Key Points
-
Psoriasis is a common immune disorder whose major manifestation is in the skin. It most frequently begins in young adults, and is a lifelong condition.
-
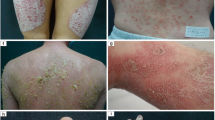
The most common form of psoriasis — psoriasis vulgaris or plaque-type psoriasis — is characterized by the presence of red, raised scaly plaques that can cover any body surface.
-
Patients with limited disease can respond well to topical treatments, but established therapies for more severe disease, such as systemic immunosuppressive drugs and phototherapy, have several drawbacks, including inconvenience and/or toxicity, which means that treatments are used intermittently.
-
So, patients experience cycles of clearance with normal quality of life that alternate with active disease and poor quality of life. There is therefore a need for therapies that result in safe and effective long-term maintenance of disease clearance for patients with moderate-to-severe psoriasis.
-
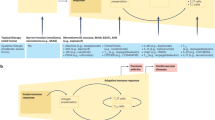
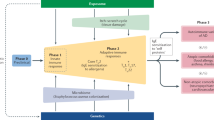
Advances in the understanding of psoriasis in recent years have highlighted the importance of aberrant activation of and migration of T cells into the skin in the pathogenesis of the disease.
-
Several agents that target the molecules involved, such as tumour-necrosis factor-α, have been recently approved for the treatment of psoriasis, or are in clinical trials. Such agents have also proved valuable in understanding disease pathogenesis.
-
This article first reviews current knowledge on the pathogenesis of psoriasis, and then discusses clinical results with immunobiological agents that have been approved or that are in development. Finally, novel potential targets for the development of drugs to treat psoriasis are highlighted.
Abstract
Psoriasis is a chronic inflammatory skin disorder that is characterized by thickened, scaly plaques, and is estimated to affect ∼1–3% of the Caucasian population. Traditional treatments, although effective in patients with limited disease, have numerous shortcomings, including inconvenience and toxicity. These drawbacks mean that many patients experience cycles of disease clearance, in which normal quality of life alternates with active disease and poor quality of life. However, as this review discusses, recent advances have highlighted the key role of the immune system in the pathogenesis of psoriasis, and have provided new defined targets for therapeutic intervention, offering hope for safe and effective psoriasis treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Gottlieb, A. B. Novel immunotherapies for psoriasis: clinical research delivers new hope for patients and scientific advances. J. Invest. Dermatol. Symp. Proc. 9, 79–83 (2004).
Gottlieb, A. B. Psoriasis. Dis. Manag. Clin. Outcomes 1, 195–202 (1998).
Heydendael, V. M. R. et al. Methotrexate versus cyclosporine in moderate-to-severe chronic plaque psoriasis. N. Engl. J. Med. 349, 658–65 (2003).
Rapp, S. R., Feldman, S. R., Exum, M. L., Fleischer, A. B. & Reboussin, D. M. Psoriasis causes as much disability as other major medical diseases. J. Am. Acad. Dermatol. 41, 401–407 (1999). This article documents the significant impact of psoriasis on mental and physical well-being.
Krueger, G. et al. The impact of psoriasis on quality of life: results of a 1998 National Psoriasis Foundation Patient-Membership Survey. Arch. Dermatol. 137, 280–284 (1998). This article emphasizes the role of quality of life in the assessment of disease severity and response to treatment.
de Korte, J., Sprangers, M. A. G., Mombers, F. M. C. & Bos, J. D. Quality of life in patients with psoriasis: a systemic literature review. J. Invest. Dermatol. Symp. Proc. 9, 140–147 (2004).
Brockbank, J. & Gladman, D. Diagnosis and management of psoriatic arthritis. Drugs 62, 2447–2457 (2002).
Gottlieb, A. B. Psoriatic arthritis: a guide for dermatology nurses. Dermatol. Nursing 15, 107–118 (2003).
Farber, E. M., Nall, M. L. & Watson, W. Natural history of psoriasis in 61 twin pairs. Arch. Dermatol. 109, 207–211 (1974).
Zhou, X. et al. Novel mechanisms of T-cell and dendritic cell activation revealed by profiling of psoriasis on the 63,100-element oligonucleotide array. Physiol. Genomics 13, 69–78 (2003). This article uses molecular genetic techniques to study the pathogenesis of psoriasis.
Helms, C. et al. A putative RUNX1 binding site variant between SLC9A3R1 and NAT9 is associated with increased susceptibility to psoriasis. Nature Genet. 35, 349–356 (2003).
Ellis, C. N. et al. Cyclosporine for plaque-type psoriasis. Results of a multidose, double-blind trial. N. Engl. J. Med. 324, 277–284 (1991). This is one of the most-cited multicentre trials demonstrating the efficacy of cyclosporine for treating psoriasis.
Bos, J. D. The pathomechanisms of psoriasis: the skin immune system and cyclosporin. Br. J. Dermatol. 118, 141–155 (1988).
Gottlieb, A. B. et al. Studies of the effect of cyclosporine in psoriasis in vivo: combined effects on activated T lymphocytes and epidermal regenerative maturation. J. Invest. Dermatol. 98, 302–309 (1992).
Baker, B. S., Swain, A. F., Fry, L. & Valdimarsson, H. Epidermal T lymphocytes and HLA-DR expression in psoriasis. Br. J. Dermatol. 110, 555–564 (1984).
Gottlieb, A. B. et al. Expression of HLA-DR molecules by keratinocytes and presence of Langerhans cells in the dermal infiltrate of active psoriatic plaques. J. Exp. Med. 164, 1013–1028 (1986). This article demonstrated increased numbers of activated T cells in psoriatic plaques.
Gottlieb, A. B. & Krueger, J. G. in Psoriasis (ed. Dubertret, L.) 63–71 (ISED, Brescie, Italy, 1994).
Gottlieb, A. B. Immunopathology and immunomodulation. Med. Dermatol. 19, 649–657 (2001).
Gottlieb, A. B. Recombinantly engineered human proteins: transforming the treatment of psoriasis. Clin. Immunol. 105, 105–116 (2002).
Wrone-Smith, T. & Nickoloff, B. J. Dermal injection of immunocytes induces psoriasis. J. Clin. lnvest. 98, 1878–1887 (1996). This paper describes one SCID mouse model of psoriasis.
Gilhar, A., David, M., Ullmann, Y., Berkutski, T. & Kalish, R. S. T-lymphocyte dependence of psoriatic pathology in human psoriatic skin grafted to SCID mice. J. Invest. Dermatol. 109, 283–288 (1997). This paper describes one SCID mouse model of psoriasis.
Gottlieb, S. L. et al. Response of psoriasis to a lymphocyte-selective toxin (DAB389IL-2) suggests a primary immune, but not keratinocyte, pathogenic basis. Nature Med. 1, 442–447 (1995). This paper demonstrated that T cells were primary pathogenic factors in the maintenance of psoriatic plaques and opened the field of biologicals in psoriasis treatment.
Gottlieb, A. B., Bacha, P., Parker, K. & Strand, V. Use of the interleukin-2 fusion protein, DAB389IL-2, for the treatment of psoriasis. Dermatol. Ther. 5, 48–63 (1998).
Gottlieb, A. B. et al. Anti-CD4 monoclonal antibody treatment of moderate to severe psoriasis vulgaris: results of a pilot, multicenter, multiple dose, placebo-controlled study. J. Am. Acad. Dermatol. 43, 595–604 (2000).
Gottlieb, A. B. et al. A multiple dose randomized double-blind pacebo-controlled study to determine the efficacy and safety of a humanized anti-CD4 monoclonal antibody in the treatment of moderate to severe chronic psoriasis vulgaris. J. Invest. Dermatol. 110, 678 (1998).
Khandke, L. et al. Cycloporine in psoriasis treatment: inhibition of keratinocyte cell-cycle progression in G1 independent of effects on transforming growth factor-α/epidermal growth factor receptor pathways. Arch. Dermatol. 127, 1172–1179 (1991).
Bachelez, H. et al. Treatment of recalcitrant plaque psoriasis with a humanized non-depleting antibody to CD4. J. Autoimmun. 11, 53–62 (1998).
Austin, L. M., Coven, T. R., Bhardwaj, N., Steinman, R. & Krueger, J. G. Intraepidermal lymphocytes in psoriatic lesions are GMP-17 (TIA-1)+CD8+CD3+ CTLs as determined by phenotypic analysis. J. Cut. Pathol. 25, 79–88 (1998).
Weinshenker, B. G., Bass, B. H., Ebers, G. C. & Rice, G. P. A. Remission of psoriatic lesions with uromonab-CD3 (Orthoclone OKT3) treatment. J. Am. Acad. Dermatol. 20, 1132–1133 (1989).
Krueger, J. The immunologic basis for the treatment of psoriasis with new biologic agents. J. Am. Acad. Dermatol. 46, 1–23 (2002).
Weinstein, G. D. Methotrexate: diagnosis and treatment drugs five years later. Ann. Intern. Med. 86, 199–204 (1977).
Bos, J. D., VanJoost, T., Powles, A. V., Meinardi, M. M. H. M. & Fry, L. Use of cyclosporin in psoriasis. Lancet 2, 1500–1502 (1989).
Ellis, C. N. et al. Cyclosporine improves psoriasis in a double-blind study. JAMA 256, 3110–3116 (1986). This is one of the earlier multicentre trials of cyclosporine for psoriasis.
Boyman, O. et al. Spontaneous development of psoriasis in a new animal model shows an essential rolse for resident T cells and tumor necrosis factor α. J. Exp. Med. 199, 731–736 (2004).
Conrad, C. et al. Crucial role for intraepidermal T cells expressing the collagen-binding integrin α1β1 in psoriasis. J. Invest. Dermatol. 122, A14–079 (2004).
Springer, T. A. et al. The lymphocyte function-associated LFA-1, CD2, and LFA-2 molecules: cell adhesion receptors of the immune system. Ann. Rev. Immunol. 5, 223–252 (1987).
Springer, T. A. Adhesions receptors of the immune system. Nature 346, 425–434 (1990).
Biedermann, T., Rocken, M. & Carballido, J. M. TH1 and TH2 lymphocyte development and regulation of TH cell-mediated immune responses of the skin. J. Invest. Dermatol. Symp. Proc. 9, 5–14 (2004).
Ridge, J. P., DiRosa, F. & Matzinger, P. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer cell. Nature 393, 474–478 (1998).
Schoenberger, S. P., Toes, R. E. M., VanDerVoort, E. I. H., Offringa, R. & Melief, C. J. M. T-cell help for cytotoxic T lymphocytes is mediated by CD40–CD40L interactions. Nature 393, 480–483 (1998).
Bennett, S. R. M. et al. Help for cytotoxic-T-cell responses is mediated by CD40 signalling. Nature 393, 478–483 (1998).
June, C. H., Ledbetter, J. A., Linsley, P. S. & Thompson, C. B. Role of the CD28 receptor in T-cell activation. Immunol. Today 11, 211–216 (1990).
Abrams, J. R. et al. CTLA4Ig-mediated blockade of T-cell costimulation in patients with psoriasis vulgaris. J. Clin. Invest. 103, 1243–1252 (1999).
Abrams, J. et al. Blockade of T lymphocyte costimulation with cytotoxic T lymphocyte-associated antigen 4-immunoglobulin (CTLA4Ig) reverses the cellular pathology of psoriatic plaques, including the activation of keratinocytes, dendritic cells, and endothelial cells. J. Exp. Med. 192, 681–94 (2000).
Krueger, J. G. et al. Blockade of T-cell costimulation with CTLA4Ig (BMS-188667) reverses pathologic inflammation and keratinocyte activation in psoriatic plaques. J. Invest. Dermatol. 108, 555 (1997).
Lebwohl, M. et al. CTLA4Ig (BMS-188667)-mediated blockade of T cell costimulation in patients with psoriasis vulgaris. J. Invest. Dermatol. 108, 570 (1997).
Gottlieb, A. B. et al. Clinical and histologic response to single-dose treatment of moderate to severe psoriasis with an anti-CD80 monoclonal antibody. J. Am. Acad. Dermatol. 47, 692–700 (2002).
Gottlieb, A. B. et al. Evaluation and safety and clinical activity of multiple doses of the anti-CD80 monoclonal antibody, galiximab, in patients with moderate to severe psoraisis. Clin. Immunol. 111, 28–37 (2004).
Najarian, D. J. & Gottlieb, A. B. Connections between psoriasis and Crohn's disease. J. Am. Acad. Dermatol. 48, 805–821 (2003). This is a comprehensive review of the similarities between Crohn's disease and psoriasis.
Chaudhari, U. et al. Efficacy and safety of infliximab monotherapy for plaque-type psoriasis: a randomised trial. Lancet 357, 1842–1847 (2001). This is the first double-blind, placebo-controlled trial of TNF blockade monotherapy for psoriasis.
Gottlieb, A. B. et al. Pharmacodynamic and pharmacokinetic response to anti-tumor necrosis factor monoclonal antibody (Infliximab) treatment of moderate to severe psoriasis vulgaris. J. Am. Acad. Dermatol. 48, 68–75 (2003).
Gottlieb, A. B. et al. Infliximab monotherapy provides rapid and sustained benefit for plaque-type psoriasis. J. Am. Acad. Dermatol. 48, 829–835 (2003).
Leonardi, C., Gottlieb, A. & Zitnik, R. Efficacy and safety of ENBREL (etanercept) in patients with psoriasis: results of a phase III study. J. Invest. Dermatol. 121, 409 (2003).
Gottlieb, A. B. et al. A randomized trial of etanercept as monotherapy for psoriasis. Arch. Dermatol. 139, 1627–1632 (2003). This is the Phase II study for etanercept monotherapy for psoriasis.
Gottlieb, A. B. et al. Infliximab induction therapy in patients with severe plaque-type psoriasis: A randomized, double-blind, placbo-controlled trial. J. Am. Acad. Dermatol. (in the press)
Nickoloff, B. J., Mitra, R. S., Elder, J. T., Fisher, G. J. & Voorhees, J. J. Decreased growth inhibition by recombinant γ-interferon is associated with increased transforming growth factor-α production in keratinocytes cultured from psoriatic lesions. Br. J. Dermatol. 121, 161–174 (1989).
Grossman, R. M. et al. Interleukin-6 (IL-6) is expressed in high levels in psoriatic skin and stimulates proliferation of cultured human keratinocytes. Proc. Natl Acad. Sci. USA 86, 6367–6371 (1989).
Hancock, G. E., Kaplan, G. & Cohn, Z. A. Keratinocyte growth regulation by the products of immune cells. J. Exp. Med. 168, 1395–1402 (1988).
Braunstein, S. et al. GM-CSF activates regenerative epidermal growth and stimulates keratinocyte proliferation in human skin in vivo. J. Invest. Dermatol. 103, 601–604 (1994).
Rich, B. & Kupper, T. Cytokines: IL-20 — a new effector in skin inflammation. Curr. Biol. 11, R531–R534 (2001).
Romer, J. et al. Epidermal overexpression of interleukin-19 and -20 mRNA in psoriatic skin disappears after short-term treatment with cyclosporine a or calcipotriol. J. Invest. Dermatol. 121, 1306–1311 (2003).
Heng, M., Kloss, S., Kuehn, C. & Chase, D. Significance and pathogenesis of basal keratinocyte herniations in psoriasis. J. Invest. Dermatol. 87, 362–366 (1986).
Vaccaro, M. et al. The dermoepidermal junction in psoriatic skin as revealed by scanning electron microscopy. Arch. Dermatol. Res. 291, 396–369 (1999).
Boehncke, W. -H., Wortmann, S., Kaufmann, R., Mielke, V. & Sterry, W. A subset of macrophages located along the basement membrane ('lining cells') is a characteristic histopathological feature of psoriasis. Am. J. Dermatopathol. 17, 139–144 (1995).
Mansbridge, J. N. & Knapp, A. M. Changes in keratinocyte maturation during wound healing. J. Invest. Dermatol. 89, 253–263 (1987). This article describes the regenerative maturation phenotype.
Smoller, B. A. et al. Recessive dystrophic epidermolysis bullosa skin displays a chronic growth-activated immunophenotype. Arch. Dermatol. 126, 78–83 (1990).
Krueger, J. G., Gilleaudeau, P., Kikuchi, T. & Lee, E. Psoriasis-related subpopulations of memory CD4+ and CD8+ T cells are selectively reduced by Alefacept. J. Invest. Dermatol. 119, 345 (2002).
Wakita, H. & Takigawa, M. E-selectin and vascular cell adhesion molecule-1 are critical for initial trafficking of helper-inducer/memory T cells in psoriatic plaques. Arch. Dermatol 130, 457–463 (1994).
daSilva, A. J. et al. Alefacept, an immunomodulatory recombinant LFA-3/IgG1 fusion protein, induces CD16 signaling and CD2/CD16-dependent apoptosis of CD2+ cells. J. Immunol. 168, 4462–4471 (2002).
Ellis, C. N., Krueger, G. G. & Group, A. C. S. Treatment of chronic plaque psoriasis by selective targeting of memory effector T lymphocytes. N. Engl. J. Med. 345, 248–255 (2001). This is the Phase II study of alefacept for psoriasis.
Krueger, G. G. et al. Afefacept Clinical Study Group. A randomized, double-blind, placebo-controlled phase III study evaluating efficacy an dtolerability of 2 courses of alefacept in patients with chronic plaque psoriasis. J. Am. Acad. Dermatol. 47, 821–833 (2002).
Lebwohl, M. et al. An international, randomized, double-blind, placebo-controlled phase 3 trial of intramuscular alefacept in patients with chronic plaque psoriasis. Arch. Dermatol. 139, 719–727 (2003). This is the Phase III trial of intramuscular alefacept for psoriasis.
Gottlieb, A. B. et al. CD4+ T-cell-directed antibody responses are maintained in patients with psoriasis receiving alefacept: results of a randomized study. J. Am. Acad. Dermatol. 49, 816–825 (2003).
Gottlieb, A. et al. Effects of administration of a single dose of a humanized monoclonal antibody to CD11a on the immunobiology and clinical activity of psoriasis. J. Am. Acad. Dermatol. 42, 428–435 (2000). This is the first report of the efficacy of efalizumab in psoriasis.
Kraan, M. C. et al. Alefacept treatment in psoriatic arthritis: reduction of the effector T cell population in peripheral blood and synovial tissue is associated with improvement of clinical signs of arthritis. Arthritis Rheum. 46, 2776–2784 (2002). This is the first report of the efficacy of alefacept for psoriatic arthritis.
Gottlieb, A. B. et al. Psoriasis as a model for T-cell-mediated disease: Immunobiologic and clinical effects of treatment with multiple doses of efalizumab, an anti-CD11a monoclonal antibody. Arch. Derm. 138, 591–600 (2002).
Gottlieb, A. G. et al. Subcutaneously administered efalizumab (antiCD11a) improves signs and symptoms of moderate to severe plaque psoriasis. J. Cut. Med. Surg. 7, 198–207 (2003).
Lebwohl, M. et al. A Novel targeted T-cell modulator, efalizumab, for plaque psoriasis. N. Engl J. Med. 349, 2004–2013 (2003). This is one of the Phase III trials of efalizumab for psoriasis.
Papp, K., B., R. et al. The treatment of moderate to severe psoriasis with a new anti-CD11a monoclonal antibody. J. Am. Acad. Dermatol. 45, 665–674 (2001).
Motensen, D. et al. The pharmacokinetics and pharmacodynamics of efalizumab following 12 weeks of subcutaneous treatment in subjects with moderate to severe plaque psoriasis in a phase I, open-label, multi–center study. J. Invest. Dermatol. 121, 379 (2003).
Chamian, F. et al. Presence of 'inflammatory' dendritic cells in psoriasis vulgaris lesions and modulation by efalizumab (anti-CD11a). J. Invest. Dermatol. 122, A41–A245 (2004).
Gordon, K. B. et al. Efalizumab for patients with moderate to severe plaque psoriasis: a randomized controlled trial. JAMA 290, 3073–3080 (2004).
Gottlieb, A. B., Hamilton, T. K., Caro, I. & Gordon, K. B. Efficacy and safety outcomes of long-term efalizumab therapy in patients with moderate to severe plaque psoriasis: an update. J. Invest. Dermatol. 122, A57–A341 (2004).
Austin, L., Ozawa, M., Kikuchi, T. & Krueger, G. Intracellular TNF-α, IFN-γ, and IL-2 identify TC1 and TH1 effector populations in psoriasis vulgaris plaque lymphocytes: single–cell analysis by flow cytometry. J. Invest. Dermatol. 110, 649 (1998).
Ettehadi, P., Greaves, M. W., Wallach, D., Aderka, D. & Camp, R. D. R. Elevated tumour necrosis factor-α (TNF-α) biological activity in psoriatic skin lesions. Clin. Exp. Immunol. 96, 146–151 (1994).
Ritchlin, C. T., Haas-Smith, S. A., Li, P., Hicks, D. G., Schwarz, E. M. Mechanisms of TNF-α- and RANKL-mediated osteoclastogenesis and bone resorption in psoriatic arthritis. J. Clin. Invest. 111, 821–831 (2003). This is a paper on the pathogenetic role of TNF in psoriatic arthritis.
Gottlieb, A. B. et al. Pharmacodynamic and pharmacokinetic response to anti-tumor necrosis factor-α monoclonal antibody (infliximab) treatment of moderate to severe psoriasis vulgaris. J. Am. Acad. Dermatol. 48, 68–75 (2003).
Nickoloff, B. J. The immunologic and genetic basis of psoriasis. Arch. Dermatol. 135, 1104–1110 (1999).
Gottlieb, A. G. Clinical research helps elucidate the role of tumor necrosis factor-α in the pathogeesis of T1-mediated immune disorders: use of targeted immunotherapeutics as pathogenic probes. Lupus 12, 190–194 (2003).
Weisfelner, M. E. & Gottlieb, A. B. The role of apoptosis in human epidermal keratinocytes. J. Drugs. Dermatol. 2, 385–391 (2003).
Mease, P. J. et al. Etanercept in the treatment of psoriatic arthritis and psoriasis: a randomised trial. Lancet 356, 385–390 (2000). This is the first double-blind, placebo controlled report of TNF blockade in psoriatic arthritis.
Konig, A. et al. Inflammatory infiltrate and interleukin-8 expression in the synovium of psoriatic arthritis — an immunohistochemical and mRNA analysis. Rheumatol. Int. 17, 159–168 (1997).
Veale, D., Barnes, L., Rogers, S. & Fitz, G. O. Immunohistochemical markers for arthritis in psoriasis. Ann. Rheum. Dis. 53, 450–454 (1994).
Hohler, T. et al. A TNF-α promoter polymorphism is associated with juvenile onset psoriasis and psoriatic arthritis. J. Invest. Dermatol. 109, 562–565 (1997).
VanDenBrande, J. M. H. et al. Infliximab but not etanercept induces apoptosis in lamina propria T-lymphocytes from patients with Crohn's Disease. Gastroenterology 12, 1774–1785 (2003).
Scallon, B. et al. Binding and functional comjparisons of two types of tumor necrosis factor antagonists. J. Pharmacol. Exp. Ther. 301, 418–426 (2002).
Scallon, B. J., Moore, M. A., Trinh, H., Knight, D. M. & Ghrayeb, J. Chimeric anti-TNF-α monoclonal anti-body cA2 binds recombinant transmembrane TNF-α and activates immune effector functions. Cytokine 7, 251–259 (1995).
tenHove, T., vanMontfrans, C., Peppelenbosch, M. P. & vanDeventer, S. J. Infliximab treatment induces apoptosis of lamina propria T lymphocytes in Crohn's disease. Gut 50, 206–211 (2002).
Maini, R. et al. Infliximab (chimeric anti-tumour necrosis factor-α monoclonal antibody) versus placebo in theumatoid arthritis patients receiving concomitant methotrexate: a randomised phase III trial. Lancet 354, 1932–1939 (1999). This is a Phase III trial of infliximab in rheumatoid arthritis.
Targan, S. R. et al. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor-α for Crohn's disease. N. Engl. J. Med. 337, 1029–1035 (1997). This is a Phase III trial of infliximab in Crohn's disease.
Present, D. H. et al. Infliximab for the treatment of fistulas in patients with Crohn's disease. N. Engl. J. Med. 340, 1398–1401 (1999).
VanOosten, B. W. et al. Increased MRI activity and immune activation in two multiple sclerosis patients treated with monoclonal anti-tumor necrosis factor antibody CA2. Neurology 47, 1531–1534 (1996).
Oh, C. J., Das, K. M. & Gottlieb, A. B. Treatment with anti-tumor necrosis factor-α (TNF-α) monoclonal antibody dramatically decreases the clinical activity of psoriasis lesions. J. Am. Acad. Dermatol. 42, 829–830 (2000).
Gottlieb, A. B. et al. Infliximab induction therapy for patients with severe plaque-type psoriasis: A randomized, double-blind, placebo-controlled trial. J. Am. Acad. Dermatol. 51, 534–542 (2004).
Antoni, C. et al. The Infliximab Multinational Psoriatic Arthritis Controlled Trial (IMPACT). Arthritis Rheum. 46, S381 (2002).
Antoni, C. et al. The one year results of the infliximab multinational psoriatic arthritis controlled trial (IMPACT). Arthritis Rheum. 48, S285 (2003).
Mease, P. et al. Etanercept treatment of psoriatic arthritis: safety, efficacy, and the effect on disease progression. Arthritis Rheum. 50, 2264–2272 (2004). This is the Phase III study of etanercept for psoriatic arthritis.
Lovell, D. et al. Etanercept in children with polyarticular juvenile rheumatoid arthritis. N. Engl J. Med. 342, 763–769 (2000).
Moreland, I. W. et al. Longterm Safety and Efficacy of Etanercept in Patients with Rheumatoid Arthritis. J. Rheumatol. 28, 1238–1244 (2001).
Davis, J. C. et al. Recombinant human tumor necrosis factor receptor (etanercept) for treating ankylosing spondylitis. Arthritis Rheum. 48, 3230–3236 (2003).
Leonardi, C. L. et al. Etanercept as monotherapy in patients with psoriasis. N. Engl. J. Med. 349, 2014–2022 (2003). This is the report of one of the Phase III trials of etanercept monotherapy for psoriasis.
Gottlieb, A. B., Gordon, K. B., Wang, A. & Zitnik, R. Durability of treatment response following withdrawal from etanercept in psoriasis patients. J. Invest. Dermatol. 122, A51–306 (2004).
Gordon, K. B. et al. Efficacy of etanercept in an integrated multistudy database of patients with psoriasis. J. Invest. Dermatol. 122, A54–321 (2004).
Day, R. Adverse reactions to TNF-α inhibitors in rheumatoid arthritis. Lancet 359, 540–541 (2002).
Keystone, E. et al. Radiographic, clinical, and functional outcomes of treatment with adalimumab (a human anti-tumor necrosis factor monoclonal antibody) in patients with active rheumatoid arthritis receiving concomitant methotrexate therapy. Arthritis Rheum. 50, 1400–1411 (2004).
Ellerin, T., Rubin, R. H. & Weinblatt, M. Infections and anti-tumor necrosis factor α therapy. Arthritis Rheum. 48, 3013–3022 (2003).
Lee, E. et al. Increased expression of interleukin 23 p19 and p40 in lesional skin of patients with psoriasis vulgaris. J. Exp. Med. 199, 125–130 (2004).
Kauffman, C. L. et al. Treatment of moderate-to-severe psoriasis with human monoclonal antibody to human IL-12. J. Invest. Dermatol. 121, 353 (2003).
Kauffman, C. L. et al. A phase I study evaluating the safety, pharmacokinetics, and clinical response of a human IL-12 p40 antibody in subjects with plaque psoriasis. J. Invest. Dermatol. 123, 1037–1044 (2004).
Heufler, C., Koch, F. & Schuler, G. Granulocyte/macrophage colony-stimulating factor and interleukin 1 mediate the maturation of murine epidermal Langerhans cells into potent immunostimulatory dendritic cells. J. Exp. Med. 167, 700–705 (1988).
Witmer-Pack, M. D., Olivier, W., Valinsky, J., Schuler, G. & Steinman, R. M. Granulocyte/macrophage colony-stimulating factor is essential for the viability and function of cultured murine epidermal Langerhans cells. J. Exp. Med. 166, 1484–1498 (1987).
Kimber, I., Cumberbatch, M., Dearman, R. J., Bhushan, M. & Griffiths, C. E. M. Cytokines and chemokines in the initiation and regulation of epidermal Langerhans cell mobilization. Br. J. Dermatol. 142, 401–412 (2000).
McInnes, I. B., Gracie, J. A. & Liew, F. Y. Interleukin-18: a novel cytokine in inflammatory rheumatic disease. Arthritis. Rheum. 44, 1481–1483 (2001).
Ye, X. J., Tang, B., Kang, A. H., Myers, L. K. & Cremer, M. A. The roles of interleukin-18 in collagen-induced arthriis in the BB rat. Clin. Exp. Immunol. 136, 440–447 (2004).
Siegmund, B. et al. Neutralization of interleukin-18 reduces severity in murine colitis and intestinal IFN-γ and TNF-α production. Am. J. Physiol. Regul. Integr. Comp. Physiol. 281, R1264–R1273 (2001).
Jameson, S. C. Maintaining the norm: T-cell homeostasis. Nature Rev. Immunol. 2, 547–555 (2002).
Ruckert, R. et al. Inhibition of keratinocyte apoptosis by IL-15: a new parameter in the pathogenesis of psoriasis? J. Immunol. 165, 2240–2250 (2000).
Schon, M. P., Zollner, T. M. & Henning Boehncke, W. The molecular basis of lymphocyte recruitment to the skin: clues for pathogenesis and selective therapies of inflammatory disorders. J. Invest. Dermatol. 121, 951–962 (2003).
Fuhlbrigge, R. C., Kieffer, J. D., Armerding, D. & Kupper, T. S. Cutaneous lymphocyte antigen is a specialized form of PSGL-1 expressed on skin-homing T cells. Nature 389, 978–981 (1997).
Kupper, T. S. Immune and inflammatory processes in cutaneous tissues. Mechanisms and speculations. J. Clin. Invest. 86, 1783–1789 (1990).
Robert, C. & Kupper, T. S. Inflammatory skin diseases, T cells, and immune surveillance. Mech. Disease 341, 1817–1828 (1999).
Schon, M. P. et al. Efomycine M, a new specific inhibitor of selectin, impairs leukocyte adhesion and alleviates cutaneous inflammation. Nature Med. 8, 366–372 (2002).
Ikegami-Kuzuhara, A. Y. T., Ohmoto, H. Inoue, Y., Saito, T. T. Therapeutic potential of a novel synthetic selectin blocker, J-R9188 in allergic dermatitis. J. Pharmacol. 134, 1498–1504 (2001).
Shand, A. F. A. Potential therapeutic role for cytokine or adhesion molecule manipulation in Crohn's disease in the shadow of infliximab? Int. J. Colorectal. Dis. 18, 1–11 (2003).
Ghosh, S. et al. Natilizumab for active Crohn's disease. N. Engl. J. Med. 348, 24–32 (2003).
De Boer, O. et al. Increased expression of adhesion receptors in both lesional and non-lesional psoriatic skin. Arch. Dermatol. Res. 286, 304–311 (1994).
deBoer, O., Verhagen, C., Visser, A., Bos, J. & Das, P. Cellular interactions and adhesion molecules in psoriatic skin. Acta Derm. Venereol. Suppl. (Stockh.) 186, 15–18 (1994).
Griffiths, C. E. M., Voorhees, J. J. & Nickoloff, B. J. Characterization of intercellular adhesion molecule-1 and HLA-DR expression in normal and inflamed skin: modulation by recombinant γ interferon and tumor necrosis factor. J. Am. Acad. Dermatol. 20, 617–629 (1989).
Lisby, S., Ralfkiaer, E., Rothlein, R. & Vejlsgaard, G. L. Intercellular adhesion molecule-I (ICAM-I) expression correlated to inflammation. Br. J. Dermatol. 120, 479–484 (1989).
Onuma, S. Immunohistochemical studies of infiltrating cells in early and chronic lesions of psoriasis. J. Dermatol. 21, 223–232 (1994).
Pauls, K. et al. Role of integrin αE(CD103)β7 for tissue-specific epidermal localization of CD8+ T lymphocytes. J. Invest. Dermatol. 117, 569–575 (2001).
Teraki, Y. & Shiohara, T. Preferential expression of αEβ7 integrin (CD103) on CD8+ T cells in the psoriatic epidermis: regulation by interleukins 4 and 12 and transforming growth factor-β. J. Invest. Dermatol. 147, 1118–1126 (2002).
Nickoloff, B. J. Role of interferon-γ in cutaneous trafficking of lymphocytes with emphasis on molecular and cellular adhesion events. Arch. Dermatol. 124, 1835–1845 (1988).
Rich, B. IL-7 promotes Th1 cell differentiation. J. Invest. Dermatol. 122, A124–753 (2004).
Ferenczi, K., Murphy, J., Krzysiek, R. & Kupper, T. S. CXCR6 and its logand are consecutively expressed in skin. J. Invest. Dermatol. 122, A130–777 (2004).
Xia, Y. P. et al. Transgenic delivery of VEGF to mouse skin leads to an inflammatory condition resembling human psoriasis. Blood 102, 161–168 (2003).
Detmar, M. Evidence for vascular endothelial growth factor (VEGF) as a modifier gene in psoriasis. J. Invest. Dermatol. 122, XIV–XV (2004).
Young, H. S., Summers, A. M., Bhushan, M., Brenchley, P. E. C. & Griffiths, C. E. Single-nucleotide polymorphisms of vascular endothelial growth factor in psoriasis of early onset. J. Invest. Dermatol. 122, 209–215 (2004).
Austin, L., Ozawa, M., Kikuchi, T., Walters, I. & Krueger, J. The majority of epidermal T cells in Psoriasis vulgaris lesions can produce type 1 cytokines, interferon-γ, interleukin-2, and tumor necrosis factor-α, defining TC1 (cytotoxic T lymphocyte) and TH1 effector populations: a type 1 differentiation bias is also measured in circulating blood T cells in psoriatic patients. J. Invest. Dermatol. 113, 752–759 (1999).
Gottlieb, A. B., Luster, A. D., Posnett, D. N. & Carter, D. M. Detection of a γ-interferon-induced protein (IP-10) in psoriatic plaques. J. Exp. Med. 168, 941–948 (1988).
Gottlieb, A. B. Immunologic mechanisms in psoriasis. J. Invest. Dermatol. 95, 18S–19S (1990).
Livden, J. K., Nilsen, R., Bjerke, J. R. & Matre, R. In situ localization of interferons in psoriatic lesions. Arch. Dermatol. Res. 281, 392–397 (1989).
Cooper, K. D. et al. Increased levels of an interleukin-1 inhibitor block interleukin-1 activity but not immunoreactivity of mRNA expression of interleukin-1β in psoriasis skin. J. Invest. Dermatol. 90, 552 (1988).
Matthews, S. J. & McCoy, C. Thalidomide: a review of approved and investigational uses. Clin.Ther. 25, 342–395 (2003).
Kumar, S., Broehm, J. & Lee, J. C. p38 MAP kinases: key signalling molecules as therapeutic targets for inflammatory diseases. Nature Rev. Drug Discov. 2, 717–726 (2003).
Thomas, P. IL-4-induced immune deviation as therapy of psoriasis. Arch. Derm. Res. 293, 30 (2001).
Asadullah, K. et al. IL-10 is a key cytokine in psoriasis. Proof of principle by IL-10 therapy: a new therapeutic approach. J. Clin. Invest. 101, 783–794 (1998).
Trepicchio, W. et al. IL-11 is an immune-modulatory cytokine which downregulates IL-12, Type 1 cytokines, and multiple inflammation-associated genes in patients with psoriasis. J. Invest. Dermatol. 112, 598 (1999).
Krueger, J. G. et al. Successful in vivo blockade of CD25 (high-affinity interleukin 2 receptor) on T cells by administration of humanized anti-Tac antibody to patients with psoriasis. J. Am. Acad. Dermatol. 43, 448–458 (2000).
Altmeyer, P. et al. Antipsoriatic effect of fumaric acid derivatives. J. Am. Acad. Dermatol. 30, 977–981 (1994).
Hoefnagel, J., Thio, H., Willemze, R. & Bavinck, J. Long-term safety aspects of systemic therapy with fumaric acid esters in severe psoriasis. Br. J. Dermatol. 149, 363–369 (2003).
Hoxtermann, S., Nuchel, C. & Altmeyer, P. Fumaric acid esters suppress peripheral CD4- and CD8-positive lymphocytes in psoriasis. Dermatology 196, 223–230 (1998).
Litjens, N. H. R. et al. Beneficial effects of fumarate therapy in psoriasis vulgaris patients coincide with downregulation of type 1 cytokines. Br. J. Dermatol. 148, 444–451 (2003).
Loewe, R. et al. Dimethylfumarate inhibits tumor-necrosis-factor-induced CD62E Expression in an NF-κB-dependent manner. J. Invest. Dermatol. 117, 1363–1368 (2001).
Mrowietz, U., Christophers, E. & Altmeyer, P. Treatment of psoriasis with fumaric acid esters: results of a prospective multicentre study. Br. J. Dermatol. 138, 456–460 (1998).
Ockenfels, H., Schultewolter, T., Ockenfels, G., Funk, R. & Goos, M. The antipsoriatic agent dimethylfumarate immunomodulates T-cell cytokine secretion and inhibits cytokines of the psoriatic cytokine network. Br. J. Dermatol. 139, 390–395 (1998).
Thio, H., van der Schroeff, J., Nugteren-Huying, W., & Vermeer, B. Long-term systemic therapy with dimethylfumarate and monoethylfumarate (Fumaderm) in psoriasis. J. Eur. Acad. Dermatol. Venereol. 4, 35–40 (1995).
Zhu, K. & Mrowietz, U. Inhibition of dendritic cell differentiation by fumaric acid esters. J. Invest. Dermatol. 116, 203–208 (2001).
Treumer, F., Zhu, K., Glaser, R. & Mrowietz, U. Dimethylfumarate is a potent inducer of apoptosis in human T cells. J. Invest. Dermatol. 121, 1383–1388 (2003).
Jones, E. L., Epinette, W. W., Hackney, V. C., Menendez, L. & Frost, P. Treatment of psoriasis with oral mycophenolic acid. J. Invest. Dermatol. 65, 537–542 (1975).
Griffiths, D. L. & Gottlieb, A. Treatment with oral pimecrolimus significantly improves psoriasis with a clear dose–response effect. J. Invest. Dermatol. 121, 391 (2003).
Singh, F. & Weinberg, J. M. Oral tazarotene and oral pimecrolimus: novel oral therapies in development for psoriasis. J. Drugs Dermatol. 3, 141–143 (2004).
Ihle, J. N. The Stat family in cytokine signaling. Curr. Opin. Cell. Biol. 13, 211–217 (2001).
Manning, A. M. & Davis, R. J. Targeting JNK for therapeutic benefit: from junk to gold? Nature Rev. Drug Discov. 2, 554–565 (2003).
Karin, M., Yamamoto, Y., & Wang, Q. M. The IKKNF-kappaB system: a treasure trove for drug development. Nature Rev. Drug Discov. 3, 17–26 (2004).
Lu, I. et al. Modulation of epidermal differentiation, tissue inflammation, and T-lymphocyte infiltration in posoriatic plaques by topical calcitriol. J. Cutan. Pathol. 23, 419–434 (1996).
Gottlieb, S. L. et al. Etretinate promotes keratinocyte terminal differentiation and reduces T-cell infiltration in psoriatic epidermis. J. Cutan. Pathol. 23, 404–418 (1996).
Gottlieb, A. B., Chaudhari, U., Baker, D. G., Perate, M. & Dooley, L. T. The National Psoriasis Foundation Psoriasis Score (NPF-PS) system versus the Psoriasis Area Severity Index (PASI) and Physician's Global Assessment (PGA): a comparison. J. Drugs Dermatol. 3, 260–266 (2003).
Frederiksson, T. & Pettersson, U. Severe psoriasis oral therapy with a new retinoid. Dermatologica 157, 238–244 (1978).
Gladman, D. D. Effectiveness of psoriatic arthritis therapies. Sem. Arthritis Rheum. 33, 29–37 (2003).
Gladman, D. D. et al. Assessment of patients with psoriatic arthritis. A review of currently available measures. Arthritis Rheum. 50, 24–35 (2004).
Abbas, A. K. & Lichtman, A. H. Cellular and Molecular Immunology 506–521 (Elsevier, Philadelphia, 2003).
Vallat, V. P. et al. PUVA bath therapy strongly suppresses immunological and epidermal activation in psoriasis: a possible cellular basis for remittive therapy. J. Exp. Med. 180, 283–296 (1994).
Ferenczi, K., Burack, L., Pope, M., Krueger, J. & Austin, L. CD69 HLA-DR and the IL-2R identify persistently activated T cells in psoriasis vulgaris lesional skin: blood and skin comparisons by flow cytometry. J. Autoimmun. 14, 63–78 (2000).
Duncan, J. I. et al. Soluble IL-2 receptor and CD25 cells in psoriasis: effects of cyclosporin A and PUVA therapy. Clin. Exp. Immunol. 85, 293–296 (1991).
Krueger, J. G. et al. Successful ultraviolet B treatment of psoriasis is accompanied by a reversal of keratinocyte pathology and by selective depletion of intraepidermal T cells. J. Exp. Med. 182, 2057–2068 (1995).
Hodak, E. et al. Climatotherapy at the Dead Sea is a remittive therapy for psoriasis: combined effects on epidermal and immunologic activation. J. Am. Acad. Dermatol. 49, 451–457 (2003).
Nickoloff, B. & Griffiths, C. γ-Interferon induces different keratinocyte expression of HLA-DR, DQ and intercellular adhesion molecule-1 (ICAM-1) antigens. J. Invest. Dermatol. 90, 592 (1988).
Kupper, T. S. Immunologic targets in psoriasis. N. Engl J. Med. 349, 1987–1990 (2003).
Gottlieb, A. B. Clinical research helps elucidate the role of tumor necrosis factor-α (TNF-α) in the pathogenesis of T1 mediated immune disorders: use of targeted immunotherapeutics as pathogenic probes. Lupus 12, 192–196 (2003).
Goebeler, M. et al. The C-X-C chemokine Mig is highly expressed in the papillae of psoriatic lesions. J. Pathol. 184, 89–95 (1998).
Homey, B. et al. Up-regulation of macrophage inflammatory protein-3 α/CCL20 and CC chemokine receptor 6 in psoriasis. J. Immunol. 164, 6621–32 (2000).
Das, P. et al. Differential expression of ICAM-1, E-selectin and VCAM-1 by endothelial cells in psoriasis and contact dermatitis. Acta Derm. Venereol. Suppl. (Stockh.) 186, 21–2 (1994).
Morganroth, G. S., Chan, L. S., Weinstein, G. D., Voorhees, J. J. & Cooper, K. D. Proliferating cells in psoriatic dermis are comprised primarily of T cells, endothelial cells, and factor XIIIa+ perivascular dendritic cells. J. Invest. Dermatol. 96, 333–340 (1991).
Nickoloff, B. J. & Griffiths, C. E. M. Lymphocyte trafficking in psoriasis: a new perspective emphasizing the dermal dendrocyte with active dermal recruitment mediated via endothelial cells followed by intra-epidermal T-cell activation. J. Invest. Dermatol. 95, 35S–37S (1990).
Gottlieb, A. B., Chang, C. K., Posnett, D. N., Fanelli, B. & Tam, J. P. Detection of transforming growth factor α in normal, malignant, and hyperproliferative human keratinocytes. J. Exp. Med. 167, 670–675 (1988).
Acknowledgements
This work was funded in part by grants from the David Ju Foundation and a Center of Excellence grant from the Federation of Clinical Immunology Societies (FOCIS).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
A.B.G. receives research funding from Amgen Inc.; Biogen Idec, Inc.; Centocor, Inc.; Genentech, Inc; WH Conzen Chair in Clinical Pharmacology; Abbott Labs; Ligand Pharmaceuticals Inc.; Beiersdorf, Inc.; Fujisawa Healthcare, Inc.; Merck, Inc.; Celgene Corp.; and Novartis AG.
A.B.G.'s Speakers' Bureau Memberships include Amgen Inc.; Biogen Idec, Inc.; Wyeth Pharmaceuticals; Centocor, Inc.; and Schering-Plough Corp.
A.B.G. currently has consulting agreements with Amgen Inc.; Biogen Idec, Inc; CellGate, Inc; Centocor, Inc.; Genentech, Inc; Novartis AG; QUATRx Pharmaceuticals Co.; Wyeth Pharmaceuticals, Schering-Plough Corp.; Eisai; Celgene Corp.; Bristol Myers Squibb Co.; Beiersdorf, Inc., Warner Chilcott, Abbott Labs., Kemia, Sankyo
Related links
Related links
DATABASES
Entrez Gene
OMIM
Glossary
- TOPICAL THERAPY
-
Therapy applied to the skin.
- PHOTOTHERAPY
-
Ultraviolet light irradiation therapy.
- KERATINOCYTE
-
Keratinocytes are a type of cell found in the epidermis (the top layer of skin). Psoriasis is characterized by hyperproliferation of keratinocytes.
- NATURAL KILLER CELLS
-
(NK cells). Lymphocytes that confer innate immunity. They were originally defined on the basis of their cytolytic activity against tumour targets, but it is now recognized that they serve a broader role in host defence against invading pathogens.
- INTEGRIN
-
Integrins are a family of adhesion molecules that mediate cell–cell, cell–extracellular matrix and cell–pathogen interactions by binding to various ligands. Integrins are non-covalently associated α/β heterodimers.
- MAJOR HISTOCOMPATIBILITY COMPLEX
-
A genetic region encoding proteins that are involved in antigen presentation to T cells. MHC class I molecules bound to antigen are most often recognized by the T-cell receptors of CD8+ T cells.
- IMMUNOLOGICAL SYNAPSE
-
A large junctional structure that is formed at the cell surface between a T cell that is interacting with an antigen-presenting cell. Important molecules involved in T-cell activation — including the T-cell receptor, numerous signal-transduction molecules and molecular adaptors — accumulate in an orderly manner at this site.
- MEMORY EFFECTOR T CELLS
-
Antigen-experienced T cells that have immediate effector capabilities and can efficiently migrate to peripheral sites of inflammation.
- φX174
-
φX174 is an experimental immunogen (vaccine) used to assess immune integrity in humans
- MUNRO'S ABSCESSES
-
Collections of neutrophils in the stratum corneum in psoriatic plaques.
- SHARP SCORES
-
A radiographic score composite of joint-space narrowing and bone erosions in joints of the hand and wrist.
Rights and permissions
About this article
Cite this article
Gottlieb, A. Psoriasis: emerging therapeutic strategies. Nat Rev Drug Discov 4, 19–34 (2005). https://doi.org/10.1038/nrd1607
Issue Date:
DOI: https://doi.org/10.1038/nrd1607
This article is cited by
-
The phytochemical profiling, pharmacological activities, and safety of malva sylvestris: a review
Naunyn-Schmiedeberg's Archives of Pharmacology (2023)
-
Piperlongumine regulates epigenetic modulation and alleviates psoriasis-like skin inflammation via inhibition of hyperproliferation and inflammation
Cell Death & Disease (2020)
-
Network meta-analysis and cost per responder of targeted Immunomodulators in the treatment of active psoriatic arthritis
BMC Rheumatology (2018)
-
Isoliquiritigenin prevents the progression of psoriasis-like symptoms by inhibiting NF-κB and proinflammatory cytokines
Journal of Molecular Medicine (2016)
-
Unleashing the therapeutic potential of human kallikrein-related serine proteases
Nature Reviews Drug Discovery (2015)