Abstract
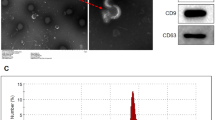
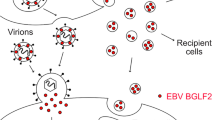
Exosomes are small secreted cellular vesicles for intercellular communications which contain proteins, mRNAs, and microRNAs (miRNAs). Recent studies have shown that exosomes play an important role in the transmission of infectious agents including hepatitis C virus, human immunodeficiency virus, and so on. However, the role of exosomes in the transfer of enterovirus 71 (EV71) between host cells remains unknown. In this study, we show that the exosomes derived from EV71-infected rhabdomyosarcoma cells contain EV71 RNA and capsid protein VP1, determined by quantitative reverse transcription-PCR (QRT-PCR) and Western blot analysis. The shedding of exosomes containing virus can establish a productive infection in human neuroblastoma cell line (SK-N-SH). A comparative analysis of neutralization by EV71-specific immunoglobulins showed different levels of neutralization of exosomes-mediated infection compared with free virus. In conclusion, exosomes from EV71-infected cells may play an important role in virus dissemination and are partially resisted to antibody neutralization. Our results suggest that there is an exosomal route of EV71 transmission infection.




Similar content being viewed by others
References
C. Thery, L. Zitvogel, S. Amigorena, Nat. Rev. Immunol. 2, 569–579 (2002)
J.S. Schorey, S. Bhatnagar, Traffic 9, 871–881 (2008)
B.T. Pan, K. Teng, C. Wu, M. Adam, R.M. Johnstone, J. Cell Biol. 101, 942–948 (1985)
R.L. Williams, S. Urbe, Nat. Rev. Mol. Cell Biol. 8, 355–368 (2007)
R.J. Simpson, S.S. Jensen, J.W.E. Lim, J.W. Lim, Proteomics 8, 4083–4099 (2008)
S.E.L. Andaloussi, I. Mager, X.O. Breakefield, M.J. Wood, Nat. Rev. Drug. Discov. 12, 347–357 (2013)
M. Simons, G. Raposo, Curr. Opin. Cell Biol. 21, 575–581 (2009)
S. Mathivanan, C.J. Fahner, G.E. Reid, R.J. Simpson, Nucleic Acids Res. 40, D1241–1244 (2012)
R. Sullivan, F. Saez, J. Girouard, G. Frenette, Blood Cells Mol. Dis. 35, 1–10 (2005)
B. Fevrier, D. Vilette, F. Archer, D. Loew, W. Faigle, M. Vidal, H. Laude, G. Raposo, Proc. Natl. Acad. Sci. USA 29, 9683–9688 (2004)
R. Valenti, V. Huber, P. Filipazzi, L. Pilla, G. Sovena, A. Villa, A. Corbelli, S. Fais, G. Parmiani, L. Rivoltini, Cancer Res. 66, 9290–9298 (2006)
J.E. Park, H.S. Tan, A. Datta, R.C. Lai, H. Zhang, W. Meng, S.K. Lim, S.K. Sze, Mol. Cell. Proteomics 9, 1085–1099 (2010)
A. Janowska-Wieczorek, M. Wysoczynski, J. Kijowski, L. Marquez-Curtis, B. Machalinski, J. Ratajczak, M.Z. Ratajczak, Int. J. Cancer 113, 752–760 (2005)
G. Camussi, M.C. Deregibus, S. Bruno, V. Cantaluppi, L. Biancone, Kidney Int. 78, 838–848 (2010)
M.A. Antonyak, B. Li, L.K. Boroughs, J.L. Johnson, J.E. Druso, K.L. Bryant, D.A. Holowka, R.A. Cerione, Proc. Natl. Acad. Sci. U.S.A. 108, 4852–4857 (2011)
L.J. Costa, N. Chen, A. Lopes, R.S. Aguiar, A. Tanuri, A. Plemenitas, B.M. Peterlin, Retrovirology 3, 33 (2006)
M. Lenassi, G. Cagney, M. Liao, T. Vaupotic, K. Bartholomeeusen, Y. Cheng, N.J. Krogan, A. Plemenitas, B.M. Peterlin, Traffic 11, 110–122 (2010)
R. Madrid, K. Janvier, D. Hitchin, J. Day, S. Coleman, C. Noviello, J. Bouchet, A. Benmerah, J. Guatelli, S. Benichou, J. Biol. Chem. 280, 5032–5044 (2005)
J. Klibi, T. Niki, A. Riedel, C. Pioche-Durieu, S. Souquere, E. Rubinstein, S. Le Moulec, J. Guigay, M. Hirashima, F. Guemira, D. Adhikary, J. Mautner, P. Busson, Blood 113, 957–1966 (2009)
F.J. Verweij, M.A. van Eijndhoven, E.S. Hopmans, T. Vendrig, T. Wurdinger, E. Cahir-McFarland, E. Kieff, D. Geerts, R. van der Kant, J. Neefjes, J.M. Middeldorp, D.M. Pegtel, EMBO J. 30, 2115–2129 (2011)
D.G. Meckes Jr, Kh Shair, A.R. Marquitz, C.-P. Kung, R.H. Edwards, N. Raab-Traub, Proc. Natl. Acad. Sci. US A 23, 20370–20375 (2010)
F.L. Cosset, M. Dreux, J. Hepatol. 60, 674–675 (2014)
F. Masciopinto, C. Giovani, S. Campagnoli, L. Galli-Stampino, P. Colombatto, M. Brunetto, T.S.B. Yen, M. Houghton, P. Pileri, S. Abrignani, Eur. J. Immunol. 34, 2834–2842 (2004)
V. Ramakrishnaiah, C. Thumann, I. Fofana, F. Habersetzer, Q. Pan, P.E. de Ruiter, R. Willemsen, J.A.A. Demmers, V. Stalin Raj, G. Jenster, J. Kwekkeboom, H.W. Tilanus, B.L. Haagmans, T.F. Baumert, L.J.W. van der Laan, L.J. van der Laan, Proc. Natl. Acad. Sci. USA 110, 13109–13113 (2013)
S.M. Robinson, G. Tsueng, J. Sin, V. Mangale, S. Rahawi, L.L. McIntyre, W. Williams, N. Kha, C. Cruz, B.M. Hancock, D.P. Nguyen, M.R. Sayen, B.J. Hilton, K.S. Doran, A.M. Segall, R. Wolkowicz, C.T. Cornell, J.L. Whitton, R.A. Gottlieb, R. Feuer, PLoS Pathog. 10, e1004045 (2014)
Nj Schmidt, E.H. Lennette, H.H. Ho, J. Infect. Dis. 129, 304–309 (1974)
J.M. Bible, P. Pantelidis, P.K.S. Chan, C.Y.W. Tong, C.Y. Tong, Rev. Med. Virol. 17, 371–379 (2007)
P.C. McMinn, FEMS Microbiol. Rev. 26, 91–107 (2002)
L.-X. Mao, Chin J Clin Lab Sci 28, 327–329 (2010)
I. Wada, D. Rindress, P.H. Cameron, W.J. Ou, J.J. Doherty 2nd, D. Louvard, A.W. Bell, D. Dignard, D.Y. Thomas, J.J. Bergeron, J. Biol. Chem. 266, 19599–19610 (1991)
P. Gastaminza, K.A. Dryden, B. Boyd, M.R. Wood, M. Law, M. Yeager, F.V. Chisari, J. Virol. 84, 10999–11009 (2010)
Acknowledgments
This study was supported by Health Science and Technology Support Program of Zhenjiang (Grant No. SHW2015014), Jiangsu Province “333” Project.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Edited by Paul Schnitzler.
Lingxiang Mao and Jing Wu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Mao, L., Wu, J., Shen, L. et al. Enterovirus 71 transmission by exosomes establishes a productive infection in human neuroblastoma cells. Virus Genes 52, 189–194 (2016). https://doi.org/10.1007/s11262-016-1292-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-016-1292-3