Abstract
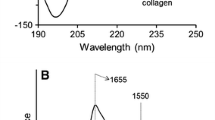
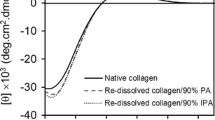
The influence of hydroxymethyl chain length of the solvents on collagen was established with conformational stability and thermal stability. Thermal stability of monomeric collagen and RTT fibres (rat tail tendon) treated with methanol, ethylene glycol (EG) and glycerol were reported using the melting temperature for helix-coil transition and the peak temperature for collagen-gelatin transition. Both melting temperature and peak temperature increases as the hydroxymethyl chain length increases. Conformational stability of collagen solution treated with lower and higher concentrations of methanol, ethylene glycol and glycerol indicates that aggregation of collagen molecule is more at higher concentrations of these solvents. The concentration dependence is greater for the increased number of OH groups. Since protein aggregation is associated with neuro degenerative diseases, aggregation of collagen molecule in the presence of solvents is of great importance for biomedical application.
Similar content being viewed by others
References
S. Vesentini, A. Redaelli and F. M. Montivecchi, J. Biomech., 38 (2005) 433.
B. Brodsky and N. K. Shah, FASEB. J., 9 (1995) 1537.
G. N. Ramachandran and G. Kartha, Nature, 174 (1954) 269.
J. Bella, M. Eaton, B. Brodsky and H. M. Berman, Science, 266 (1994) 75.
R. Z. Kramer, L. Vitagliano, J. Bella, R. Berisio, L. Mazzarella, B. Brodsky, A. Zagari and H. M. Berman, J. Mol. Biol., 280 (1998) 623.
R. Z. Kramer, J. Bella, B. Brodsky and H. M. Berman, J. Mol. Biol., 311 (2001) 131.
T. V. Burjanadze and A. Veis, Connect. Tiss. Res., 36 (1997) 347.
R. Usha and T. Ramasami, J. Polym. Sci. B. Polym. Phys., 37 (1999) 1397.
R. Usha and T. Ramasami, Thermochim. Acta, 338 (1999) 17.
R. Usha and T. Ramasami, Thermochim. Acta, 409 (2004) 201.
G. J. Hart, A. E. Russell and D. R. Cooper, Biochem. J., 123 (1971) 599.
K. Gekko and S. N. Timasheff, Biochem., 20 (1981) 4677.
Y. Nazaki and C. Tanford, J. Biol. Chem., 240 (1965) 3568.
C. A. Miles and T. V. Burjanadze, Biophysical J., 80 (2001) 1480.
C. A. Miles and M. Gbelishvilli, Biophys. J., 76 (1999) 3243.
G. Chandrakasan, D. A. Torchia and K. A. Piez, J. Biol. Chem., 251 (1976) 6060.
J. F. Woessner Jr., Arch. Biochem. Biophys., 93 (1961) 440.
K. A. Piez and M. R. Sherman, Biochemistry, 9 (1970) 4129.
M. G. Venugopal, A. W. Ramshaw, E. Brasswell, D. Zhu and B. Brodsky, Biochemistry, 33 (1994) 7948.
B. Saccà, C. Renner and L. Moroder, J. Mol. Biol., 324 (2002) 309.
S. K. Holmgran, K. M. Taylor, L. E. Bretscher and R. T. Raines, Nature, 392 (1998) 666.
A. V. Periskov, J. A. M. Ramshaw, A. Krikpatrick and B. Brodsky, Biochemistry, 9 (2000)14960.
R. Gayatri, R. Rajaram and T. Ramasami, Biochim. Biophys. Acta, 1524 (2000) 228.
R. Usha and T. Ramasami, Colloids Surf. B. Biointerface, 41 (2005) 21.
V. Vetri and V. Militello, Biophys. Chem.,113 (2005) 83.
N. Kuznetsova, S. L. Chi and S. Leikin, Biochemistry, 37 (1998) 11888.
E. I. Tiktopulo and A. V. Kajava, Biochemistry, 37 (1998) 8147.
P. F. Davison and M. P. Drake, Biochemistry, 5 (1966) 313.
R. J. Davidson and D. R. Cooper, Nature, 217 (1968) 168.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Usha, R., Ramasami, T. Role of solvents in stability of collagen. J Therm Anal Calorim 93, 541–545 (2008). https://doi.org/10.1007/s10973-006-7887-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-006-7887-5