Abstract
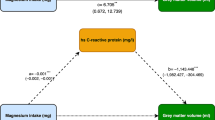
Mediation analysis aims to discover the role of intermediate variables from exposure to disease. The current study was performed to evaluate how vitamin D mediates the association between acrylamide hemoglobin biomarkers and obesity. Data were collected on 10,377 adults participating in the National Health and Nutrition Examination Survey (NHANES) 2003–2006 and 2013–2014 aged ≥ 18 years. Obesity was assessed through body mass index and abdominal circumference measurements. Generalized linear and restricted cubic spline (RCS) regression were used to estimate the association between vitamin D and acrylamide hemoglobin biomarkers, and the mediation effect of vitamin D was also discussed. After adjusting for potentially confounding factors, vitamin D had strong negative associations with serum concentrations of acrylamide hemoglobin adducts (HbAA, HbGA, and HbAA + HbGA). The RCS plots demonstrated that vitamin D was inversely and nonlinearly associated with HbAA and HbAA + HbGA while inversely and linearly associated with HbGA, and also a striking difference when vitamin D was lower than 60 nmol/L. Mediation analysis suggested that a negative correlation between acrylamide and obesity was mediated by vitamin D. The current study is expected to offer a fresh perspective on reducing the toxicity of acrylamide.



Similar content being viewed by others
Data availability
Not applicable.
Change history
08 November 2021
A Correction to this paper has been published: https://doi.org/10.1007/s11356-021-17332-6
References
Afshin A Forouzanfar, MH Reitsma, MB Sur, P Estep, K Lee A, et al (2017) Health effects of overweight and obesity in 195 countries over 25 years. GBD 2015 Obesity Collaborators. N Engl J Med 377(1):13–27
Christakos S, Dhawan P, Verstuyf A, Verlinden L, Carmeliet G (2016) Vitamin D: metabolism, molecular mechanism of action, and pleiotropic effects. Physiol Rev 96(1):365–408
Chu P-L, Lin L-Y, Chen P-C, Su T-C, Lin C-Y (2017) Negative association between acrylamide exposure and body composition in adults: NHANES, 2003–2004. Nutr Diabetes 7(3):e246
Chu P-L, Liu H-S, Wang C, Lin C-Y (2020) Association between acrylamide exposure and sex hormones in males: NHANES, 2003–2004. PLoS One 15(6):e0234622
Curtis T, Halford N (2016) Reducing the acrylamide-forming potential of wheat. Food Energy Secur 5:153–164
Daniel RM, De Stavola BL, Cousens SN, Vansteelandt S (2015) Causal mediation analysis with multiplemediators. Biometrics 71(1):1–14
Davuljigari CB, Ekuban FA, Zong C, Fergany AAM, Morikawa K, Ichihara G (2021) Nrf2 activation attenuates acrylamide-induced neuropathy in mice. Int J Mol Sci 22(11):5995
Duarte-Salles T, von Stedingk H, Granum B, Gützkow KB, Rydberg P et al (2013) Dietary acrylamide intake during pregnancy and fetal growth-results from the Norwegian mother and child cohort study (MoBa). Environ Health Perspect 121(3):374–379
Dzik KP, Kaczor JJ (2019) Mechanisms of vitamin D on skeletal muscle function: oxidative stress, energy metabolism and anabolic state. Eur J Appl Physiol 119(4):825–839
Erkekoglu P, Baydar T (2014) Acrylamide neurotoxicity. Nutr Neurosci 17:49–57
European Food Safety Authority (2009) Results on the monitoring of acrylamide levels in food. EFSA J 7: RN-285
Ghanayem BI, Bai Re, Kissling GE, Travlos G, Hoffler U (2010) Diet-induced obesity in male mice is associated with reduced fertility and potentiation of acrylamide-induced reproductive toxicity. Biol Reprod 82(1):96–104
Ghosh S, Bouchard C (2017) Convergence between biological, behavioural and genetic determinants of obesity. Nat Rev Genet 18(12):731–748
Grant WB, Cross HS, Garland CF, Gorham ED, Moan J, Peterlik M, Porojnicu AC, Reichrath J, Zittermann A (2009) Estimated benefit of increased vitamin D status in reducing the economic burden of disease in western Europe. Prog Biophys Mol Biol 99(2–3):104–13
Gonzalez FJ (2005) Role of cytochromes P450 in chemical toxicity and oxidative stress: studies with CYP2E1. Mutat Res 569:101–110
Hogervorst J, Vesper HW, Madhloum N, Gyselaers W, Nawrot T (2021) Cord blood acrylamide levels and birth size, and interactions with genetic variants in acrylamide-metabolising genes. Environ Health 20:35
Hsu C-N, Hou C-Y, Pei-Chen Lu, Chang-Chien G-P, Lin S, Tain Y-L (2020) Association between acrylamide metabolites and cardiovascular risk in children with early stages of chronic kidney disease. Int J Mol Sci 21(16):5855
IARC (1994) Some Industrial Chemicals IARC monographs on evaluation of carcinogenic risks to humans. Volume 60. World Health Organization, International Agency for Research on Cancer; Lyon, 1994. Acrylamide, pp 389–433
Ingrid LD, Nenseth CB, Bere E, Bjørnarå HBT, Helland SH, Øverby NC et al (2017) The association between time scarcity, sociodemographic correlates and consumption of ultra-processed foods among parents in Norway: a cross-sectional study. BMC Public Health 17:447
JECFA 2011 Evaluation of Certain Food Additives and Contaminants. 72nd Report of the Joint FAO/Who Expert Committee on Food Additive. WHO Press, Geneva
Jiang G, Zhang L, Wang H, Chen Q, Wu X, Yan X, Chen Y, Xie M (2018) Protective effects of a Ganoderma atrum polysaccharide against acrylamide induced oxidative damage via a mitochondria mediated intrinsic apoptotic pathway in IEC-6 cells. Food Funct 9(2):1133–1143
Jones G, Prosser DE, Kaufmann M (2014) Cytochrome P450-mediated metabolism of vitamin D. J. Lipid Res 55(1):13–31
Lee GH, Oh KJ, Kim HR, Han HS, Lee HY, Park KG et al (2016) Effect of BI-1 on insulin resistance through regulation of CYP2E1. Sci Rep. 6:32229
Lin CY, Lee HL, Chen YC, Lien GW, Lin LY, Wen LL et al (2013) Positive association between urinary levels of 8-hydroxydeoxyguanosine and the acrylamide metabolite N-acetyl-S-(propionamide)-cysteine in adolescents and young adults. J Haz Mat 261:372–377
Lin CY, Lin LY, Chen YC, Wen LL, Chien KL, Sung FC et al (2015) Association between measurements of thyroid function and the acrylamide metabolite N-Acetyl-S-(propionamide)-cysteine in adolescents and young adults. Environ Res 136:246–252
Lin CY, Lin YC, Kuo HK, Hwang JJ, Lin JL, Chen PC et al (2009) Association among acrylamide, blood insulin, and insulin resistance in adults. Diabetes Care 32:2206–2211
Linhart K, Bartsch H, Seitz HK (2014) The role of reactive oxygen species (ROS) and cytochrome P-450 2E1 in the generation of carcinogenic etheno-DNA adducts. Redox Biol 3:56–62
Ma Q, Cai S, Jia Y, Sun X, Yi Junjie, Jiang D (2020) Effects of Hot-Water Extract from Vine Tea (Ampelopsis grossedentata) on Acrylamide Formation, Quality and Consumer Acceptability of Bread. Foods 9(3):373
Meyerhardt JA, Niedzwiecki D, Hollis D, Saltz LB, Mayer RJ, Nelson H, el at, (2008) Impact of body mass index and weight change after treatment on cancer recurrence and survival in patients with stage III colon cancer: findings from Cancer and Leukemia Group B 89803. J Clin Oncol 26:4109–4115
Miśkiewicz K, Nebesny E, Rosicka-Kaczmarek J, Żyżelewicz D, Budryn G (2018) The effects of baking conditions on acrylamide content in shortcrust cookies with added freeze-dried aqueous rosemary extract. J Food Sci Technol 55(10):4184–4196
Mojska H, Gielecińska I, Jasińska-Melon E, Winiarek J, Sawicki W (2020) Are AAMA and GAMA levels in urine after childbirth a suitable marker to assess exposure to acrylamide from passive smoking during pregnancy?—a pilot study. Int J Environ Res Public Health 17(20):7391
Mogire RM, Mutua A, Kimita W, Kamau A, Bejon P, Pettifor JM, el at, (2020) Prevalence of vitamin D deficiency in Africa: a systematic review and meta-analysis. Lancet Glob Health 8(1):e134–e142
Mottram DS, Wedzicha BL, Dodson AT (2002) Acrylamide is formed in the Maillard reaction. Nature 419(6906):448–449
Mucci LA, Wilson KM (2008) Acrylamide intake through diet and human cancer risk. J Agric Food Chem 56(15):6013–6019
Norman RE, Carpenter DO, Scott J, Brune MN, Sly PD (2013) Environmental exposures: an underrecognized contribution to noncommunicable diseases. Rev Environ Health 28(1):59–65
Nowak A, Zakłos-Szyda M, Żyżelewicz D, Koszucka A, Motyl I (2020) Acrylamide decreases cell viability, and provides oxidative stress, DNA damage, and apoptosis in human colon adenocarcinoma cell line Caco-2. Molecules 25(2):368
Palus K, Bulc M, Całka J (2020) Effect of acrylamide supplementation on the population of vasoactive intestinal peptide (VIP)-like immunoreactive neurons in the porcine small intestine. Int J Mol Sci 21(24):9691
Regnier SM, Sargis RM (2014) Adipocytes under assault: environmental disruption of adipose physiology. Biochim Biophys Acta 1842(3):520–533
Roberto CA, Swinburn B, Hawkes C, Huang TT, Costa SA, Ashe M et al (2015) Patchy progress on obesity prevention: emerging examples, entrenched barriers, and new thinking. Lancet 385(9985):2400–2409
Rifai L, Saleh Fatima A (2020) A Review on Acrylamide in Food: Occurrence, Toxicity, and Mitigation Strategies. Int J Toxicol 39(2):93–102
Risk Factor Collaborators (2017) Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. GBD 2017. Lancet. 2018 392(10159):1923–1994.
Sarocka A, Kovacova V, Omelka R, Grosskopf B, Kapusta E, Goc Z et al (2019) Single and simultaneous effects of acrylamide and ethanol on bone microstructure of mice after one remodeling cycle. BMC Pharmacol Toxicol 20:38
Sievert K, Lawrence M, Naika A, Baker P (2019) Processed foods and nutrition transition in the pacific: regional trends, patterns and food system drivers. Nutrients 11(6):1328
Vanderweele TJ, Vansteelandt S (2010) Odds ratios for mediation analysis for a dichotomous outcome. Am J Epidemiol 172(12):1339–1348
Wolk R, Somers VK (2006) Obesity-related cardiovascular disease: implications of obstructive sleep apnea. Diabetes Obes Metab 8:250–260
Wang RS, McDaniel LP, Manjanatha MG, Shelton SD, Doerge DR, Mei N (2010) Mutagenicity of acrylamide and glycidamide in the testes of big bluemice. Toxicol Sci 117:72–80
Acknowledgements
We thank all the participants in the CDC for conducting the NHANES survey and NCHS staff for providing us with consultation on the application of survey weights.
Funding
This work was supported by a National Science Foundation grant (Grant No.81770396).
Author information
Authors and Affiliations
Contributions
TY and FX designed and conceived the study proposal and drafted the manuscript; SS and SL conducted statistical analyses and analyzed the data; XT was responsible for supervision and software; YZ and HZ studied the literature; YZ and XL led the design of the study and revised the manuscript. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The CDC/NCHS Ethics Review Board approved the NHANES study and gave approval for public dissemination. The consent form was signed by all participants in the survey, and participants consented to store specimens of their blood for future research.
Consent for publication
Not applicable. There is no individual-level data in our publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible editor: Ludek Blaha.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The statement for the equal contribution of the Authors is corrected in the original published proof.
Highlights
• Reconfirmed the negative correlation between acrylamide and obesity in a large sample representative population in the USA.
• First put forward the impacts of vitamin D to mediate the associations between acrylamide and obesity.
• Exposure to acrylamide is nonlinearly and inversely associated with vitamin D, and there is a striking difference when vitamin D is lower than 60 nmol/L.
Rights and permissions
About this article
Cite this article
Yin, T., Xu, F., Shi, S. et al. Vitamin D mediates the association between acrylamide hemoglobin biomarkers and obesity. Environ Sci Pollut Res 29, 17162–17172 (2022). https://doi.org/10.1007/s11356-021-16798-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-16798-8