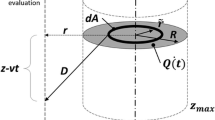
Abstract
Surgical robots have been proposed ex vivo to drill precise holes in the temporal bone for minimally invasive cochlear implantation. The main risk of the procedure is damage of the facial nerve due to mechanical interaction or due to temperature elevation during the drilling process. To evaluate the thermal risk of the drilling process, a simplified model is proposed which aims to enable an assessment of risk posed to the facial nerve for a given set of constant process parameters for different mastoid bone densities. The model uses the bone density distribution along the drilling trajectory in the mastoid bone to calculate a time dependent heat production function at the tip of the drill bit. Using a time dependent moving point source Green’s function, the heat equation can be solved at a certain point in space so that the resulting temperatures can be calculated over time. The model was calibrated and initially verified with in vivo temperature data. The data was collected in minimally invasive robotic drilling of 12 holes in four different sheep. The sheep were anesthetized and the temperature elevations were measured with a thermocouple which was inserted in a previously drilled hole next to the planned drilling trajectory. Bone density distributions were extracted from pre-operative CT data by averaging Hounsfield values over the drill bit diameter. Post-operative \(\mu\)CT data was used to verify the drilling accuracy of the trajectories. The comparison of measured and calculated temperatures shows a very good match for both heating and cooling phases. The average prediction error of the maximum temperature was less than 0.7 °C and the average root mean square error was approximately 0.5 °C. To analyze potential thermal damage, the model was used to calculate temperature profiles and cumulative equivalent minutes at 43 °C at a minimal distance to the facial nerve. For the selected drilling parameters, temperature elevation profiles and cumulative equivalent minutes suggest that thermal elevation of this minimally invasive cochlear implantation surgery may pose a risk to the facial nerve, especially in sclerotic or high density mastoid bones. Optimized drilling parameters need to be evaluated and the model could be used for future risk evaluation.







Similar content being viewed by others
References
Arrazola, P., T. Özel, D. Umbrello, M. Davies, and I. Jawahir. Recent advances in modelling of metal machining processes. CIRP Ann. Manuf. Technol. 62:695–718, 2013.
Aslan, A., H. S. Vatansever, G. G. Aslan, G. Eskiizmir, and G. Giray. Effect of thermal energy produced by drilling on the facial nerve: histopathologic evaluation in guinea pigs. J. Laryngol. Otol. 119:600–605, 2005.
Augustin, G., S. Davila, K. Mihoci, T. Udiljak, D. S. Vedrina, and A. Antabak. Thermal osteonecrosis and bone drilling parameters revisited. Arch. Orthop. Trauma Surg. 128:71–7, 2008.
Augustin, G., S. Davila, T. Udilljak, T. Staroveški, D. Brezak, and S. Babić. Temperature changes during cortical bone drilling with a newly designed step drill and an interally cooled drill. Int. Orthop. 36:1449–1456, 2012.
Augustin, G., T. Zigman, S. Davila, T. Udilljak, T. Staroveski, D. Brezak, and S. Babic. Cortical bone drilling and thermal osteonecrosis. Clin. Biomech. (Bristol, Avon) 27:313–25, 2012.
Bell, B., N. Gerber, T. Williamson, K. Gavaghan, W. Wimmer, M. Caversaccio, and S. Weber. In Vitro accuracy evaluation of image-guided robot system for direct cochlear access. Otol. Neurotol. 34:1284–1290, 2013.
Bell, B., C. Stieger, N. Gerber, M. Caversaccio, and S. Weber. A self-developed and constructer robot for minimally invasive cochlear implantation. Acta oto-laryngol. 132:355–360, 2012.
Carslaw, H. and J. Jeager. Conduction of Heat in Solids. 2nd edn. Oxford: Oxford University Press, 1986.
Carter, D. and W. Hayes. The compressive behavior of bone as a two-phase porous structure. J. Bone Joint Surg. 59:954–962, 1977.
Chen, H. L. and A. A. Gundjian. Specific heat of bone. Med. Biol. Eng. 14:548, 1974.
Davidson, S. R. and D. F. James. Measurement of thermal conductivity of bovine cortical bone. Med. Eng. Phys. 22:741–7, 2000.
Dolan, E. B., M. G. Haugh, M. C. Voisin, and D. Tallon. Thermally induced osteocyte damage initiates a remodelling signaling cascade. Plos One 10:1–17, 2015.
Drake, R. L., A. W. Vogl, and A. W. Mitchel. Gray’s Anatomy. 3rd edn. Philadelphia: Churchill Livingstone, 2014.
Eriksson, R. and T. Albrektsson. The effect of heat on bone regeneration: An experimental study in the rabbit using the bone growth chamber. J. Oral Maxillofac. Surg. 42:705–711, 1984.
Fayad, J. N., G. B. Wanna, J. N. Micheletto, and S. C. Parisier. Facial nerve paralysis following cochlear implant surgery. The Laryngoscope 113:1344–1346, 2003.
Gerber, N., B. Bell, K. Gavaghan, C. Weisstanner, and M. Caversaccio. Surgical planning tool for robotically assisted hearing aid implantation. Int. J. CARS 9:11–20, 2014.
Gross, T., D. H. Pahr, and P. K. Zysset. Morphology—elasticity relationships using decreasing fabric information of human trabecular bone from three major anatomical locations. Biomech. Model Mechanobiol. 12:793–800, 2013.
Gulya, A. J. and H. Schuknecht. Anatomy of the termporal bone with surgical implications. 3rd edn. Washington: Informa Healthcare, 2007.
Havemann, J. van der Zee, J. Wondergems, J. Effects of hypertermia on the peripheral nervous system: a review. Int. J. Hyperth. 20:371–391, 2004.
Kobler, J.-P., M. Schoppe, G. J. Lexow, T. S. Rau, O. Majdani, L. A. Kahrs, and T. Ortmaier. Temporal bone borehole accuracy for cochlear implantation influenced by drilling strategy: an in vitro study. Int. J. Comput. Assist. Radiol. Surg. 9:1033–1043, 2014.
Labadie, R. F., R. Balachandran, J. H. Noble, G. S. Blachon, J. E. Mitchell, F. A. Reda, B. M. Dawant, and J. M. Fitzpatrick. Minimally invasive image-guided cochlear implantation surgery: first report of clinical implementation. The Laryngoscope 9:1–8, 2013.
Lee, J., B. A. Gozen, and O. B. Ozdoganlar. Modeling and experimentation of bone drilling forces. J. Biomech. 45:1076–1083, 2011.
Lee, J., O. B. Ozdoganlar, and Y. Rabin. An experimental investigation on thermal exposure during bone drilling. Med. Eng. Phys. 34:1510–1520, 2012.
Lee, J., Y. Rabin, and O. B. Ozdoganlar. A new thermal model for bone drilling with applications to orthopaedic surgery. Med. Eng. Phys. 33:1234–1244, 2011.
Lughmani, W. A., K. Bouazza-Marouf, and I. Ashcroft. Finite element modeling and experimentation of bone drilling forces. J. Phys. Conf. Ser. 451:012034, 2013.
Maani, N., K. Farhang, and M. Hodaei. A model for the prediction of thermal response of bone in surgical drilling. J. Therm. Sci. Eng. Appl. 6:041005, 2014.
Misch, C. E.: Contemporary Implant Dentistry. 3rd edn. St. Louis: Elsevier, 2007.
Paek, U. and F. Gagliano. Thermal analysis of laser drilling process. IEEE J. Quantum Electron. 8:112–119, 1972.
Sapareto, S. A. and W. C. Dewey. Thermal dose determination in cancer therapy. Int. J. Radiat. Oncol. Biol. Phys. 10:787–800, 1984.
Schipper, J., A. Aschendorff, and R. Laszig. Navigation as a quality management tool in cochlear implant surgery. J. Laryngol. Otol. 118:764–760, 2004.
Shield, B. Evaluation of the social and economic costs of hearing impairment. A report for Hear-it. Technical Report October, 2012.
Soares, H. B. and L. Lavinsky. Histology of sheep temporal bone. Braz. J. Otorhinolaryngol. 77:285–292, 2011.
Sugita, N. and M. Mitsuishi. Specifications for machining the bovine cortical bone in relation to its microstructure. J. Biomech. 42:2826–2829, 2009.
de Vrind, H. H., J. Wondergem, and H. de Vrind. Hyperthermia-induced damage to rat sciatic nerve assessed in vivo with functional methods and with electrophysiology. J. Neurosci. Methods 45:165–174, 1992.
Williamson, T. M., B. J. Bell, N. Gerber, L. Salas, P. Zysset, M. Caversaccio, and S. Weber. Estimation of tool pose based on force-density correlation during robotic drilling. IEEE Trans. Biomed. Eng. 60:969–76, 2013.
Acknowledgements
The authors wish to thank Christina Precht from the Vetsuisse Department for her help in conducting the in vivo sheep studies. The authors would also like to thank Nano Tera and the Swiss National Science Foundation for funding the research within the hear restore project (RTD 2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Sean Kohles oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Feldmann, A., Anso, J., Bell, B. et al. Temperature Prediction Model for Bone Drilling Based on Density Distribution and In Vivo Experiments for Minimally Invasive Robotic Cochlear Implantation. Ann Biomed Eng 44, 1576–1586 (2016). https://doi.org/10.1007/s10439-015-1450-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-015-1450-0