Abstract
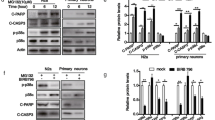
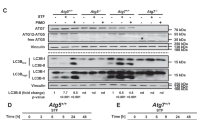
Increased autophagic vacuoles (AVs) occur in injured or degenerating neurons, under both developmental and pathological situations. Although an induced autophagy has been shown in inflammation response to cell factors, the underlying mechanism(s) remain(s) unknown. Here, we show that both cell factor IL-6 and environmental toxin MPP+ promote the formation of vacuolation in SHSY5Y cells. By electron and immunofluorescent microscopy analyses, we showed that these structures are acid autolysosomes, containing cellular debris, and labeled by LC3 or LAMP1, markers of autophagosomes or lysosomes, respectively. Combining MPP+ and IL-6 do not further increase vacuolation of SHSY5Y cells, and the vacuolation is less than that in the MPP+-treated group. MPP+-induced vacuolation results from significant increase in autophagy formation and delay in autophagy degradation, in relation to a decline of the lysosomal activity of arylsulfatase A. At molecular level, we show that this defect in autolysosomal maturation is independent of mammalian target of rapamycin and p38 inhibitions. Most importantly, we provide the first evidence that activation of ERK pathway is sufficient to commit cell to autophagic vacuolation. The sustained activation is required for MPP+ to disrupt the autophagic pathway. IL-6 also induces a temporary and significant activation of ERK, but not sustained activation, and change sustained activation in MPP+-treated group into temporary activation. Taken together, these findings strongly support that IL-6 promotes the maturation of autophagosomes into functional autolysosomes by regulating ERK.







Similar content being viewed by others
References
Itakura E, Mizushima N (2010) Characterization of autophagosome formation site by a hierarchical analysis of mammalian Atg proteins. Autophagy 6:764–776
Klionsky DJ, Emr SD (2000) Autophagy as a regulated pathway of cellular degradation. Science 290:1717–1721
Kirkegaard K, Taylor MP, Jackson WT (2004) Cellular autophagy: surrender, avoidance and subversion by microorganisms. Nat Rev Microbiol 2:301–314
Dagda RK, Zhu JH, Kulich SM, Chu CT (2008) Mitochondrially localized ERK2 regulates mitophagy and autophagic cell stress implications for Parkinson’s disease. Autophagy 4:770–782
Arico S, Petiot A, Bauvy C, Dubbelhuis PF, Meijer AJ, Codogno P, Ogier-Denis E (2001) The tumor suppressor PTEN positively regulates macroautophagy by inhibiting the phosphatidylinositol 3-kinase/protein kinase B pathway. J Biol Chem 276:35243–35246
Feng Z, Zhang H, Levine AJ, Jin S (2005) The coordinate regulation of the p53 and mTOR pathways in cells. Proc Natl Acad Sci USA 102:8204–8209
Zhu JH, Horbinski C, Guo FL, Watkins S, Uchiyama Y, Chu CT (2007) Regulation of Autophagy by Extracellular Signal-Regulated Protein Kinases During 1-Methyl-4-Phenylpyridinium-Induced Cell Death. Am J Pathol 170:75–86
Yoon S, Woo SU, Kang JH, Kim K, Kwon MH, Park S, Shin HJ, Gwak HS, Chwae YJ (2010) STAT3 transcriptional factor activated by reactive oxygen species induces IL6 in starvation-induced autophagy of cancer cells. Autophagy 6:1125–1138
Kimura A, Ishida Y, Wada T, Hisaoka T, Morikawa Y, Sugaya T, Mukaida N, Kondo T (2010) The absence of interleukin-6 enhanced arsenite-induced renal injury by promoting autophagy of tubular epithelial cells with aberrant extracellular signal-regulated kinase activation. Am J Pathol 176:40–50
Mograbi B, Corcelle E, Defamie N, Samson M, Nebout M, Segretain D, Fénichel P, Pointis G (2003) Aberrant connexin 43 endocytosis by the carcinogen lindane involves activation of the ERK/mitogen-activated protein kinase pathway. Carcinogenesis 24:1415–1423
Le’Negrate G, Selva E, Auberger P, Rossi B, Hofman P (2000) Sustained polymorphonuclear leukocyte transmigration induces apoptosis in T84 intestinal epithelial cells. J Cell Biol 150:1479–1488
Eskelinen EL (2005) Maturation of autophagic vacuoles in mammalian cells. Autophagy 1:1–10
Salthun-Lassalle B, Hirsch EC, Wolfart J, Ruberg M, Michel PP (2004) Rescue of mesencephalic dopaminergic neurons in culture by low-level stimulation of voltage-gated sodium channels. J Neurosci 24:5922–5930
Koster A, Saftig P, Matzner U, von Figura K, Peters C, Pohlmann R (1993) Targeted disruption of the M(r) 46,000 mannose 6-phosphate receptor gene in mice results in misrouting of lysosomal proteins. EMBO J 12:5219–5223
Kim I, Lemasters JJ (2010) Mitophagy selectively degrades individual damaged mitochondria after photoirradiation. Antioxid Redox Signal 14:1919–1928
Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, Kominami E, Ohsumi Y, Yoshimori T (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19:5720–5728
Petiot A, Pattingre S, Arico S, Meley D, Codogno P (2002) Diversity of signaling controls of macroautophagy in Mammalian cells. Cell Struct Funct 27:431–441
Yamamoto A, Tagawa Y, Yoshimori T, Moriyama Y, Masaki R, Tashiro Y (1998) Bafilomycin A1 prevents maturation of autophagic vacuoles by inhibiting fusion between autophagosomes and lysosomes in rat hepatoma cell line, H-4-II-E cells. Cell Struct Funct 23:33–42
Aoki H, Takada Y, Kondo S, Sawaya R, Aggarwal BB, Kondo Y (2007) Evidence that curcumin suppresses the growth of malignant gliomas in vitro and in vivo through induction of autophagy: role of Akt and extracellular signal-regulated kinase signaling pathways. Mol Pharmacol 72:29–39
Arany I, Megyesi JK, Nelkin BD, Safirstein RL (2006) STAT3 attenuates EGFR-mediated ERK activation and cell survival during oxidant stress in mouse proximal tubular cells. Kidney Int 70:669–674
Roca H, Varsos ZS, Sud S, Craig MJ, Ying C (2009) Pienta KJ.CCL2 and Interleukin-6 promote survival of human CD11b+ peripheral blood mononuclear cells and induce M2-type macrophage polarization. J Biol Chem 284:34342–34354
Shintani T, Klionsky DJ (2004) Autophagy in health and disease: a double-edged sword. Science 306:990–995
Tanida I, Minematsu-Ikeguchi N, Ueno T, Kominami E (2005) Lysosomal turnover, but not a cellular level, of endogenous LC3 is a marker for autophagy. Autophagy 1:c2–c9
Corcelle E, Djerbi N, Mari M, Nebout M, Fiorini C, Fénichel P, Hofman P, Poujeol P, Mograbi B (2007) Control of the autophagy maturation step by the MAPK ERK and p38: lessons from environmental carcinogens. Autophagy 3:57–59
Min Y, Xu W, Liu D, Shen S, Lu Y, Zhang L, Wang H (2010) Autophagy promotes BCG-induced maturation of human dendritic cells. Acta Biochim Biophys Sin 42:177–182
Jung Y, Yi YS, Yoo DS, Kim JH, Yang WS, Lee J, Park KW, Kweon DH, Hong S, Cho JY (2013) 8-(Tosylamino) quinoline inhibits tumour progression through targeting phosphoinositide-3-kinase/Akt pathway. Pharmazie 68(2):146–152
Robert G, Jullian V, Jacquel A, Ginet C, Dufies M, Torino S, Pottier A, Peyrade F, Tartare-Deckert S, Bourdy G, Deharo E, Auberger P (2012) Simalikalactone E (SkE), a new weapon in the armamentarium of drugs targeting cancers that exhibit constitutive activation of the ERK pathway. Oncotarget 3(12):1688–1699
Pattingre S, Bauvy C, Codogno P (2003) Amino acids interfere with the ERK1/2-dependent control of macroautophagy by controlling the activation of Raf -1 in human colon cancer HT-29 cells. J Biol Chem 278:16667–16674
Marshall CJ (1995) Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell 80:179–185
Acknowledgments
This study was supported by the Board of Health of Zhenjiang, China (No. WS0907), grants from the Zhenjiang social development project (No. SH2010038) and the Jiangsu Natural Science Foundation of China (No. BK2008249).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, Xz., Sui, Cy., Chen, Q. et al. Promotion of autophagy at the maturation step by IL-6 is associated with the sustained mitogen-activated protein kinase/extracellular signal-regulated kinase activity. Mol Cell Biochem 380, 219–227 (2013). https://doi.org/10.1007/s11010-013-1676-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1676-9