Summary
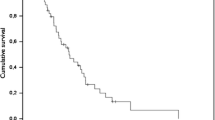
Thirty-two patients with recurrent glioma who had previously received radiation therapy and chemotherapy with nitrosoureas were treated with intravenous carboplatin every 3 weeks, starting at a dose of 350 mg/m2, with a dose escalation of 25 mg/m2 every 6 weeks until a level 4 hematologic toxicity was reached. Of the 28 patients who could be evaluated for a response, 50% demonstrated a response or had stabilization of their disease after two infusions of carboplatin. Their median time to tumor progression and median duration of survival were 19 weeks and 38 weeks. Thrombocytopenia was the major toxicity and was severe in one-third of the patients. No neurologic or renal toxicities were noted. Carboplatin has demonstrated activity against recurrent gliomas in patients who have already had extensive chemotherapy. Increasing the dose of carboplatin may improve the rate of response and the duration of progression-free survival in patients with recurrent glioma.
Similar content being viewed by others
References
Harsh GR IV, Levin VA, Gutin PH, Seager M, Silver P, Wilson CB: Reoperation for recurrent glioblastoma and anaplastic astrocytoma. Neurosurgery 21: 615–621, 1987
Leibel SA, Gutin PH, Wara WM, Silver PS, Larson DA, Edwards MSB, Lamb SA, Ham B, Weaver KA, Barnett C, Phillips TL: Survival and quality of life after interstitial implantation of removable high-activity iodine-125 sources for the treatment of patients with recurrent malignant gliomas. Int J Radiat Oncol Biol Phys 17: 1129–1139, 1989
Kornblith PL, Walker M: Chemotherapy for malignant gliomas. J Neurosurg 68: 1–17, 1988
Prados MD: Treatment strategies for patients with recurrent brain tumors. Semin Radiat Oncol 1: 62–68, 1991
Muggia FM: Overview of carboplatin. Replacing, complementing and extending the therapeutic horizons of cisplatin. Semin Oncol 16(Suppl 5): 7–13, 1989
de Graeff A, Slebos RJC, Rodenhuis S: Resistance to cisplatin and analogues: Mechanisms and potential clinical implications. Cancer Chemother Pharmacol 21: 325–332, 1988
Buckner JC, Brown LD, Cascino TL: Phase II evaluation of infusional etoposide and cisplatin in patients with recurrent astrocytoma. J Neurooncol 9: 249–254, 1990
Spence AM, Berger MS, Livingston RB: Phase II evaluation of high-dose intravenous cisplatin for treatment of adult malignant gliomas recurrent after chloroethylnitrosourea failure. J Neurooncol 12: 187–191, 1992
Stewart DJ, O'Bryan RM, Al-Sarraf M: Phase II study of cisplatin in recurrent astrocytomas in adults. A Southwest Oncology Group study. J Neurooncol 1: 145–147, 1983
Doz F, Berens ME, Dougherty DV, Rosenblum ML: Comparison of the cytotoxic activities of cisplatin and carboplatin against glioma cell lines at pharmacologically relevant drug exposures. J Neurooncol 11: 27–35, 1991
Poisson M, Pereon Y, Chiras J, Delattre JY: Treatment of recurrent malignant supratentorial gliomas with carboplatin (CBDCA). J Neurooncol 10: 139–144, 1991
Twelves CJ, Ash CM, Miles DW, Thomas DG, Souhami RL: Activity and toxicity of carboplatin and iproplatin in relapsed high-grade glioma. Cancer Chemother Pharmacol 27: 481–483, 1991
Walker RW, Dantis E, Shapiro WR: Treatment of recurrent glioma with carboplatin (CBDCA). Proc Am Soc Clin Oncol 6: 72, 1987
Yung WK, Mechtler L, Gleason MJ: Intravenous carboplatin for recurrent malignant glioma. A phase II study. J Clin Oncol 9: 860–864, 1991
Kaplan EL, Meier P: Nonparametric estimation from incomplete observations. J Am Stat Assoc 53: 457–481, 1958
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Warnick, R.E., Prados, M.D., Mack, E.E. et al. A phase II study of intravenous carboplatin for the treatment of recurrent gliomas. J Neuro-Oncol 19, 69–74 (1994). https://doi.org/10.1007/BF01051050
Issue Date:
DOI: https://doi.org/10.1007/BF01051050