Abstract
Background
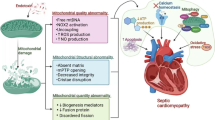
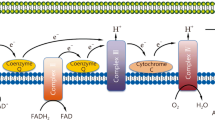
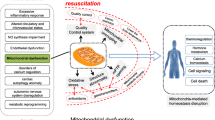
Sepsis-induced myocardial dysfunction is associated with worse clinical outcomes and high mortality, but no effective therapeutic intervention has been explored, reinforcing the urgent need to develop innovative strategies. Mitochondrial dysfunction underlies the pathogenesis of sepsis-induced myocardial dysfunction. Herein, we assessed the effect of mitochondrial transplantation on sepsis-induced myocardial dysfunction in a rat model of cecal ligation and puncture (CLP)-induced sepsis.
Methods
Male Wistar rats (n = 80, 12 weeks old, 250-300 g) were divided into groups with/without CLP-induced sepsis receiving mitochondrial transplantation in single or two repetitive injections (1 h or 1 and 7 h post-CLP, respectively). Mitochondria were isolated from donor rats and injected intravenously (400 µl of mitochondrial suspension containing 7.5 × 106 mitochondria/ml of respiration buffer) in recipient groups. Twenty-four hours post-operation, LDH and cTn-I levels, mitochondrial functional endpoints, expression of mitochondrial biogenesis (SIRT-1 and PGC-1α) and fission/fusion (Drp1/Mfn1 and Mfn2) genes, and inflammatory cytokines (TNF-α, IL-1β, and IL-6) levels were evaluated. Survival was tested over 72 h post-operation.
Results
Mitotherapy significantly improved 72-hours survival (P < .05) and decreased LDH and cTn-I levels (P < .01). It also restored mitochondrial function and expression of mitochondrial biogenesis and fusion genes, and decreased the expression of mitochondrial fission gene and the levels of inflammatory cytokines (P < .05 to P < .01). Mitotherapy with repetitive injections at 1 and 7 h post-CLP provided noticeable mitoprotection in comparison with the group receiving mitotherapy at single injection.
Conclusion
Mitotherapy improved mitochondrial function, biogenesis, and dynamic associated with SIRT-1/PGC-1α network and suppressed inflammatory response in CLP-induced sepsis model, therefore, offers a promising strategy to overcome life-threatening sepsis challenge.





Similar content being viewed by others
Availability of data and materials
The datasets used during the present work are available from the corresponding authors on reasonable request.
References
Preau S et al (2021) Energetic dysfunction in sepsis: a narrative review. Ann Intensiv Care 11(1):1–21
Lin Y, Xu Y, Zhang Z (2020) Sepsis-induced myocardial dysfunction (SIMD): the pathophysiological mechanisms and therapeutic strategies targeting mitochondria. Inflammation 43(4):1184–1200
Liu Y et al (2019) SS31 ameliorates sepsis-induced heart injury by inhibiting oxidative stress and inflammation. Inflammation 42(6):2170–2180
Liang D et al (2018) Protective effects of exogenous NaHS against sepsis-induced myocardial mitochondrial injury by enhancing the PGC-1α/NRF2 pathway and mitochondrial biosynthesis in mice. Am J Translational Res 10(5):1422
Yang H, Zhang Z (2021) Sepsis-induced myocardial dysfunction: the role of mitochondrial dysfunction. Inflamm Res 70(4):379–387
Popov LD (2020) Mitochondrial biogenesis: an update. J Cell Mol Med 24(9):4892–4899
Wang J, Toan S, Zhou H (2020) Mitochondrial quality control in cardiac microvascular ischemia-reperfusion injury: new insights into the mechanisms and therapeutic potentials. Pharmacol Res 156:104771
Hernandez-Resendiz S et al (2020) Targeting mitochondrial fusion and fission proteins for cardioprotection. J Cell Mol Med 24(12):6571–6585
Habimana R et al (2020) Sepsis-induced cardiac dysfunction: a review of pathophysiology. Acute and Critical Care 35(2):57
Wu Y, Yao Y-M, Lu Z-Q (2019) Mitochondrial quality control mechanisms as potential therapeutic targets in sepsis-induced multiple organ failure. J Mol Med 97(4):451–462
Park A et al (2021) Mitochondrial transplantation as a novel therapeutic strategy for mitochondrial diseases. Int J Mol Sci 22(9):4793
van der Slikke EC et al (2021) Sepsis is associated with mitochondrial DNA damage and a reduced mitochondrial mass in the kidney of patients with sepsis-AKI. Crit Care 25(1):1–13
Ibáñez B, Villena-Gutierrez R (2021) Cardiac Mitochondrial Transplantation: The Force Awakens. American College of Cardiology Foundation Washington DC. p. 1089–1092
Kaza AK et al (2017) Myocardial rescue with autologous mitochondrial transplantation in a porcine model of ischemia/reperfusion. J Thorac Cardiovasc Surg 153(4):934–943
Doulamis IP et al (2020) Mitochondrial transplantation for myocardial protection in diabetic hearts. Eur J Cardiothorac Surg 57(5):836–845
Zhang Z et al (2019) Muscle-derived autologous mitochondrial transplantation: a novel strategy for treating cerebral ischemic injury. Behav Brain Res 356:322–331
Jabbari H et al (2020) Mitochondrial transplantation ameliorates ischemia/reperfusion-induced kidney injury in rat. Biochimica et Biophysica Acta (BBA)-Molecular basis of Disease. 1866:1658098
Moskowitzova K et al (2020) Mitochondrial transplantation enhances murine lung viability and recovery after ischemia-reperfusion injury. Am J Physiology-Lung Cell Mol Physiol 318(1):L78–L88
Ko SF et al (2020) Hepatic 31P-magnetic resonance spectroscopy identified the impact of melatonin‐pretreated mitochondria in acute liver ischaemia‐reperfusion injury. J Cell Mol Med 24(17):10088–10099
Fang S-Y et al (2021) Transplantation of viable mitochondria attenuates neurologic injury after spinal cord ischemia. J Thorac Cardiovasc Surg 161(5):e337–e347
Orfany A et al (2020) Mitochondrial transplantation ameliorates acute limb ischemia. J Vasc Surg 71(3):1014–1026
Mokhtari B et al (2022) An Overview on Mitochondrial-Based Therapies in Sepsis-Related Myocardial Dysfunction: Mitochondrial Transplantation as a Promising Approach. Canadian Journal of Infectious Diseases and Medical Microbiology, 2022
Xin C et al (2020) Irisin attenuates myocardial ischemia/reperfusion injury and improves mitochondrial function through AMPK pathway in diabetic mice. Front Pharmacol 11:565160
Ling H et al (2021) LncRNA GAS5 inhibits mir-579-3p to activate SIRT1/PGC-1α/Nrf2 signaling pathway to reduce cell pyroptosis in sepsis-associated renal injury. Am J Physiology-Cell Physiol 321(7):C117–C133
Peng K et al (2017) The interaction of mitochondrial biogenesis and fission/fusion mediated by PGC-1α regulates rotenone-induced dopaminergic neurotoxicity. Mol Neurobiol 54(5):3783–3797
Haileselassie B et al (2019) Drp1/Fis1 interaction mediates mitochondrial dysfunction in septic cardiomyopathy. J Mol Cell Cardiol 130:160–169
Zhang H, Feng Y-w, Yao Y-m (2018) Potential therapy strategy: targeting mitochondrial dysfunction in sepsis. Military Med Res 5(1):1–11
Su H et al (2020) Salvianolic acid B protects against sepsis–induced liver injury via activation of SIRT1/PGC–1α signaling. Experimental and Therapeutic Medicine 20(3):2675–2683
Stanzani G, Duchen MR, Singer M (2019) The role of mitochondria in sepsis-induced cardiomyopathy. Biochimica et Biophysica Acta (BBA)-Molecular basis of Disease. 1865:759–7734
Zhang S et al (2020) Carbon monoxide attenuates LPS-induced myocardial dysfunction in rats by regulating the mitochondrial dynamic equilibrium. Eur J Pharmacol 889:173726
Tan Y et al (2019) Irisin ameliorates septic cardiomyopathy via inhibiting DRP1-related mitochondrial fission and normalizing the JNK-LATS2 signaling pathway. Cell Stress Chaperones 24(3):595–608
Zhu X et al (2022) Verbascoside protects from LPS-induced septic cardiomyopathy via alleviating cardiac inflammation, oxidative stress and regulating mitochondrial dynamics. Ecotoxicol Environ Saf 233:113327
Lee S et al (2022) Inhibition of mitoNEET attenuates LPS-induced inflammation and oxidative stress. Cell Death Dis 13(2):1–9
Eyenga P et al (2018) Time course of liver mitochondrial function and intrinsic changes in oxidative phosphorylation in a rat model of sepsis. Intensive care medicine experimental 6(1):1–17
Wang Y et al (2020) Crocetin attenuates Sepsis-Induced Cardiac Dysfunction via Regulation of Inflammatory Response and mitochondrial function. Front Physiol 11:514
Lightowlers RN, Chrzanowska-Lightowlers ZM, Russell OM (2020) Mitochondrial transplantation—A possible therapeutic for mitochondrial dysfunction? Mitochondrial transfer is a potential cure for many diseases but proof of efficacy and safety is still lacking. EMBO Rep 21(9):e50964
Kong C, Song W, Fu T (2022) Systemic inflammatory response syndrome is triggered by mitochondrial damage. Mol Med Rep 25(4):1–8
Hwang JW et al (2021) The immune modulatory effects of mitochondrial transplantation on cecal slurry model in rat. Crit Care 25(1):1–12
Yan C et al (2020) Mitochondrial transplantation attenuates brain dysfunction in sepsis by driving microglial M2 polarization. Mol Neurobiol 57(9):3875–3890
Zhang Z et al (2021) Muscle-derived mitochondrial transplantation reduces inflammation, enhances bacterial clearance, and improves survival in Sepsis. Shock 56(1):108–118
Acknowledgements
The authors appreciate the help of Dr. Soleyman Bafadam (PhD student of Medical Physiology at Tabriz University of Medical Sciences) in animal handling.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by Drug Applied Research Center at Tabriz University of Medical Sciences, Tabriz-Iran.
Author information
Authors and Affiliations
Contributions
B.M. performed the experimental tests, gathered and analyzed the data, and wrote the manuscript. M.H. contributed to the experimental tests. R.B. and A.M. did the study design and supervised the whole project. B.M. and R.B. critically revised and finalized the manuscript. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
All experimental protocols and procedures were approved by the Institutional Animal Ethical Committee at the Faculty of Medicine of Tabriz University of Medical Sciences (Ethics approval number: IR.TBZMED.VCR.REC.1399.114).
Consent for publication
Not Applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mokhtari, B., Hamidi, M., Badalzadeh, R. et al. Mitochondrial transplantation protects against sepsis-induced myocardial dysfunction by modulating mitochondrial biogenesis and fission/fusion and inflammatory response. Mol Biol Rep 50, 2147–2158 (2023). https://doi.org/10.1007/s11033-022-08115-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-08115-4