Abstract
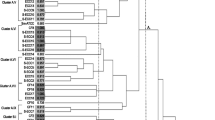
The aim of the study was to identify the number and distribution of genotypes of Streptococcus mutans (S. mutans) and Streptococcus sobrinus (S. sobrinus) isolated from caries-free and caries-active subjects. Non-stimulated saliva, buccal smooth surface of the right upper teeth, fissures of sound occlusal surface, and carious surface were sampled from 7 caries-free and 7 caries-active (DMFT ≥ 6) students aged 22–24 years. S. mutans and S. sobrinus were isolated using Chelex-100 and primarily identified by colony morphology and biochemical characteristics. The isolates of S. mutans were genotyped using arbitrarily primed polymerase chain reaction. A total of 516 isolates of S. mutans were genotyped from 47 sites in 14 students, and 44 different genotypes were determined. All of the caries-free individuals harbored S. mutans but not S. sobrinus, although individuals 3 and 7 had no S. mutans in their saliva. The CFU value of S. mutans on carious surfaces was the highest, and values in saliva, fissures, and occlusal surfaces were higher in caries-active individuals than in caries-free individuals. We detected 28 genotypes of S. mutans in caries-free individuals, each of who carried more than 3 genotypes. However, we found only 16 genotypes of S. mutans in caries-active individuals, each of who carried no more than 3 genotypes. More genotypes are harbored in the saliva, fissures, and smooth surfaces of caries-free individuals than of caries-active individuals. The proportion of samples positive for S. mutans and S. sobrinus was significantly higher in caries-active individuals than in caries-free individuals, and the presence of these species is a risk factor for high DMFT in dental caries. Isolates of S. mutans exist that have apparent genetic diversity. The genotypes of isolates might relate to differences in caries susceptibility.
Similar content being viewed by others
References
Whiley RA, Beighton D (1998) Current classification of the oral streptococci. Oral Microbiol Immunol 13:195–216
Beighton D (2005) The complex oral microflora of high-risk individuals and groups and its role in the caries process. Community Dent Oral Epidemiol 33:248–255
Bratthall D, Hoszek A, Zhao X (1996) Evaluation of a simplified method for site-specific determination of mutans streptococci levels. Swed Dent J 20:215–220
Mattos-Graner RO, Napimoga MH, Fukushima K, Duncan MJ, Smith DJ (2004) Comparative analysis of Gtf isozyme production and diversity in isolates of Streptococcus mutans with different biofilm growth phenotypes. J Clin Microbiol 42:4586–4592
Napimoga MH, Kamiya RU, Rosa RT, Rosa EA, Höfling JF, Mattos-Graner RO, Gonçalves RB (2004) Genotypic diversity and virulence traits of Streptococcus mutans in caries-free and caries-active individuals. J Med Microbiol 53:697–703
Redmo Emanuelsson I, Thornqvist E (2000) Genotypes of mutans streptococci tend to persist in their host for several years. Caries Res 34:133–139
Kreulen CM, De Soet HJ, Hogeveen R, Veerkamp JS (1997) Streptococcus mutans in children using nursing bottles. ASDC J Dent Child 64:107–111
Alaluusua S, Takei T, Ooshima T, Hamada S (1991) Mutacin activity of strains isolated from children with varying levels of mutans streptococci and caries. Arch Oral Biol 36:251–255
Longo PL, Mattos-Graner RO, Mayer MPA (2003) Determination of mutacin activity and detection of mutA genes in Streptococcus mutans genotypes from caries-free and caries-active children. Oral Microbiol Immunol 18:144–149
Schaeken MJ, van der Hoeven JS, Franken HC (1986) Comparative recovery of Streptococcus mutans on five isolation media, including a new simple selective medium. J Dent Res 65:906–908
Shklair IL, Keene HJ (1974) A biochemical scheme for the separation of the five varieties of Streptococcus mutans. Arch Oral Biol 19:1079–1081
Giraffa G, Rossetti L, Neviani E (2000) An evaluation of chelex-based DNA purification protocols for the typing of lactic acid bacteria. J Microbiol Methods 42:175–184
Huang X, Liu T, Chen G (2001) Typing of Streptococcus mutans (serotype C) by arbitrarily primed polymerase chain reaction. Zhonghua Kou Qiang Yi Xue Za Zhi 36:281–284
Liu J, Bian Z, Fan M, He H, Nie M, Fan B, Peng B, Chen Z (2004) Typing of mutans streptococci by arbitrarily primed PCR in patients undergoing orthodontic treatment. Caries Res 38:523–529
Redmo Emanuelsson IM, Carlsson P, Hamberg K, Bratthall D (2003) Tracing genotypes of mutans streptococci on tooth sites by random amplified polymorphic DNA (RAPD) analysis. Oral Microbiol Immunol 18:24–29
Berkowitz RJ (2006) Mutans streptococci acquisition and transmission. Pediatr Dent 28:106–109
Seki M, Yamashita Y, Shibata Y, Torigoe H, Tsuda H, Maeno M (2006) Effect of mixed mutans streptococci colonization on caries development. Oral Microbiol Immunol 21:47–52
Powell LV (1998) Caries risk assessment: relevance to the practitioner. J Am Dent Assoc 129:349–352
Mattos-Graner RO, Smith DJ, King WF, Mayer MP (2000) Water-insoluble glucan synthesis by mutans streptococcal strains correlates with caries incidence in 12- to 30-month-old children. J Dent Res 79:1371–1377
Nascimento MM, Hofinig FJ, Gonealves RB (2004) Streptococcus mutans genotypes isolated from root and coronal caries. Caries Res 38:454–463
Kulkarni GV, Chan KH, Sandham HJ (1989) An investigation into the use of restriction endonuclease analysis for the study of transmission of mutans streptococci. J Dent Res 68:1155–1161
Saarela M, Alaluusua S, Takei T, Asikainen S (1993) Genetic diversity within isolates of mutans streptococci recognized by an rRNA gene probe. J Clin Microbiol 31:584–587
Kozai K, Nakayama R, Tedjosasongko U, Kuwahara S, Suzuki J, Okada M, Nagasaka N (1999) Intrafamilial distribution of mutans streptococci in Japanese families and possibility of father-to-child transmission. Microbiol Immunol 43:99–106
Welsh J, McClelland M (1990) Fingerprinting genomes using PCR with arbitrary primers. Nucleic Acids Res 18:7213–7218
James V, Thearith K, James RL (1991) Distribution of repetitive DNA sequences in eubacteria and application to fingerprinting of bacterial genomes. Nucleic Acids Res 19:6823–6831
Aas JA, Griffen AL, Dardis SR, Lee AM, Olsen I, Dewhirst FE, Leys EJ, Paster BJ (2008) Bacteria of dental caries in primary and permanent teeth in children and young adults. J Clin Microbiol 46:1407–1417
Li Y, Ge Y, Saxena D, Caufield PW (2007) Genetic profiling of the oral microbiota associated with severe early-childhood caries. J Clin Microbiol 45:81–87
Okada M, Soda Y, Hayashi F, Doi T, Suzuki J, Miura K, Kozai K (2002) PCR detection of Streptococcus mutans and S. sobrinus in dental plaque samples from Japanese pre-school children. J Med Microbiol 51:443–447
Palmer RJ Jr, Diaz PI, Kolenbrander PE (2006) Rapid Succession within the Veillonella population of a developing human oral biofilm in situ. J Bacteriol 188:4117–41124
Becker MR, Paster BJ, Leys EJ, Moeschberger ML, Kenyon SG, Galvin JL, Boches SK, Dewhirst FE, Griffen AL (2002) Molecular analysis of bacterial species associated with childhood caries. J Clin Microbiol 40:1001–1009
Conflict of interest
The authors have no commercial, proprietary, or financial interest in the products or companies described in this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, Q., Yu, M., Min, Z. et al. AP-PCR detection of Streptococcus mutans and Streptococcus sobrinus in caries-free and caries-active subjects. Mol Cell Biochem 365, 159–164 (2012). https://doi.org/10.1007/s11010-012-1255-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1255-5