Abstract
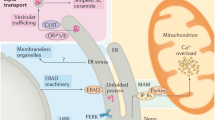
The endoplasmic reticulum (ER) is an intracellular organelle involved in maintaining protein and ion homeostasis and is vital for preserving cardiovascular health. Within the ER are a number of important proteins involved in sustaining cellular homeostasis, including molecular chaperones, Ca2+ binding proteins, and proteins involved in ER stress sensing and communication with other organelles. When ER homeostasis is disrupted, either by fluctuations in ion concentration or by a buildup of mis-folded protein, ER stress is triggered followed by a specific cellular response termed the Unfolded Protein Response (UPR). A number of conditions trigger ER stress, including hypoxia, ischemia/reperfusion, glucose starvation and several disease states. The UPR has three main effects; cessation of protein synthesis into the ER, up-regulation in expression of ER protein chaperones and up-regulation in ER-associated degradation (ERAD) of mis-folded proteins. The UPR can also trigger apoptosis if the insult is prolonged or severe. The cardiovascular system has recently been linked to ER stress with an ability to protect the heart, but is also observed to be pathologically damaging. Suppression of overall protein synthesis with up-regulation of ER resident molecular chaperones such as calreticulin and BiP/GRP78 may initially resolve the ER stress and restore ER homeostasis, but if ER stress conditions are extenuating, the UPR will stimulate apoptosis, characteristic of ischemia/reperfusion (I/R) injury in the heart. Pressure overload can also trigger ER stress, performing an important role in heart recovery. ER stress is implicated in several disease conditions including I/R of the brain or heart, heart failure and diabetes. UPR can be specifically activated in cardiac tissues by decreases in ATP, ER Ca2+, or UDP-glucose. An increase in UPR components such as ATF6 occurs within 24 hours of myocardial infarction in mice, as well as oxygen, serum and glucose deprivation in rats. As a number of cardiovascular diseases involve ER stress and the UPR, the understanding and management of ER stress and the UPR is of paramount importance.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- ASK1:
-
apoptosis signaling kinase 1
- ATF4:
-
activating transcription factor 4
- ATF6:
-
activating transcription factor 6
- ATG:
-
autophagic protein
- Bax:
-
Bcl2 associated X protein
- Bcl2:
-
B-cell CLL/lymphoma 2
- BH3:
-
Bcl-2 homology domain 3
- Bid:
-
BH3 interacting domain death agonist
- BiP:
-
immunoglobulin heavy chain-binding protein
- CHOP:
-
C/EBP-homologous protein
- CRE:
-
cyclic AMP response element
- CREB:
-
cAMP responsive element binding protein
- EDEM1:
-
endoplasmic reticulum degradation enhancer, mannosidase alpha-like 1
- eIF2α:
-
eukaryotic translation initiation factor 2A
- Elk1:
-
ETS domain containing protein
- ER:
-
endoplasmic reticulum
- ERAD:
-
endoplasmic reticulum associated degradation
- ERdj3:
-
endoplasmic reticulum DnaJ homolog 3
- ERdj4:
-
endoplasmic reticulum DnaJ homolog 4
- ERK1/2:
-
extracellular signal-regulated kinases 1 and 2
- ERp57:
-
ER protein 57
- ERp72:
-
endoplasmic reticulum protein 72
- ERp59:
-
ER protein 59
- ERSE:
-
endoplasmic reticulum stress element
- GADD34:
-
growth arrest and DNA damage inducible protein 34
- GRP94:
-
glucose related protein 94
- GRP78:
-
glucose related protein 78
- HSP40:
-
heat shock protein 40
- Hyou1:
-
hypoxia up regulated 1
- iNOS:
-
inducible nitric oxide synthase
- Ins3PR:
-
inositol 3-phosphate receptor
- I/R:
-
ischemia/reperfusion
- IRE1:
-
inositol requiring protein 1
- JIK:
-
c-Jun N-terminal inhibitory kinase
- JNK:
-
c-Jun kinase
- MEF2c:
-
myocyte enhancer factor 2C
- MCP1:
-
monocyte chemotactic protein 1
- MCPIP:
-
MCP1 inhibitory protein
- c-Myc:
-
myelocytomatosis viral oncogene homolog
- NFAT:
-
nuclear factor of activated T-cells
- Oasis:
-
old astrocyte specifically-induced substance
- PARM1:
-
prostatic androgen repressed message 1
- P58IPK:
-
protein kinase inhibitor of 58-kDa
- PDIa6:
-
protein disulfide isomerase family A, member 6
- PDI:
-
protein disulfide isomerase
- PERK:
-
protein kinase RNA-like endoplasmic reticulum kinase
- PI3K:
-
phosphoinositides 3-kinase
- RCAN1:
-
regulator of calcineurin 1
- ROS:
-
reactive oxygen species
- S1P:
-
site 1 protease
- S2P:
-
site 2 protease
- SERCA2:
-
sarco/endoplasmic reticulum Ca2+ ATPase 2
- SOD:
-
superoxide dismutase 1
- Stat3:
-
signal transducer and activator of transcription 3
- TRAF2:
-
tumor necrosis factor receptor associated factor 2
- UPR:
-
unfolded protein response
- UPRE:
-
unfolded protein response element
- XBP1:
-
X-box binding protein 1
References
Amuthan G, Biswas G, Ananadatheerthavarada HK, Vijayasarathy C, Shephard HM, Avadhani NG (2002) Mitochondrial stress-induced calcium signaling, phenotypic changes and invasive behavior in human lung carcinoma A549 cells. Oncogene 21(51):7839–7849
Araki E, Oyadomari S, Mori M (2003) Impact of endoplasmic reticulum stress pathway on pancreatic beta-cells and diabetes mellitus. Exp Biol Med (Maywood) 228(10):1213–1217
Avery J, Etzion S, DeBosch BJ, Jin X, Lupu TS, Beitinjaneh B, Grand J, Kovacs A, Sambandam N, Muslin AJ (2010). TRB3 function in cardiac endoplasmic reticulum stress. Circ Res 106(9):1516–1523
Azfer A, Niu J, Rogers LM, Adamski FM, Kolattukudy PE (2006) Activation of endoplasmic reticulum stress response during the development of ischemic heart disease. Am J Physiol Heart Circ Physiol 291(3):H1411–H1420
Bassik MC, Scorrano L, Oakes SA, Pozzan T, Korsmeyer SJ (2004) Phosphorylation of BCL-2 regulates ER Ca2+ homeostasis and apoptosis. Embo J 23(5):1207–1216
Belmont PJ, Chen WJ, San Pedro MN, Thuerauf DJ, Gellings Lowe N, Gude N, Hilton B, Wolkowicz R, Sussman MA, Glembotski CC (2010) Roles for endoplasmic reticulum-associated degradation and the novel endoplasmic reticulum stress response gene Derlin-3 in the ischemic heart. Circ Res 106(2):307–316
Belmont PJ, Tadimalla A, Chen WJ, Martindale JJ, Thuerauf DJ, Marcinko M, Gude N, Sussman MA, Glembotski CC (2008) Coordination of growth and endoplasmic reticulum stress signaling by regulator of calcineurin 1 (RCAN1), a novel ATF6-inducible gene. J Biol Chem. 283(20):14012–14021
Brostrom MA, Brostrom CO (2003) Calcium dynamics and endoplasmic reticular function in the regulation of protein synthesis: implications for cell growth and adaptability. Cell Calcium 34(4–5):345–363
Bicknell AA, Tourtellotte J, Niwa M (2010) Late phase of the endoplasmic reticulum stress response pathway is regulated by Hog1 MAP kinase. J Biol Chem 285(23):17545–17555
Dong S, Teng Z, Lu FH, Zhao YJ, Li H, Ren H, Chen H, Pan ZW, Lv YJ, Yang BF, Tian Y, Xu CQ, Zhang WH (2010) Post-conditioning protects cardiomyocytes from apoptosis via PKC(epsilon)-interacting with calcium-sensing receptors to inhibit endo(sarco)plasmic reticulum-mitochondria crosstalk. Mol Cell Biochem 341(1–2):195–206
Doroudgar S, Thuerauf DJ, Marcinko MC, Belmont PJ, Glembotski CC (2009) Ischemia activates the ATF6 branch of the endoplasmic reticulum stress response. J Biol Chem 284(43):29735–29745
Du K, Herzig S, Kulkarni RN, Montminy M (2003) TRB3: a tribbles homolog that inhibits Akt/PKB activation by insulin in liver. Science 300(5625):1574–1577
Eizirik DL, Cardozo AK, Cnop M (2008) The role for endoplasmic reticulum stress in diabetes mellitus. Endocr Rev 29(1):42–61
Fujita Y, Ito M, Nozawa Y, Yoneda M, Oshida Y, Tanaka M (2007) CHOP (C/EBP homologous protein) and ASNS (asparagine synthetase) induction in cybrid cells harboring MELAS and NARP mitochondrial DNA mutations. Mitochondrion 7(1–2):80–88
Galagudza M, Kurapeev D, Minasian S, Valen G, Vaage J (2004) Ischemic postconditioning: brief ischemia during reperfusion converts persistent ventricular fibrillation into regular rhythm. Eur J Cardiothorac Surg 25(6):1006–1010
Gargalovic PS, Gharavi NM, Clark MJ, Pagnon J, Yang WP, He A, Truong A, Baruch-Oren T, Berliner JA, Kirchgessner TG, Lusis AJ (2006) The unfolded protein response is an important regulator of inflammatory genes in endothelial cells. Arterioscler Thromb Vasc Biol 26(11):2490–2496
George I, Sabbah HN, Xu K, Wang N, Wang J (2011) Beta-adrenergic receptor blockade reduces endoplasmic reticulum stress and normalizes calcium handling in a coronary embolization model of heart failure in canines. Cardiovasc Res 91(3):447–455
Gill MB, Perez-Polo JR (2008) Hypoxia ischemia-mediated cell death in neonatal rat brain. Neurochem Res 33(12):2379–89
Glembotski CC (2007) Endoplasmic reticulum stress in the heart. Circ Res 101(10):975–984
Glembotski CC (2008) The role of the unfolded protein response in the heart. J Mol Cell Cardiol 44(3):453–459
Gorlach A, Klappa P, Kietzmann T (2006) The endoplasmic reticulum: folding, calcium homeostasis, signaling, and redox control. Antioxid Redox Signal 8(9–10):1391–1418
Gottlieb RA, Mentzer RM (2010) Autophagy during cardiac stress: joys and frustrations of autophagy. Annu Rev Physiol 72:45–59
Groenendyk J, Sreenivasaiah PK, Kim do H, Agellon LB, Michalak M (2010) Biology of endoplasmic reticulum stress in the heart. Circ Res 107(10):1185–1197
Gustafsson AB, Gottlieb RA (2009) Autophagy in ischemic heart disease. Circ Res 104(2):150–158
Hamada H, Suzuki M, Yuasa S, Mimura N, Shinozuka N, Takada Y, Nishino T, Nakaya H, Koseki H, Aoe T 2004) Dilated cardiomyopathy caused by aberrant endoplasmic reticulum quality control in mutant KDEL receptor transgenic mice. Mol Cell Biol 24(18):8007–8017
Isodono K, Takahashi T, Imoto H, Nakanishi N, Ogata T, Asada S, Adachi A, Ueyama T, Oh H, Matsubara H (2010) PARM-1 is an endoplasmic reticulum molecule involved in endoplasmic reticulum stress-induced apoptosis in rat cardiac myocytes. PLoS ONE 5(3):e9746
Jousse C, Deval C, Maurin AC, Parry L, Cherasse Y, Chaveroux C, Lefloch R, Lenormand P, Bruhat A, Fafournoux P (2007) TRB3 inhibits the transcriptional activation of stress-regulated genes by a negative feedback on the ATF4 pathway. J Biol Chem 282(21):15851–15861
Jung JH, Choi HJ, Kim SY, Hong, GS, Min SK, Nam MH, Kim CW, Koh YH, Seo JB (2011) Proteomic and biochemical analyses reveal the activation of unfolded protein response, ERK-1/2 and ribosomal protein S6 signaling in experimental autoimmune myocarditis rat model. BMC Genomics 12(1):520
Kin H, Zhao ZQ, Sun HY, Wang NP, Corvera JS, Halkos ME, Kerendi F, Guyton RA, Vinten-Johansen J (2004) Postconditioning attenuates myocardial ischemia-reperfusion injury by inhibiting events in the early minutes of reperfusion. Cardiovasc Res 62(1):74–85
Lecca MR, Wagner U, Patrignani A, Berger EG, Hennet T (2005) Genome-wide analysis of the unfolded protein response in fibroblasts from congenital disorders of glycosylation type-I patients. FASEB J 19(2):240–242
Li Y, Schwabe RF, DeVries-Seimon T, Yao PM, Gerbod-Giannone MC, Tall AR, Davis RJ, Flavell R, Brenner DA, Tabas I (2005) Free cholesterol-loaded macrophages are an abundant source of tumor necrosis factor-alpha and interleukin-6: model of NF-kappaB- and map kinase-dependent inflammation in advanced atherosclerosis. J Biol Chem 280(23):21763–21772
Liu XH, Zhang ZY, Andersson KB, Husberg C, Enger UH, Raeder MG, Christensen G, Louch WE (2011) Cardiomyocyte-specific disruption of Serca2 in adult mice causes sarco(endo)plasmic reticulum stress and apoptosis. Cell Calcium 49(4):201–207
Liu XH, Zhang ZY, Sun S, Wu XD (2008) Ischemic postconditioning protects myocardium from ischemia/reperfusion injury through attenuating endoplasmic reticulum stress. Shock 30(4):422–427
Marciniak SJ, Yun CY, Oyadomari S, Novoa I, Zhang Y, Jungreis R, Nagata K, Harding HP, Ron D (2004) CHOP induces death by promoting protein synthesis and oxidation in the stressed endoplasmic reticulum. Genes Dev 18(24):3066–3077
Martindale JJ, Fernandez R, Thuerauf D, Whittaker R, Gude N, Sussman MA, Glembotski CC (2006) Endoplasmic reticulum stress gene induction and protection from ischemia/reperfusion injury in the hearts of transgenic mice with a tamoxifen-regulated form of ATF6. Circ Res 98(9):1186–1193
Martinet W, Agostinis P, Vanhoecke B, Dewaele M, De Meyer GR (2009) Autophagy in disease: a double-edged sword with therapeutic potential. Clin Sci (Lond) 116(9):697–712
Martinet W, Knaapen MW, Kockx MM, De Meyer GR (2007) Autophagy in cardiovascular disease. Trends Mol Med 13(11):482–491
McCullough KD, Martindale JL, Klotz LO, Aw TY, Holbrook NJ (2001) Gadd153 sensitizes cells to endoplasmic reticulum stress by down-regulating Bcl2 and perturbing the cellular redox state. Mol Cell Biol 21(4):1249–1259
Minamino T Kitakaze M (2010) ER stress in cardiovascular disease. J Mol Cell Cardiol 48(6):1105–1110
Morishima N, Nakanishi K, Takenouchi H, Shibata T, Yasuhiko Y (2002) An endoplasmic reticulum stress-specific caspase cascade in apoptosis. Cytochrome c-independent activation of caspase-9 by caspase-12. J Biol Chem 277(37):34287–34294
Nakagawa T, Zhu H, Morishima N, Li E, Xu J, Yankner BA, Yuan J (2000) Caspase-12 mediates endoplasmic-reticulum-specific apoptosis and cytotoxicity by amyloid-beta. Nature 403(6765):98–103
Nishitoh H, Matsuzawa A, Tobiume K, Saegusa K, Takeda K, Inoue K, Hori S, Kakizuka A, Ichijo H (2002) ASK1 is essential for endoplasmic reticulum stress-induced neuronal cell death triggered by expanded polyglutamine repeats. Genes Dev 16(11):1345–1355
Nishitoh H, Saitoh M, Mochida Y, Takeda K, Nakano H, Rothe M, Miyazono K, Ichijo H (1998) ASK1 is essential for JNK/SAPK activation by TRAF2. Mol Cell 2(3):389–395
Niu J, Kolattukudy PE (2009) Role of MCP-1 in cardiovascular disease: molecular mechanisms and clinical implications. Clin Sci (Lond) 117(3):95–109
Oda Y, Okada T, Yoshida H, Kaufman RJ, Nagata K, Mori K (2006) Derlin-2 and Derlin-3 are regulated by the mammalian unfolded protein response and are required for ER-associated degradation. J Cell Biol 172(3):383–393
Ohoka N, Yoshii S, Hattori T, Onozaki K, Hayashi H (2005) TRB3, a novel ER stress-inducible gene, is induced via ATF4-CHOP pathway and is involved in cell death. Embo J 24(6):1243–1255
Ogata M, Hino S, Saito A, Morikawa K, Kondo S, Kanemoto S, Murakami T, Taniguchi M, Tanii I, Yoshinaga K, Shiosaka S, Hammarback JA, Urano F, Imaizumi K (2006) Autophagy is activated for cell survival after endoplasmic reticulum stress. Mol Cell Biol 26(24):9220–9231
Okada K, Minamino T, Tsukamoto Y, Liao Y, Tsukamoto O, Takashima S, Hirata A, Fujita M, Nagamachi Y, Nakatani T, Yutani C, Ozawa K, Ogawa S, Tomoike H, Hori M, Kitakaze M (2004) Prolonged endoplasmic reticulum stress in hypertrophic and failing heart after aortic constriction: possible contribution of endoplasmic reticulum stress to cardiac myocyte apoptosis. Circulation 110(6):705–712
Okada T, Yoshida H, Akazawa R, Negishi M, Mori, K (2002) Distinct roles of activating transcription factor 6 (ATF6) and double-stranded RNA-activated protein kinase-like endoplasmic reticulum kinase (PERK) in transcription during the mammalian unfolded protein response. Biochem J 366(Pt 2):585–594
Oyadomari S, Araki E, Mori M (2002) Endoplasmic reticulum stress-mediated apoptosis in pancreatic beta-cells. Apoptosis 7(4):335–345
Pan YX, Lin L, Ren AJ, Pan XJ, Chen H, Tang CS, Yuan WJ (2004) HSP70 and GRP78 induced by endothelin-1 pretreatment enhance tolerance to hypoxia in cultured neonatal rat cardiomyocytes. J Cardiovasc Pharmacol 44(Suppl 1):S117–S120
Papp S, Zhang X, Szabo E, Michalak M, Opas M (2008) Expression of Endoplasmic Reticulum Chaperones in Cardiac Develop-ment. Open Cardiovasc Med J 2:31–35
Peskoff A, Post JA, Langer GA (1992) Sarcolemmal calcium binding sites in heart: II. Mathematical model for diffusion of calcium released from the sarcoplasmic reticulum into the diadic region. J Membr Biol 129(1):59–69
Rao RV, Castro-Obregon S, Frankowski H, Schuler M, Stoka V, del Rio G, Bredesen DE, Ellerby HM (2002) Coupling Endoplasmic Reticulum Stress to the Cell Death Program. An Apaf-1-independent intrinsci pathway. J Biol Chem 277(24):21836–21842
Rao RV, Poksay KS, Castro-Obregon S, Schilling B, Row RH, del Rio G, Gibson BW, Ellerby HM, Bredesen DE (2004) Molecular components of a cell death pathway activated by endoplasmic reticulum stress. J Biol Chem 279(1):177–187
Ravingerova T (2007) Intrinsic defensive mechanisms in the heart: a potential novel approach to cardiac protection against ischemic injury. Gen Physiol Biophys 26(1):3–13
Reimold AM, Etkin A, Clauss I, Perkins A, Friend DS, Zhang J, Horton HF, Scott A, Orkin SH, Byrne MC, Grusby MJ, Glimcher LH (2000) An essential role in liver development for transcription factor XBP-1. Genes Dev 14(2):152–157
Renna M, Caporaso MG, Bonatti S, Kaufman RJ, Remondelli P (2007) Regulation of ERGIC-53 gene transcription in response to endoplasmic reticulum stress. J Biol Chem 282(31):22499–22512
Rzymski T, Milani M, Singleton DC, Harris AL (2009) Role of ATF4 in regulation of autophagy and resistance to drugs and hypoxia. Cell Cycle 8(23):3838–3847
Saito A, Hino S, Murakami T, Kondo S, Imaizumi K (2007) A novel ER stress transducer, OASIS, expressed in astrocytes. Antioxid Redox Signal 9(5):563–571
Schroder M (2008) Endoplasmic reticulum stress responses. Cell Mol Life Sci 65(6):862–894
Schroder M, Kaufman RJ (2005) ER stress and the unfolded protein response. Mutat Res 569(1–2):29–63
Shi Y (2004) Caspase activation: revisiting the induced proximity model. Cell 117(7):855–858
Shintani-Ishida K, Nakajima M, Uemura K, Yoshida K (2006) Ischemic preconditioning protects cardiomyocytes against ischemic injury by inducing GRP78. Biochem Biophys Res Commun 345(4):1600–1605
Simms MG, Walley KR (1999) Activated macrophages decrease rat cardiac myocyte contractility: importance of ICAM-1-dependent adhesion. Am J Physiol 277(1 Pt 2):H253–H260
Sun Y, Liu G, Song T, Liu F, Kang W, Zhang Y, Ge Z (2008) Upregulation of GRP78 and caspase-12 in diastolic failing heart. Acta Biochim Pol 55(3):511–516
Szegezdi E, Duffy A, O’Mahoney ME, Logue SE, Mylotte LA, O’Brien T, Samali A (2006) ER stress contributes to ischemia-induced cardiomyocyte apoptosis. Biochem Biophys Res Commun 349(4):1406–1411
Tabas I (2010) The role of endoplasmic reticulum stress in the progression of atherosclerosis. Circ Res 107(7):839–850
Takagi H, Matsui Y, Sadoshima J (2007) The role of autophagy in mediating cell survival and death during ischemia and reperfusion in the heart. Antioxid Redox Signal 9(9):1373–1381
Teixeira PF, Cerca F, Santos SD, Saraiva MJ (2006) Endoplasmic reticulum stress associated with extracellular aggregates. Evidence from transthyretin deposition in familial amyloid polyneuropathy. J Biol Chem 281(31):21998–22003
Thuerauf DJ, Marcinko M, Gude N, Rubio M, Sussman MA, Glembotski CC (2006) Activation of the unfolded protein response in infarcted mouse heart and hypoxic cultured cardiac myocytes. Circ Res 99(3):275–282
Toldo S, Severino A, Abbate A, Baldi A (2011) The role of PDI as a survival factor in cardiomyocyte ischemia. Methods Enzymol 489:47–65
Toth A, Nickson P, Mandl A, Bannister ML, Toth K, Erhardt P (2007) Endoplasmic reticulum stress as a novel therapeutic target in heart diseases. Cardiovasc Hematol Disord Drug Targets 7(3):205–218
Urano F, Wang X, Bertolotti A, Zhang Y, Chung P, Harding HP, Ron D (2000) Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science 287(5453):664–666
Vitadello M, Penzo D, Petronilli V, Michieli G, Gomirato S, Menabo R, Di Lisa F, Gorza L (2003) Overexpression of the stress protein Grp94 reduces cardiomyocyte necrosis due to calcium overload and simulated ischemia. FASEB J 17(8):923–925
Wang ZV, Rothermel BA, Hill JA (2010) Autophagy in hypertensive heart disease. J Biol Chem 285(12):8509–8514
Xin W, Lu X, Li X, Niu K, Cai J 2011 Attenuation of endoplasmic reticulum stress-related myocardial apoptosis by SERCA2a gene delivery in ischemic heart disease. Mol Med 17(3–4):201–210
Xu C, Bailly-Maitre B, Reed JC (2005) Endoplasmic reticulum stress: cell life and death decisions. J Clin Invest 115(10):2656–2664
Yoneda T, Imaizumi K, Oono K, Yui D, Gomi F, Katayama T, Tohyama M (2001) Activation of caspase-12, an endoplastic reticulum (ER) resident caspase, through tumor necrosis factor receptor-associated factor 2-dependent mechanism in response to the ER stress. J Biol Chem 276(17):13935–13940
Yorimitsu T, Klionsky DJ (2007) Eating the endoplasmic reticulum: quality control by autophagy. Trends Cell Biol 17(6):279–785
Younce CW, Kolattukudy PE (2010) MCP-1 causes cardiomyoblast death via autophagy resulting from ER stress caused by oxidative stress generated by inducing a novel zinc-finger protein, MCPIP. Biochem J 426(1):43–53
Zhang ZY, Liu XH, Hu WC, Rong F, Wu XD (2010) The calcineurin-myocyte enhancer factor 2c pathway mediates cardiac hypertrophy induced by endoplasmic reticulum stress in neonatal rat cardiomyocytes. Am J Physiol Heart Circ Physiol 298(5):H1499–H1509
Zhao ZQ, Corvera JS, Halkos ME, Kerendi F, Wang NP, Guyton RA, Vinten-Johansen J (2003) Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol 285(2):H579–H588
Acknowledgment
Work is our laboratory is supported by grants from the Canadian Institutes of Health Research, and Alberta Innovates-Heath Solutions.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Groenendyk, J., Michalak, M. (2012). Cardiovascular Disease and Endoplasmic Reticulum Stress. In: Agostinis, P., Afshin, S. (eds) Endoplasmic Reticulum Stress in Health and Disease. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-4351-9_15
Download citation
DOI: https://doi.org/10.1007/978-94-007-4351-9_15
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-4350-2
Online ISBN: 978-94-007-4351-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)