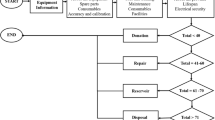
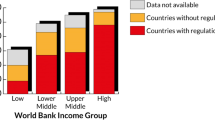
Abstract
Most of the world population is being treated in low-income countries, where there are not only harsh environmental conditions but also a failure to meet international standards and minimum requirements of the medical devices and locations. This can jeopardize the safe and efficient functioning of the medical devices. This paper draws on 5 field studies that took place in Sub-Saharan Africa, presenting few examples of donated medical devices and discussing the possible steps in order to strive for a more universal free healthcare coverage.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Arasaratnam, A., Humphreys, G.: Emerging economies drive frugal innovation. World Health Organ. Bull. World Health Organ. 91(1), 6 (2013)
WHO: Sustainable development goals. [cited 2018 14/12/2018]. Available from: https://sustainabledevelopment.un.org/?menu=1300
Iadanza, E., Dyro, J.: Clinical Engineering Handbook. Elsevier, Amsterdam (2004)
Williams, D.B., Kohler, J.C.J.C.P.: Maximizing the value of donated medical equipment in resource-limited settings: the roles of donors and end-users. CMBES Proceedings, 39(1) (2016)
Shah, M.T., et al.: Assessment of the availability of technology for trauma care in India. World J. Surg. 39(2), 363–372 (2015)
LaVigne, A.W., et al.: Cervical cancer in low and middle income countries: addressing barriers to radiotherapy delivery. Gynecol. Oncol. Rep. 22, 16–20 (2017)
Taylor, A., et al.: Innovating for global health: study of healthcare technology failure in southern Malawi. Ann. Glob. Health 82(3), 600–601 (2016)
Rusatira, J.C., et al.: Enabling access to medical and health education in rwanda using mobile technology: needs assessment for the development of mobile medical educator apps. JMIR Med. Educ. 2(1) (2016)
Pecchia, L.: Medical devices in low-resource settings. SCOPE 27(4), 41–44 (2018)
De Maria, C., et al.: Safe innovation: on medical device legislation in Europe and Africa. Health Policy Technol. (2018)
Pecchia, L.: Health technology assessment of medical devices in low and middle income countries: study design and preliminary results. In EMBEC & NBC 2017, pp. 225–228. Springer, Berlin (2017)
Approvazione dell’atto di indirizzo e coordinamento alle regioni e alle province autonome di Trento e di Bolzano, in materia di requisiti strutturali, tecnologici ed organizzativi minimi per l’esercizio delle attività sanitarie da parte delle strutture pubbliche e private (1997)
Invacare. Invacare® Perfecto2™ Oxygen Concentrator [cited 2018 12/12/2018]. Available from: https://www.invacare.com/doc_files/1143482.pdf
Zhang, W., Chatwin, C., Lui, R.: The Chinese medical device market: market drivers and investment prospects. J. Commer. Biotechnol. 22(2), 33–39 (2016)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this paper
Cite this paper
Piaggio, D., Medenou, D., Houessouvo, R.C., Pecchia, L. (2020). Donation of Medical Devices in Low-Income Countries: Preliminary Results from Field Studies. In: Badnjevic, A., Škrbić, R., Gurbeta Pokvić, L. (eds) CMBEBIH 2019. CMBEBIH 2019. IFMBE Proceedings, vol 73. Springer, Cham. https://doi.org/10.1007/978-3-030-17971-7_64
Download citation
DOI: https://doi.org/10.1007/978-3-030-17971-7_64
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-17970-0
Online ISBN: 978-3-030-17971-7
eBook Packages: EngineeringEngineering (R0)