Abstract
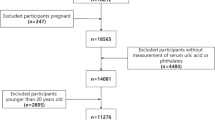
The aim of this study is to investigate the associations between phthalate exposure and UI in a nationally representative sample of US adults. Cross-sectional data from the National Health and Nutrition Examination Survey (NHANES) database was used for analysis. In total, 2,818 participants with measurements for phthalate metabolites and complete UI questionnaire data were enrolled in our study. Furthermore, seven phthalate metabolites were measured, which were obtained from urine samples and creatinine-standardized in the subsequent analyses. After dividing these phthalate metabolites into three groups, multivariable regression models were performed to evaluate the association between phthalate metabolites and UI rates. Moreover, interaction analyses and subgroup analyses stratified by gender were performed. In these seven phthalate metabolites, high level of mono-carboxynonyl phthalate (MCNP), mono-carboxyoctyl phthalate (MCOP), mono-isobutyl phthalate (MiBP), mono-n-butyl phthalate (MBP), and mono-3-carboxypropyl phthalate (MCPP) showed increased risk of UI (odds ratio (OR) = 1.52, 1.42, 1.43, 1.50, 1.51, respectively, all p value < 0.05). Trend test showed that incidence of UI increased significantly with concentration. A higher incidence of UI among participants was observed in the maximal tertile of phthalate when comparing with the lowest tertile. Subgroup analysis found that different phthalates have varying influence for different types of UI. Moreover, the analyses stratified for sex indicated that the high concentrations of MCNP and median concentrations of MCCP were associated with increase of the odds of UI in women and in men, respectively. Overall, the exposure to phthalates was positively associated with UI among US adults. Notably, different phthalates have varying influence for different types of UI, and male and female exposure to phthalate could result in the different prevalence of UI.

Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abrams P et al (2010) Fourth International Consultation on Incontinence Recommendations of the International Scientific Committee: Evaluation and treatment of urinary incontinence, pelvic organ prolapse, and fecal incontinence. Neurourol Urodyn 29(1):213–240
Abrams P et al (2018) 6th International Consultation on Incontinence. Recommendations of the International Scientific Committee: Evaluation and treatment of urinary incontinence, pelvic organ prolapse and faecal incontinence. Neurourol Urodyn 37(7):2271–2272
Adamiak-Godlewska A et al (2018) Stress urinary incontinent women, the influence of age and hormonal status on estrogen receptor alpha and beta gene expression and protein immunoexpression in paraurethral tissues. J Physiol Pharmacol 69(1):53–59
Albro PW et al (1982) Pharmacokinetics, interactions with macromolecules and species differences in metabolism of DEHP. Environ Health Perspect 45:19–25
Aoki Y et al (2017) Urinary incontinence in women. Nat Rev Dis Primers 3:17042
Barr DB et al (2003) Assessing human exposure to phthalates using monoesters and their oxidized metabolites as biomarkers. Environ Health Perspect 111(9):1148–1151
Bart S et al (2008) Stress urinary incontinence and obesity. Prog Urol 18(8):493–498
Botelho GG et al (2009) Reproductive effects of di(2-ethylhexyl)phthalate in immature male rats and its relation to cholesterol, testosterone, and thyroxin levels. Arch Environ Contam Toxicol 57(4):777–784
Buser MC, Murray HE, Scinicariello F (2014) Age and sex differences in childhood and adulthood obesity association with phthalates: analyses of NHANES 2007–2010. Int J Hyg Environ Health 217(6):687–694
Farrington N et al (2015) Exploring the role of practical nursing wisdom in the care of patients with urinary problems at the end of life: a qualitative interview study. J Clin Nurs 24(19–20):2745–2756
Ferguson KK, Loch-Caruso R, Meeker JD (2011) Urinary phthalate metabolites in relation to biomarkers of inflammation and oxidative stress: NHANES 1999–2006. Environ Res 111(5):718–726
Gao DW, Wen ZD (2016) Phthalate esters in the environment: a critical review of their occurrence, biodegradation, and removal during wastewater treatment processes. Sci Total Environ 541:986–1001
Hakimi S et al (2020) Prevalence and risk factors of urinary/anal incontinence and pelvic organ prolapse in healthy middle-aged Iranian women. J Menopausal Med 26(1):24–28
Hannas BR et al (2011) Dose-response assessment of fetal testosterone production and gene expression levels in rat testes following in utero exposure to diethylhexyl phthalate, diisobutyl phthalate, diisoheptyl phthalate, and diisononyl phthalate. Toxicol Sci 123(1):206–216
Hauser R, Calafat AM (2005) Phthalates and human health. Occup Environ Med 62(11):806–818
Hoppin JA, Ulmer R, London SJ (2004) Phthalate exposure and pulmonary function. Environ Health Perspect 112(5):571–574
Huang PC et al (2007) Associations between urinary phthalate monoesters and thyroid hormones in pregnant women. Hum Reprod 22(10):2715–2722
Jia LX et al (2015) Mechanical stretch-induced endoplasmic reticulum stress, apoptosis and inflammation contribute to thoracic aortic aneurysm and dissection. J Pathol 236(3):373–383
Kasahara E et al (2002) Role of oxidative stress in germ cell apoptosis induced by di(2-ethylhexyl)phthalate. Biochem J 365(Pt 3):849–856
Keller DL (2010) Idiopathic urgency urinary incontinence. N Engl J Med 363(27):2672; author reply 2672
Kim MM, Kreydin EI (2018) The association of serum testosterone levels and urinary incontinence in women. J Urol 199(2):522–527
Kim KN et al (2016) Urinary phthalate metabolites and depression in an elderly population: National Health and Nutrition Examination Survey 2005–2012. Environ Res 145:61–67
Li M et al (2011) Increased transient receptor potential vanilloid type 1 (TRPV1) signaling in idiopathic overactive bladder urothelial cells. Neurourol Urodyn 30(4):606–611
Lightner DJ et al (2019) Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU Guideline Amendment 2019. J Urol 202(3):558–563
Long SE et al (2021) Urinary phthalate metabolites and alternatives and serum sex steroid hormones among pre- and postmenopausal women from NHANES, 2013–16. Sci Total Environ 769:144560
Lucas MG et al (2012) EAU guidelines on surgical treatment of urinary incontinence. Eur Urol 62(6):1118–1129
Mariana M et al (2016) The effects of phthalates in the cardiovascular and reproductive systems: a review. Environ Int 94:758–776
Markland AD et al (2011) Prevalence and trends of urinary incontinence in adults in the United States, 2001 to 2008. J Urol 186(2):589–593
Meeker JD (2010) Exposure to environmental endocrine disrupting compounds and men’s health. Maturitas 66(3):236–241
Meeker JD, Ferguson KK (2014) Urinary phthalate metabolites are associated with decreased serum testosterone in men, women, and children from NHANES 2011–2012. J Clin Endocrinol Metab 99(11):4346–4352
Meeker JD et al (2009a) Urinary phthalate metabolites in relation to preterm birth in Mexico city. Environ Health Perspect 117(10):1587–1592
Meeker JD, Sathyanarayana S, Swan SH (2009b) Phthalates and other additives in plastics: human exposure and associated health outcomes. Philos Trans R Soc Lond B Biol Sci 364(1526):2097–2113
Moser H et al (2018) Pelvic floor muscle activity during impact activities in continent and incontinent women: a systematic review. Int Urogynecol J 29(2):179–196
Nishioka J et al (2012) Di-(2-ethylhexyl) phthalate induces production of inflammatory molecules in human macrophages. Inflamm Res 61(1):69–78
Pintos-Díaz MZ et al (2019) Living with urinary incontinence: potential risks of women's health? A qualitative study on the perspectives of female patients seeking care for the first time in a specialized center. Int J Environ Res Public Health 16(19)
Qaseem A et al (2014) Nonsurgical management of urinary incontinence in women: a clinical practice guideline from the American College of Physicians. Ann Intern Med 161(6):429–440
Radke EG et al (2018) Phthalate exposure and male reproductive outcomes: a systematic review of the human epidemiological evidence. Environ Int 121(Pt 1):764–793
Rogers RG et al (2018) An international Urogynecological association (IUGA)/international continence society (ICS) joint report on the terminology for the assessment of sexual health of women with pelvic floor dysfunction. Int Urogynecol J 29(5):647–666
Rusyn I et al (2001) Phthalates rapidly increase production of reactive oxygen species in vivo: role of Kupffer cells. Mol Pharmacol 59(4):744–750
Silva MJ et al (2004) Urinary levels of seven phthalate metabolites in the U.S. population from the National Health and Nutrition Examination Survey (NHANES) 1999–2000. Environ Health Perspect 112(3):331–8.
Sun S, Liu D, Jiao Z (2016) Coffee and caffeine intake and risk of urinary incontinence: a meta-analysis of observational studies. BMC Urol 16(1):61
Swan SH (2006) Prenatal phthalate exposure and anogenital distance in male infants. Environ Health Perspect 114(2):A88–A89
Swan SH et al (2015) First trimester phthalate exposure and anogenital distance in newborns. Hum Reprod 30(4):963–972
Tähtinen RM et al (2016) Long-term impact of mode of delivery on stress urinary incontinence and urgency urinary incontinence: a systematic review and meta-analysis. Eur Urol 70(1):148–158
Trasande L et al (2013) Race/ethnicity-specific associations of urinary phthalates with childhood body mass in a nationally representative sample. Environ Health Perspect 121(4):501–506
Treister-Goltzman Y, Peleg R (2018) Urinary incontinence among Muslim women in Israel: risk factors and help-seeking behavior. Int Urogynecol J 29(4):539–546
Varshavsky JR et al (2018) Dietary sources of cumulative phthalates exposure among the U.S. general population in NHANES 2005–2014. Environ Int 115:417–429
Vetrano AM et al (2010) Inflammatory effects of phthalates in neonatal neutrophils. Pediatr Res 68(2):134–139
Wang Z et al (2020) Constitutive androstane receptor (CAR) mediates dieldrin-induced liver tumorigenesis in mouse. Arch Toxicol 94(8):2873–2884
Woodward MJ et al (2020) Phthalates and sex steroid hormones among men from NHANES, 2013–2016. J Clin Endocrinol Metab 105(4):e1225–e1234
Zhou D et al (2010) Di-n-butyl phthalate (DBP) exposure induces oxidative damage in testes of adult rats. Syst Biol Reprod Med 56(6):413–419
Zuo HX et al (2014) Di-(n-butyl)-phthalate-induced oxidative stress and depression-like behavior in mice with or without ovalbumin immunization. Biomed Environ Sci 27(4):268–280
Funding
This study was supported by grants from National Natural Science Foundation of China (82070784, 81702536), a grant from Science & Technology Department of Sichuan Province, China (2022JDRC0040) to J. A., and a grant from 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYGD18011) to H.L.
Author information
Authors and Affiliations
Contributions
J. Ai, Q. Wei, and X. Yi conceived the project and drafted the manuscript, K. Jin, X. Xiong, T. Zhang, and G. Peng searched the databases, S. Qiu, X. Zheng, D. Liao, and H. Xu analyzed data, and H. Li and L. Yang revised the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Not applicable.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xianyanling Yi, Kun Jin, and Shi Qiu contributed to this work equally.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yi, X., Jin, K., Qiu, S. et al. Phthalate exposure enhances incidence of urinary incontinence: US NHANES, 2003–2004 and 2005–2006. Environ Sci Pollut Res 29, 64692–64703 (2022). https://doi.org/10.1007/s11356-022-20307-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-20307-w