Abstract
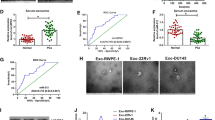
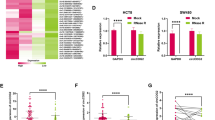
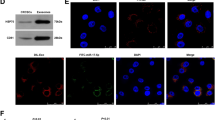
Accumulating evidence supports that exosomal RNAs are crucial in tumor microenvironment and may be used as diagnostic biomarkers for cancers. This study aimed to determine the role of exosomal circular RNA_protein tyrosine phosphatase receptor type A (circ_PTPRA) in colorectal cancer (CRC). The morphology of exosomes was identified by transmission electron microscopy (TEM), and several exosome-specific proteins were quantified by western blot. The expression of circ_PTPRA, miR-671-5p and SMAD family member 4 (SMAD4) was detected using quantitative polymerase chain reaction (qPCR). Cell cycle was assessed using flow cytometry assay. Cell proliferation was assessed by MTT assay. Radiosensitivity was observed according to colony growth and cell apoptosis rate by colony formation assay and flow cytometry assay. The protein levels of proliferation- and apoptosis-related markers and SMAD4 were measured by western blot. The predicted relationship between miR-671-5p and circ_PTPRA or SMAD4 was verified by dual-luciferase reporter assay. Animal study was performed to investigate the role of exosomal circ_PTPRA in vivo. Circ_PTPRA expression was declined in serumal exosomes from CRC patients and CRC cell lines. Exosomal circ_PTPRA induced CRC cell cycle arrest and inhibited cell proliferation. Besides, exosomal circ_PTPRA promoted radiosensitivity of CRC cells, leading to inhibitory colony formation and increased apoptotic rate. In mechanism, circ_PTPRA functioned as a competing endogenous RNA (ceRNA) to increasing SMAD4 level by binding to miR-671-5p. Rescue experiments concluded that circ_PTPRA inhibited CRC growth and radioresistance by decreasing miR-671-5p expression, and miR-671-5p inhibition also inhibited CRC growth and radioresistance by enriching SMAD4 expression. Moreover, exosomal circ_PTPRA blocked tumor growth in vivo. Exosomal circ_PTPRA enhanced CRC cell radiosensitivity and inhibited CRC malignant development partially by regulating the miR-671-5p/SMAD4 pathway, hinting that exosomal circ_PTPRA might be used as a potential predicted and therapeutic target for CRC.







Similar content being viewed by others
Data availability
Data analyzed for this study will be available on a reasonable request.
References
Araghi M, Soerjomataram I, Jenkins M et al (2019) Global trends in colorectal cancer mortality: projections to the year 2035. Int J Cancer 144:2992–3000
Bai S, Wu Y, Yan Y et al (2020) Construct a circRNA/miRNA/mRNA regulatory network to explore potential pathogenesis and therapy options of clear cell renal cell carcinoma. Sci Rep 10:13659
Barbagallo D, Condorelli A, Ragusa M et al (2016) Dysregulated miR-671-5p / CDR1-AS / CDR1 / VSNL1 axis is involved in glioblastoma multiforme. Oncotarget 7:4746–4759
Beshara A, Ahoroni M, Comanester D et al (2020) Association between time to colonoscopy after a positive guaiac fecal test result and risk of colorectal cancer and advanced stage disease at diagnosis. Int J Cancer 146:1532–1540
Cui X, Wang J, Guo Z et al (2018) Emerging function and potential diagnostic value of circular RNAs in cancer. Mol Cancer 17:123
Fanale D, Taverna S, Russo A, Bazan V (2018) Circular RNA in exosomes. Adv Exp Med Biol 1087:109–117
Guan Y, Cao Z, Du J, Liu T, Wang T (2020) Circular RNA circPITX1 knockdown inhibits glycolysis to enhance radiosensitivity of glioma cells by miR-329-3p/NEK2 axis. Cancer Cell Int 20:80
Guo W, Gao Y, Li N et al (2017) Exosomes: new players in cancer (review). Oncol Rep 38:665–675
Haney MJ, Klyachko NL, Zhao Y et al (2015) Exosomes as drug delivery vehicles for Parkinson’s disease therapy. J Control Release 207:18–30
He Q, Huang L, Yan D et al (2019) CircPTPRA acts as a tumor suppressor in bladder cancer by sponging miR-636 and upregulating KLF9. Aging (albany NY) 11:11314–11328
Hong X, Liu N, Liang Y et al (2020) Circular RNA CRIM1 functions as a ceRNA to promote nasopharyngeal carcinoma metastasis and docetaxel chemoresistance through upregulating FOXQ1. Mol Cancer 19:33
Itatani Y, Kawada K, Fujishita T et al (2013) Loss of SMAD4 from colorectal cancer cells promotes CCL15 expression to recruit CCR1+ myeloid cells and facilitate liver metastasis. Gastroenterology 145:1064–1075
Jiang L, Gu Y, Du Y, Liu J (2019) Exosomes: diagnostic biomarkers and therapeutic delivery vehicles for cancer. Mol Pharm 16:3333–3349
Jin W, Shi J, Liu M (2019) Overexpression of miR-671-5p indicates a poor prognosis in colon cancer and accelerates proliferation, migration, and invasion of colon cancer cells. Onco Targets Ther 12:6865–6873
Kim KM, Abdelmohsen K, Mustapic M, Kapogiannis D, Gorospe M (2017) RNA in extracellular vesicles. Wiley Interdiscip Rev RNA 8:1413
Kuipers EJ, Grady WM, Lieberman D et al (2015) Colorectal Cancer. Nat Rev Dis Primers 1:15065
Li X, Diao H (2019) Circular RNA circ_0001946 acts as a competing endogenous RNA to inhibit glioblastoma progression by modulating miR-671-5p and CDR1. J Cell Physiol 234:13807–13819
Li X, Nie C, Tian B et al (2019) miR-671-5p Blocks The Progression Of Human Esophageal Squamous Cell Carcinoma By Suppressing FGFR2. Int J Biol Sci 15:1892–1904
Li Y, Zheng F, Xiao X et al (2017) CircHIPK3 sponges miR-558 to suppress heparanase expression in bladder cancer cells. EMBO Rep 18:1646–1659
Liang L, Zhang L, Zhang J, Bai S, Fu H (2020) Identification of circRNA-miRNA-mRNA networks for exploring the fundamental mechanism in lung adenocarcinoma. Onco Targets Ther 13:2945–2955
Lim W, Kim HS (2019) Exosomes as therapeutic vehicles for cancer. Tissue Eng Regen Med 16:213–223
Liu L, Nie J, Chen L et al (2013) The oncogenic role of microRNA-130a/301a/454 in human colorectal cancer via targeting Smad4 expression. PLoS ONE 8:55532
Liu Y, Xu Y, Xiao F et al (2020) Comprehensive analysis of a circRNA-miRNA-mRNA network to reveal potential inflammation-related targets for gastric adenocarcinoma. Mediators Inflamm 2020:9435608
Qiu T, Wang K, Li X, Jin J (2018) miR-671-5p inhibits gastric cancer cell proliferation and promotes cell apoptosis by targeting URGCP. Exp Ther Med 16:4753–4758
Shang A, Gu C, Wang W et al (2020) Exosomal circPACRGL promotes progression of colorectal cancer via the miR-142-3p/miR-506-3p- TGF-beta1 axis. Mol Cancer 19:117
Siegel RL, Miller KD, Jemal A (2019) Cancer statistics, 2019. CA Cancer J Clin 69:7–34
Voorneveld PW, Kodach LL, Jacobs RJ et al (2014) Loss of SMAD4 alters BMP signaling to promote colorectal cancer cell metastasis via activation of Rho and ROCK. Gastroenterology 147:196–208
Wei S, Zheng Y, Jiang Y et al (2019) The circRNA circPTPRA suppresses epithelial-mesenchymal transitioning and metastasis of NSCLC cells by sponging miR-96-5p. EBioMedicine 44:182–193
Wu QB, Sheng X, Zhang N, Yang MW, Wang F (2018) Role of microRNAs in the resistance of colorectal cancer to chemoradiotherapy. Mol Clin Oncol 8:523–527
Acknowledgements
None.
Funding
There is no funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial conflicts of interest.
Ethical approval
All the procedures involving human subjects were compliant with the ethical guideline of the Declaration of Helsinki and were allowed by the Ethics Committee of Hunan Cancer Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, Y., Yang, N. & Jiang, J. Exosomal circ_PTPRA inhibits tumorigenesis and promotes radiosensitivity in colorectal cancer by enriching the level of SMAD4 via competitively binding to miR-671-5p. Cytotechnology 74, 51–64 (2022). https://doi.org/10.1007/s10616-021-00506-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-021-00506-y