Abstract
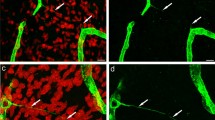
The purpose of the study was to examine the nanoscale distribution and density of the VEGFR-2 membrane receptor on the endothelial cell surface of glioma microvasculature. Immunofluorescence and atomic force microscopy combined with immunogold labeling techniques were used to characterize and determine the position of the glioma microvasculature endothelial cell surface receptor VEGFR-2. We aimed to indirectly detect the distribution of VEGFR-2 on the cell membrane at the nanoscale level and to analyze VEGFR-2 quantitatively. Immunofluorescence imaging showed a large amount of VEGFR-2 scattered across the endothelial cell surface; atomic force microscopy imaging also showed two globular structures of different sizes scattered across the endothelial cell surface. The difference between the average diameter of the small globular structure outside the cell surface (43.67 ± 5.02 nm) and that of IgG (44.61 ± 3.19 nm) was not statistically significant (P > 0.05). The three-dimensional morphologies of the small globular structure outside the cell surface and IgG were similar. The difference between the average diameter of the large globular structure outside the cell surface (74.19 ± 9.10 nm) and that of IgG–SpA-CG (74.54 ± 15.93 nm) was also not statistically significant (P > 0.05). The three-dimensional morphologies of this large globular structure outside the cell surface and IgG–SpA-CG were similar. The total density of these two globular structures within the unit area was 92 ± 19 particles μm2. No globular structures were seen on the cell surface in the control group. The large globular structure on the surface of glioma microvascular endothelial cells was categorized as a VEGFR-2–IgG–SpA-CG immune complex, whereas the small globular structure was categorized as a VEGFR-2–IgG immune complex. The positions of the globular structures were the same as the positions of the VEGFR-2 molecules. A large amount of VEGFR-2 was scattered across glioma microvascular endothelial cell surfaces; the receptor density was about 92 per square micron.






Similar content being viewed by others
References
Almqvist N, Bhatia R, Primbs G et al (2004) Elasticity and adhesion force mapping reveals real-time clustering of growth factor receptors and associated changes in local cellular rheological properties. Biophys J 86(3):1753–1762
Argyriou AA, Giannopoulou E, Kalofonos HP (2009) Angiogenesis and anti-angiogenic molecularly targeted therapies in malignant gliomas. Oncology 77(1):1–11
Cai W, Chen K, Mohamedali KA et al (2006) PET of vascular endothelial growth factor receptor expression. J Nucl Med 47(12):2048–2056
Chamberlain MC, Raizer J (2009) Antiangiogenic therapy for high-grade gliomas. CNS Neurol Disord Drug Targets 8(3):184–194
Dexiang Z, Xiaodan J, Ruxiang X et al (2008) Assessing the cytoskeletal system and its elements in C6 glioma cells and astrocytes by atomic force microscopy. Cell Mol Neurobiol 28(6):895–905
Engel A, Schoenenberger CA, Müller DJ (1997) High resolution imaging of native biological sample surfaces using scanning probe microscopy. Curr Opin Struct Biol 7(2):279–284
Lamszus K, Brockmann MA, Eckerich C et al (2005) Inhibition glioblastoma angiogenesis and invasion by combined treatments directed against vascular endothelial growth factor receptor-2, epidermal growth factor receptor, and vascular endothelial-cadherin. Clin Cancer Res 11(13):4934–4940
Li JT, Yan Q, Yu HL (2009) Expression of VEGF and NGF in gliomas of human. Sichuan Da Xue Xue Bao Yi Xue Ban 40(3):408–411
Lucio-Eterovic AK, Piao Y, de Groot JF (2003) Mechanisms of tumor cell invasion and angiogenesis in the central nervous system. Front Biosci 8:e289–e304
Norden AD, Drappatz J, Wen PY (2009) Antiangiogenic therapy for high-grade gliomas. Nat Rev Neurol 5(11):610–620
Rustem IL, Joel SB, John WW et al (2005) Multi-step fibrinogen binding to the integrin αIIbβ3 detected using force spectroscopy. Biophys J 89:2824–2834
Shibuya M (2006) Differential roles of vascular endothelial growth factor receptor-1 and receptor-2 in angiogenesis. J Biochem Mol Biol 39(5):469–478
Solis FJ, Bash R, Yodh J et al (2004) A statistical thermodynamic model applied to experimental AFM population and location data is able to quantify DNA-histone binding strength and internucleosomal interaction differences between acetylated and unacetylated nucleosomal arrays. Biophys J 87(5):3372–3387
Tanaka K, Mitsushima A, Yamagata N et al (1991) Direct visualization of colloidal gold-bound molecules and a cell-surface receptor by ultrahigh-resolution scanning electron microscopy. J Microsc 161(Pt 3):455–461
Wang FK, Liu L, Zhao DP et al (2009) Immunofluorescent staining on differentiation of mononuclear cells from rat umbilical cord blood into endothelial progenitor cells. China Bio-Beauty (4):10–15
Yu YG, Xu RX, Jiang XD et al (2005) Comparative study of the localization and conformation of immunocomplexes single molecule of NMDA receptor protein on neuron membrane by AFM. Neurosci Bull 21(2):117–128
Acknowledgments
This study was supported by grants from the Natural Science Fund of China (No. 30270491), the Funds for Key Sci-Tech Research Projects of Guangdong (No. 2008A030201019) and Guangzhou (No. 09B52120112-2009J1-C418-2, No. 2008A1-E4011-6).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, D., Zhan, S., Zhou, D. et al. A study of the Distribution and Density of the VEGFR-2 Receptor on Glioma Microvascular Endothelial Cell Membranes. Cell Mol Neurobiol 31, 687–694 (2011). https://doi.org/10.1007/s10571-011-9665-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-011-9665-6