Abstract
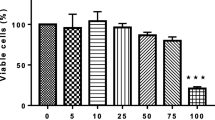
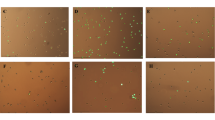
The phytochemical epigallocatechin gallate (EGCG) has been reported to alleviate age-associated immune disorders and organ dysfunction. However, information regarding the mechanistic role of EGCG in the suppression of cellular senescence is limited. The present study thus assessed the effects and underlying mechanisms of EGCG in the inhibition of senescence as well as its potential to selectively eliminate senescent cells (senolytics) using 3T3-L1 preadipocytes. Premature senescence was established in cells by repeated exposure of H2O2 at a sub-lethal concentration (150 μM). H2O2 treated cells showed characteristic senescence-associated features including increased cell size, senescence-associated β-galactosidase activity (SA-β-gal), development of senescence-associated secretory phenotype (SASP), activation of reactive oxygen species (ROS) and pathways, DNA damage as well as induction of cell cycle inhibitors (p53/p21WAF1/p16INK4a). In addition, a robust activation of PI3K/Akt/mTOR and AMPK pathways was also observed in H2O2 treated cells. Presence of EGCG (50 and 100 μM) showed significant downregulation of PI3K/Akt/mTOR and AMPK signaling along with the suppression of ROS, iNOS, Cox-2, NF-κB, SASP and p53 mediated cell cycle inhibition in preadipocytes. In addition, EGCG treatment also suppressed the accumulation of anti-apoptotic protein Bcl-2 in senescent cells thereby promoting apoptosis mediated cell death. Our results collectively show that EGCG acts as an mTOR inhibitor, SASP modulator as well as a potential senolytic agent thereby indicating its multi-faceted attributes that could be useful for developing anti-aging or age-delaying therapies.











Similar content being viewed by others
Abbreviations
- EGCG:
-
Epigallocatechin gallate
- mTOR:
-
Mechanistic target of rapamycin
- H2O2 :
-
Hydrogen peroxide
- SASP:
-
Senescence-associated secretory phenotype
- ROS:
-
Reactive oxygen species
- iNOS:
-
Inducible nitric oxide synthase
- Cox-2:
-
Cyclooxygenase-2
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- AMPK:
-
Adenosine monophosphate-activated protein kinase
References
Bent EH, Gilbert LA, Hemann MT (2016) A senescence secretory switch mediated by PI3K/AKT/mTOR activation controls chemoprotective endothelial secretory responses. Genes Dev 30(16):1811–1821
Bhatia-Dey N, Kanherkar RR, Stair SE, Makarev EO, Csoka AB (2016) Cellular senescence as the causal nexus of aging. Front Genet 7:13
Biran A, Zada L, Abou Karam P, Vadai E, Roitman L, Ovadya Y, Porat Z, Krizhanovsky V (2017) Quantitative identification of senescent cells in aging and disease. Aging Cell 16(4):661–671
Blagosklonny MV (2008) Aging: ROS or TOR. Cell Cycle 7(21):3344–3354
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brown MK, Evans JL, Luo Y (2006) Beneficial effects of natural antioxidants EGCG and alpha-lipoic acid on life span and age-dependent behavioral declines in Caenorhabditis elegans. Pharmacol Biochem Behav 85(3):620–628
Campisi J (2013) Aging, cellular senescence, and cancer. Annu Rev Physiol 75:685–705
Carroll B, Korolchuk VI (2017) Dysregulation of mTORC1/autophagy axis in senescence. Aging 9(8):1851–1852
Chen JH, Ozanne SE, Hales CN (2007) Methods of cellular senescence induction using oxidative stress. Methods Mol Biol 371:179–189
Choo KB, Tai L, Hymavathee KS, Wong CY, Nguyen PN, Huang CJ, Cheong SK, Kamarul T (2014) Oxidative stress-induced premature senescence in Wharton’s jelly-derived mesenchymal stem cells. Int J Med Sci 11(11):1201–1207
Chung HY, Lee EK, Choi YJ, Kim JM, Kim DH, Zou Y, Kim CH, Lee J, Kim HS, Kim ND, Jung JH, Yu BP (2011) Molecular inflammation as an underlying mechanism of the aging process and age-related diseases. J Dental Res 90:830–840
Davalli P, Mitic T, Caporali A, Lauriola A, D’Arca D (2016) ROS, cell senescence, and novel molecular mechanisms in aging and age-related diseases. Oxid Med Cell Longev 2016:3565127
De la Fuente M, Miquel J (2009) An update of the oxidation-inflammation theory of aging: the involvement of the immune system in oxi-inflamm-aging. Curr Pharm Des 15(26):3003–3026
Green DR, Reed JC (1998) Mitochondria and apoptosis. Science 281:1309–1312
Han DW, Lee MH, Kim B, Lee JJ, Hyon SH, Park JC (2012) Preventive effects of epigallocatechin-3-O-gallate against replicative senescence associated with p53 acetylation in human dermal fibroblasts. Oxid Med Cell Longev 2012:850684
Johnson SC, Rabinovitch PS, Kaeberlein M (2013) mTOR is a key modulator of ageing and age-related disease. Nature 493(7432):338–345
Kennedy BK, Lamming DW (2016) The mechanistic target of rapamycin: the grand conductor of metabolism and aging. Cell Metab 23(6):990–1003
Kirkland JL, Tchkonia T (2017) Cellular senescence: a translational perspective. EBioMedicine 21:21–28
Lambert JD, Lee MJ, Lu H, Meng X, Hong JJ, Seril DN, Sturgill MG, Yang CS (2003) Epigallocatechin-3-gallate is absorbed but extensively glucuronidated following oral administration to mice. J Nutr 133(12):4172–4177
Laplante M, Sabatini DM (2012) mTOR signaling in growth control and disease. Cell 149:274–293
Mária J, Ingrid Ž (2017) Effects of bioactive compounds on senescence and components of senescence associated secretory phenotypes in vitro. Food Funct 8(7):2394–2418
Nacarelli T, Azar A, Sell C (2015) Aberrant mTOR activation in senescence and aging: a mitochondrial stress response? Exp Gerontol 68:66–70
Niu Y, Na L, Feng R, Gong L, Zhao Y, Li Q, Li Y, Sun C (2013) The phytochemical, EGCG, extends lifespan by reducing liver and kidney function damage and improving age-associated inflammation and oxidative stress in healthy rats. Aging Cell 12(6):1041–1049
Nogueira V, Park Y, Chen CC, Xu PZ, Chen ML, Tonic I, Unterman T, Hay N (2008) Akt determines replicative senescence and oxidative or oncogenic premature senescence and sensitizes cells to oxidative apoptosis. Cancer Cell 14(6):458–470
Sadowska-Bartosz I, Bartosz G (2014) Effect of antioxidants supplementation on aging and longevity. Biomed Res Int 2014:404680
Sharma R, Kapila R, Haq MR, Salingati V, Kapasiya M, Kapila S (2014) Age-associated aberrations in mouse cellular and humoral immune responses. Aging Clin Exp Res 26(4):353–362
Sharma R, Sharma A, Kumari A, Kulurkar PM, Raj R, Gulati A, Padwad YS (2017) Consumption of green tea epigallocatechin-3-gallate enhances systemic immune response, antioxidative capacity and HPA axis functions in aged male swiss albino mice. Biogerontology 18(3):367–382
Swami M (2008) Akt: a double-edged sword. Nat Rev Cancer 2008(9):76. https://doi.org/10.1038/nrc2586
Trabucco SE, Zhang H (2016) Finding Shangri-La: limiting the impact of senescence on aging. Cell Stem Cell 18(3):305–306
Verburgh K (2015) Nutrigerontology: why we need a new scientific discipline to develop diets and guidelines to reduce the risk of aging-related diseases. Aging Cell 14(1):17–24
Wiley CD, Campisi J (2016) From ancient pathways to aging cells-connecting metabolism and cellular senescence. Cell Metab 23(6):1013–1021
Zhang L, Jie G, Zhang J, Zhao B (2009) Significant longevity-extending effects of EGCG on Caenorhabditis elegans under stress. Free Radic Biol Med 46(3):414–421
Zhang J, Wang X, Vikash V, Ye Q, Wu D, Liu Y, Dong W (2016) ROS and ROS-mediated cellular signaling. Oxid Med Cell Longev 2016:4350965
Zhou L, Chen X, Liu T, Gong Y, Chen S, Pan G, Cui W, Luo ZP, Pei M, Yang H, He F (2015) Melatonin reverses H2O2-induced premature senescence in mesenchymal stem cells via the SIRT1-dependent pathway. J Pineal Res 59(2):190–205
Zhu X, Yue H, Guo X, Yang J, Liu J, Liu J, Wang R, Zhu W (2017) The preconditioning of berberine suppresses hydrogen peroxide-induced premature senescence via regulation of sirtuin 1. Oxid Med Cell Longev 2017:2391820
Zoico E, Di Francesco V, Olioso D, Fratta Pasini AM, Sepe A, Bosello O, Cinti S, Cominacini L, Zamboni M (2010) In vitro aging of 3T3-L1 mouse adipocytes leads to altered metabolism and response to inflammation. Biogerontology 1:111–122
Zwerschke W, Mazurek S, Stöckl P, Hütter E, Eigenbrodt E, Jansen-Dürr P (2003) Metabolic analysis of senescent human fibroblasts reveals a role for AMP in cellular senescence. Biochem J 376(Pt 2):403–411
Acknowledgements
Authors are grateful to the Director, CSIR-IHBT for constant encouragement and support. This work was supported by grants from Department of Science and Technology, Government of India under the INSPIRE Faculty scheme (IFA17-LSPA79) and CSIR in-house project MLP0204.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumar, R., Sharma, A., Kumari, A. et al. Epigallocatechin gallate suppresses premature senescence of preadipocytes by inhibition of PI3K/Akt/mTOR pathway and induces senescent cell death by regulation of Bax/Bcl-2 pathway. Biogerontology 20, 171–189 (2019). https://doi.org/10.1007/s10522-018-9785-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-018-9785-1