postantibiotic effect and the postantibiotic sub-MIC effect of meropenem. J Antimicrob Chemother 1993 Jun; 31: 881–92
Nadler HL, Pitkin DH, Sheikh W The postantibiotic effect of meropenem and imipenem against selected bacteria. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 225–31
Martin CMM, Fuentes MF, Izquierdo IJ, et al. In-vivo and in-vitro study of the postantibiotic effect of meropenem. J Antimicrob Chemother 1993 Dec; 32: 917–8
MacKenzie FM, Gould IM, Chapman DG, et al. Postantibiotic effect of meropenem on members of the family Enterobacteriaceae determined by five methods. Antimicrob Agents Chemother 1994 Nov; 38: 2583–9
Majcherczyk PA, Livermore DM. Penicillin-binding protein (PBP) 2 and the post-antibiotic effect of carbapenems [letter]. J Antimicrob Chemother 1990 Oct; 26: 593–4
Livermore DM. Interplay of impermeability and chromosomal beta-lactamase activity in imipenem-resistant Pseudomonas aeruginosa. Antimicrob Agents Chemother 1992 Sep; 36: 2046–8
Margaret BS, Drusano GL, Standiford HC. Emergence of resistance to carbapenem antibiotics in Pseudomonas aeruginosa. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 161–7
Zhou XY, Kitzis M-D, Gutmann L. Role of cephalosporinase in carbapenem resistance of clinical isolates of Pseudomonas aeruginosa. Antimicrob Agents Chemother 1993 Jun; 37: 1387–9
Livermore DM. Mechanisms of resistance to β-lactam antibiotics. Scand J Infect Dis 1991; 78 Suppl.: 7–16
Cornaglia G, Guan L, Fontana R, et al. Diffusion of meropenem and imipenem through the outer membrane of Escherichia coli K-12 and correlation with their antibacterial activities. Antimicrob Agents Chemother 1992 Sep; 36: 1902–8
Gotoh N, Tanaka S, Nishino T. Permeability of the outer membrane of Moraxella catarrhalis for β-lactam antibiotics. J Antimicrob Chemother 1992; 29: 279–85
Chen HY, Yuan M, Elmagboul IBI, et al. Multi-centre UK survey of drug resistance in P. aeruginosa. Zeneca Pharmaceuticals (Data on file)
Raimondi A, Traverso A, Nikaido H. Imipenem- and meropenem-resistant mutants of Enterobacter cloacae and Proteus rettgeri lack porins. Antimicrob Agents Chemother 1991 Jun; 35: 1174–80
Satake S, Yoneyama H, Nakae T. Role of 0mpD2 and chromosomal beta-lactamase in carbapenem resistance in clinical isolates of Pseudomonas aeruginosa. J Antimicrob Chemother 1991 Aug; 28: 199–207
Vurma-Rapp U, Kayser FH, Hadorn K, et al. Mechanism of imipenem resistance acquired by three Pseudomonas aeruginosa strains during imipenem therapy. Eur J Clin Microbiol Infect Dis 1990 Aug; 9: 580–7
Trias J, Nikaido H. Outer membrane protein D2 catalyzes facilitated diffusion of carbapenems and penems through the outer membrane of Pseudomonas aeruginosa. Antimicrob Agents Chemother 1990 Jan; 34: 52–7
Lee EH, Nicolas MH, Kitzis MD, et al. Association of two resistance mechanisms in a clinical isolate of Enterobacter cloacae with high-level resistance to imipenem. Antimicrob Agents Chemother 1991 Jun; 35: 1093–8
Nikaido H. Prevention of drug access to bacterial targets: permeability barriers and active efflux. Science 1994; 264: 382
Li X-Z, Livermore DM, Nikaido H. Role of efflux pump (s) in intrinsic resistance of Pseudomonas aeruginosa: resistance to tetracycline, chloramphenicol, and norfloxacin. Antimicrob Agents Chemother 1994; 38: 1732–41
Krcmery V, Trupl J. Nosocomial outbreak of meropenem resistant Pseudomonas aeruginosa infections in a cancer centre [letter]. J Hosp Infect 1994 Jan; 26: 69–71
van den Broek PJ, Buys LFM, van den Barselaar MT, et al. Interaction between human monocytes and penicillin G in relation to the antibacterial effect on Staphylococcus aureus. J Infect Dis 1985; 152: 521–8
van den Broek PJ, Buys LFM, Mattie H, et al. Effect of penicillin G on Staphylococcus aureus phagocytosed by human monocytes. J Infect Dis 1986; 153: 586–92
Cuffini AM, Tullio V, Allocco A, et al. The entry of meropenem into human macrophages and its immunomodulating activity. J Antimicrob Chemother 1993 Nov; 32: 695–703
Easmon CS. Interaction of meropenem with humoral and phagocytic defences. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 259–64
Traub WH, Leonhard B, Bauer D. Intracellular bactericidal activities of cefpirome and meropenem as compared with ciprofloxacin against Serratia marcescens. Med Microbiol Lett 1994 Jun 15; 3: 173–80
Edwards J, Williams S, Nairn K. Therapeutic activity of meropenem in experimental infections. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 279–85
Nairn K, Shepherd GL, Edwards JR. Efficacy of meropenem in experimental meningitis. J Antimicrob Chemother. In press
Bastain W, Bax RP, Featherstone A, et al. The pharmacokinetics in volunteers of SM 7338 anew, broad spectrum, DHP-stable, carbapenem [abstract]. In: 28th Interscience Conference on Antimicrobial Agents and Chemotherapy 1988: 215
Bax RP, Bastain W, Featherstone A, et al. The pharmacokinetics of meropenem in volunteers. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 311–20
Burman LA, Nilsson-Ehle I, Hutchison M, et al. Pharmacokinetics of meropenem and its metabolite ICI 213,689 in healthy subjects with known renal metabolism of imipenem. J Antimicrob Chemother 1991 Feb; 27: 219–24
Drusano GL. Pharmacokinetics of meropenem. Official Satellite Symposium at the 9th Mediterranean Congress of Chemotherapy; 1994 June 14; Milan 1994
Ljungberg B, Nilsson-Ehle I. Pharmacokinetics of meropenem and its metabolite in young and elderly healthy men. Antimicrob Agents Chemother 1992 Jul; 36: 1437–40
Nakashima M, Kanamaru M, Uematsu T. The pharmacokinetics and safety of SM-7338, a new carbapenem, evaluated in healthy volunteers [abstract]. In: 29th Interscience Conference on Antimicrobial Agents and Chemotherapy 1989: 140
Nilsson-Ehle I, Hutchison M, Haworth SJ, et al. Pharmacokinetics of meropenem compared to imipenem-cilastatin in young healthy males. Eur J Clin Microbiol Infect Dis 1991 Feb; 10: 85–8
Wise R, Logan M, Cooper M, et al. Meropenem pharmacokinetics and penetration into an inflammatory exudate. Antimicrob Agents Chemother 1990 Aug; 34: 1515–7
Leroy A, Fillastre JP, Borsa-Lebas F, et al. Pharmacokinetics of meropenem (ICI 194,660) and its metabolite (ICI 213,689) in healthy subjects and in patients with renal impairment. Antimicrob Agents Chemother 1992 Dec; 36: 2794–8
Kelly HC, Hutchison M, Haworth SJ. A comparison of the pharmacokinetics of meropenem after intravenous administration by injection over 5 min and infusion over 30 min. J Antimicrob Chemother. In press
Drusano GL, Hutchison M. The pharmacokinetics of meropenem. Scand J Infect Dis 1995; 96 Suppl.: 11–6
Granai F, Smart HL, Triger DR. A study of penetration of meropenem into bile using endoscopic retrograde cholangio-graphy. J Antimicrob Chemother 1992 Jun; 29: 711–8
Mouton JW, Michel MF. Pharmacokinetics of meropenem in serum and suction blister fluid during continuous and intermittent infusion. J Antimicrob Chemother 1991 Dec; 28: 911–8
Sano T, Sakurai M, Dohi S, et al. Investigation of meropenem levels in the human bone marrow blood, bone, joint fluid and joint tissues [in Japanese]. Jpn J Antibiot 1993 Feb; 46: 159–63
Thys J-P. Penetration of meropenem into lung, bronchial mucosa, pleural tissues, and bronchial secretions: preliminary data [abstract]. 8th Mediterranean Congress on Chemotherapy; May 24-29, 1992
Bergogne-Bérézin E, Muller-Serieys C, Aubier M, et al. Concentration of meropenem in serum and in bronchial secretions in’ patients undergoing fibreoptic bronchoscopy. Eur J Clin Pharmacol 1994; 46(1): 87–8
Newsom SWB, Palsingh J, Wells FC, et al. Penetration of meropenem into heart valve tissue. J Antimicrob Chemother. In press
Dagan R, Velghe L, Rodda JL. Penetration of meropenem into the cerebrospinal fluid of patients with inflamed meninges. J Antimicrob Chemother 1994 Jul; 34: 175–9
Yagami Y, Hanada S, Ikuta K, et al. Basic and clinical studies on meropenem in obstetrics and gynecology [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 676–81
Matsuda S, Hirayama H, Oh K, et al. Pharmacokinetic and clinical studies of meropenem in the field of obstetrics and gynecology [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 652–6
Hori H, Uemura T, Ishikawa H, et al. Pharmacokinetic and clinical studies of meropenem in the field of obstetrics and gynecology [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 669–75
Wise R, Andrews JM, Ashby JP, et al. The pharmacokinetic, inflammatory fluid and peritoneal fluid penetration of meropenem [abstract]. In: 30th Interscience Conference on Antimicrobial Agents and Chemotherapy 1990: 282
Wise R. Penetration of meropenem into intraperitoneal fluid [abstract]. 8th Mediterranean Congress on Chemotherapy; 1992 May 24–29 1992
Miyazaki Y, Higashiyama Y, Matsuda H, et al. Laboratory and clinical studies on meropenem [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 412–20
Ooishi M, Sakaue F, Tazawa H, et al. Basic and clinical studies on meropenem in ophthalmology [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 689–700
Harada Y, Yajin K, Tagashira N, et al. Concentration of meropenem in tissues and clinical efficacy in otorhinolaryngology [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 719–24
Harrison MP, Haworth SJ, Moss SR, et al. The disposition and metabolic fate of 14C-meropenem in man. Xenobiotica 1993 Nov; 23: 1311–23
Fujii R, Yoshioka H, Fujita K, et al. Pharmacokinetic and clinical studies with meropenem in the pediatric field [in Japanese]. Jpn J Antibiot 1992 Jun; 45: 697–717
Martinkova J, de Groot R, Chladek J, et al. Meropenem (MEM) pharmacokinetics (Pk) in pre-term and full-term neonates. Zeneca Pharmaceuticals. Abstract No. A71 (Data on file)
Parker EM, Hutchinson M, Blumer JL. The pharmacokinetics of meropenem in infants and children: a population analysis. J Antimicrob Chemother. In press
Chimata M, Nagase M, Suzuki Y, et al. Pharmacokinetics of meropenem in patients with various degrees of renal function, including patients with end-stage renal disease. Antimicrob Agents Chemother 1993 Feb; 37: 229–33
Leroy A, Fillastre JP, Etienne I, et al. Pharmacokinetics of meropenem in subjects with renal insufficiency. Eur J Clin Pharmacol 1992; 42: 535–8
Christensson BA, Nilsson-Ehle I, Hutchison M, et al. Pharmacokinetics of meropenem in subjects with various degrees of renal impairment. Antimicrob Agents Chemother 1992 Jul; 36: 1532–7
Christensson BA, Ljungberg B, Eriksson L, et al., editors. Pharmacokinetics of meropenem in cystic fibrosis patients [abstract no. A73]. Proceedings and Abstracts of the 34th Interscience Conference on Antimicrobial Agents and Chemotherapy; Orlando 1994
Geroulanos SJ. Antibiotics and the abdomen. Curr Opin Infect Dis 1994 Oct; 7 Suppl. 1: 17–22
Brismar B, Malmborg AS, Tuncrall G, et al. Meropenem versus imipenem/cilastatin in the treatment of intra-abdominal infections. Report from a Swedish Study Group. J Antimicrob Chemother 1995; 35: 139–48
Geroulanos SJ, Meropenem Study Group. Meropenem versus imipenem/cilastatin in intra-abdominal infections requiring surgery. J Antimicrob Chemother. In press
Kanellakopoulou K, Giamarellou H, Papadothomakos P, et al. Meropenem versus imipenem/cilastatin in the treatment of intraabdominal infections requiring surgery. Eur J Clin Microbiol Infect Dis 1993 Jun; 12: 449–53
Condon RE, Walker AP, Sirinek KR, et al. Meropenem compared to tobramycin plus clindamycin in intraabdominal infections: results of a prospective, randomized, double-blind clinical trial. Clin Infect Dis. In press
Heseltine PNR, Berne TV, Yellin AE, et al. Carbapenems reduce hospital stay for perforated & gangrenous appendicitis compared with clindamycin/aminoglycoside [abstract]. Clin Infect Dis 1994 Sep; 19: 595
Huizinga WKJ, Warren BL, Baker LW, et al. Antibiotic monotherapy with meropenem in the surgical management of intra-abdominal infections. J Antimicrob Chemother. In press
Meropenem — efficacy in the management of critically ill patients. Zeneca Pharmaceuticals (Data on file)
Del Favero A. Empiric monotherapy in neutropenia — a realistic goal? [abstract]. Scand J Infect Dis In press
Boogaerts MA. Anti-infective strategies in neutropenia. J Anti-microb Chemother. In press
The Meropenem Study Group of Leuven L and N. Equivalent efficacies of meropenem and ceftazidime as empirical mono-therapy of febrile neutropenic patients. J Antimicrob Chemother. In press
Masaoka T, Sugiyama H, Nagai K, et al. Therapeutic effects of meropenem against severe infections in patients with haematopoietic disorders [abstract]. 8th Mediterranean Congress on Chemotherapy; 1992 May 24–29 1992
McGee ZA, Baringer JR. Acute Meningitis. In: Mandell GL, Douglas RG, Bennett JE, editors. Principles and Practice of Infectious Diseases. Third ed. New York, Edinburgh, London, Melbourne: Churchill Livingstone, 1990: 741–55
Buckley MM, Brogden RN, Barradell LB, et al. Imipenem/cilastatin: a reappraisal of its antibacterial activity, pharmacokinetic properties and therapeutic efficacy. Drugs 1992; 44(3): 408–44
Scheid WM. Is there a place for carbapenems in the therapy of meningitis? Curr Opin Infect Dis 1994 Oct; 7 Suppl. 1: 33–7
Klugman KP, Dagan R, Meningitis SG. Efficacy and safety of meropenem in the treatment of meningitis. Antimicrob Agents Chemother 1995; 39: 1140–6
Lopez E. Meropenem versus cefotaxime or ceftriaxone for bacterial meningitis [abstract]. In: 33rd Interscience Conference on Antimicrobial Agents and Chemotherapy 1993: 236
Pelser HH, Meropenem SG. Meropenem or cephalosporins for bacterial meningitis [abstract]. In: 18th International Congress of Chemotherapy 1993: 265
Schmutzhard E, Williams KJ, Vukmirovits G, et al. A randomised comparison of meropenem with cefotaxime or ceftriaxone for the treatment of bacterial meningitis in adults. J Antimicrob Chemother. In press
Donnelly JP, Horrevorts AM, Sauerwein RW, et al. High-dose meropenem in meningitis due to Pseudomonas aeruginosa. Lancet 1992 May 2; 339: 1117
Chmelik V, Gutvirth J. Meropenem treatment of post-traumatic meningitis due to Pseudomonas aeruginosa. J Antimicrob Chemother 1993 Dec; 32: 922–3
Hamacher J, Vogel F, Lichey J, et al. Treatment of acute bacterial exacerbations of chronic obstructive pulmonary disease in hospitalised patients — a comparison of meropenem and imipenem/cilastatin. J Antimicrob Chemother. In press
Hara K, Sakamoto A, Komori K, et al. A comparative study of meropenem and imipenem/cilastatin sodium in bacterial pneumonia [in Japanese]. Chemotherapy (Tokyo) 1992 Nov; 40: 1343–64
Hara K, Kono S, Koga H, et al. A comparative study of meropenem and imipenem/cilastatin sodium in chronic respiratory tract infections [in Japanese]. Chemotherapy (Tokyo) 1992 Dec; 40: 1426–50
Hara K, Kono S, Koga H, et al. Dose-finding study on meropenem in bacterial pneumonia [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 447–63
Rizzato G, Montemurro L, Fanti D, et al. Meropenem versus imipenem: relationship between microbiological parameters and clinical outcome in lower respiratory tract infections. Curr Ther Res 1993 Dec; 54: 731–52
Romanelli G, Cravarezza P, Italian Intramuscular Meropenem Study Group. Intramuscular meropenem in the treatment of bacterial infections of the urinary and lower respiratory tracts. J Antimicrob Chemother. In press
Mouton YJ, Beuscart C, Meropenem Study Group. Empirical monotherapy with meropenem in serious bacterial infections. J Antimicrob Chemother. In press
Sieger B, Geckler R. A comparison of meropenem and ceftazidime plus tobramycin in the treatment of hospital-acquired lower respiratory infections [abstract]. In: 33rd Interscience Conference on Antimicrobial Agents and Chemotherapy 1993: 236
Vetter N. Efficacy of meropenem in the treatment of respiratory tract infection: a comparative evaluation [abstract]. 8th Mediterranean Congress on Chemotherapy; 1992 May 24–29 1992
Byrne S, Maddison J, Connor P, et al. Clinical evaluation of meropenem versus ceftazidime for the treatment of Pseudomonas spp. infections in cystic fibrosis patients. J Antimicrob Chemother. In press
Cox CE, Holloway WJ, Geckler RW. A multicenter comparative study of meropenem and imipenem/cilastatin in the treatment of complicated urinary tract infections in hospitalized patients. Clin Infect Dis. In press
Kumazawa J, Matsumoto T, Masatoshi T, et al. Clinical evaluation of meropenem for the treatment of complicated urinary tract infections–a double-blind controlled study using imipenem/cilastatin. Nishinippon Hinyokika 1992; 54(6): 954–69
Kumazawa J, Matsumoto T, Tanaka M, et al. Dose finding study of meropenem in complicated urinary tract infection [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 631–45
Naber KG. Efficacy of meropenem in both complicated and uncomplicated urinary tract infection: comparison with ceftazidime [abstract]. 8th Mediterranean Congress on Chemotherapy; 1992 May 24–29 1992
Lami JL, Wilson SE, Hopkins JA. Adjunctive antimicrobials in surgery of soft tissue infections: evaluation of cephalosporins and carbapenems. Am Surg 1991 Dec; 57: 769–74
Nakayama I, Yamaji E, Kawamura H, et al. Meropenem in abdominal skin and soft tissue infections in the field of surgery [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 486–99
Nakanishi H, Tamaki T, Minami H, et al. Skin tissue concentration and clinical evaluation of meropenem [in Japanese]. Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 746–50
Smith J, Muzik A, Nichols RL, et al. Randomised comparative study of meropenem vs imipenem/cilastatin in the treatment of skin and skin structure infections [abstract]. In: 31st Interscience Conference on Antimicrobial Agents and Chemotherapy 1991: 201 (N)
Nichols RL, Smith JW, Geckler RW, et al. Meropenem versus imipenem/cilastatin in the treatment of hospitalised patients with skin and soft tissue infections. South Med J 1995; 88: 397–404
Hemsell D, Martens M. A comparison of meropenem and clindamycin-gentamicin in the treatment of obstetric and gynaecologic infections [abstract]. In: 9th Mediterranean Congress of Chemotherapy 1994: 100
Cho N, Araki H, Kimura T, et al. Basic and clinical studies of meropenem in obstetrics and gynecology [in Japanese], Chemotherapy (Tokyo) 1992 Apr; 40 Suppl. 1: 657–68
Matsuda S. Clinical study of SM-7338 in obstetrics and gynecology [in Japanese]. Chemotherapy (Tokyo) 1992 Jan; 40: 133
Matsuda S. Pharmacokinetic and clinical studies of meropenem in the field of obstetrics and gynaecological infection [abstract], 8th Mediterranean Congress on Chemotherapy; 1992 May 24–29 1992
Solberg CO, Sjursen H. Safety and efficacy of meropenem in patients with septicaemia: a randomised comparison with ceftazidime, alone or combined with amikacin. J Antimicrob Chemother. In press
Blumer JL. Carbapenems in paediatrics. Scand J Infect Dis. In press
Schuler D, Merrem/Meronem Paediatric Study Group. Safety and efficacy of meropenem in hospitalised children: randomised comparison with cefotaxime, alone and combined with metronidazole or amikacin. Zeneca Pharmaceuticals (Data on file)
Blumer JL. Meropenem (M) compared with cefotaxime (C) —based regimens in pediatric (P) infections. Zeneca Pharmaceuticals. Abstract no. M28; (Data on file)
Norrby SR, Newell PA, Faulkner KL, et al. Safety profile of meropenem: international clinical experience based on the first 3125 patients treated with meropenem. J Antimicrob Chemother. In press
Winston DJ, Ho WG, Bruckner DA, et al. Beta-lactam antibiotic therapy in febrile granulocytopenic patients: a randomized trial comparing cefoperazone plus piperacillin, ceftazidime plus piperacillin, and imipenem alone. Ann Intern Med 1991; 115: 849–59
Freifeld AG, Walsh T, Marshall D, et al. Monotherapy for fever and neutropenia in cancer patients: a randomized comparison of ceftazidime versus imipenem. J Clin Oncol 1995; 13: 165–76
Bergan T, Nord CE, Thorsteinsson SB. Effect of meropenem on the intestinal microflora. Eur J Clin Microbiol Infect Dis 1991 Jun; 10: 524–7
Patel J, Giles RE. Meropenem: evidence of lack of proconvulsive tendency in mice. J Antimicrob Chemother 1989 Sep; 24 Suppl. A: 307–9
De Sarro A, Ammendola D, Zappala M, et al. Relationship between structure and convulsant properties of some β-lactam antibiotics following intracerebroventricular microinjection in rats. Antimicrob Agents Chemother 1995; 39: 232–7
Wong VK, Wright HT, Ross LA, et al. Imipenem/cilastatin treatment of bacterial meningitis in children. Pediatr Infect Dis J 1991; 10: 122–5
Prescription products: Meronem vials. Pharm J 1995; 254: 299
Core prescribing information: essential information document. Zeneca Pharmaceuticals (Data on file)
Zeneca. Merrem/meronem technical monograph. Cheshire, England, 1995.
Del Favero A. Clinically important aspects of carbapenem safety. Curr Opin Infect Dis 1994; 7 Suppl. 1: S38–42
Author information
Authors and Affiliations
Additional information
Various sections of the manuscript reviewed by: J.L. Blunter, Departments of Pediatrics and Pharmacology, University Hospitals of Cleveland, Cleveland, Ohio, USA; R. Dagan, Paediatric Infectious Disease Unit, Ben Gurion University of the Neger, Beer-Sheva, Israel; J.A. Garcia-Rodriguez, Department of Microbiology, Hospital Clinico Universitario, Salamanca, Spain; S.J. Geroulanos, Onassis Cardiac Surgery Center, Athens, Greece; W.K.J. Huizinga, Department of Surgery, Natal University Medical School, Durban, South Africa; R.N. Jones, Department of Pathology, University of Iowa Hospitals and Clinics, Iowa City, Iowa, USA; G. Ravizzola, Institute of Microbiology, University of Brescia, Brescia, Italy; E. Schmutzhard, Neurology Department, University Clinic of Innsbruck, Innsbruck, Austria; H. Sjursen, College of Medicine, Haukeland Hospital, Bergen, Norway; Y. Sumita, Episome Institute, Gunma, lapan; J. Turnidge, Microbiology and Infectious Diseases Department, Monash Medical Centre, Clayton, Victoria, Australia; S.E. Wilson, Department of Surgery, California College of Medicine, Orange, California, USA; R. Wise, Department of Medical Microbiology, Dudley Road Hospital, Birmingham, England.
Rights and permissions
About this article
Cite this article
Wiseman, L.R., Wagstaff, A.J., Brogden, R.N. et al. Meropenem. Drugs 50, 73–101 (1995). https://doi.org/10.2165/00003495-199550010-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199550010-00007