Abstract
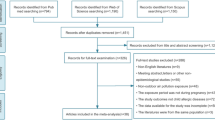
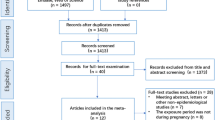
According to the “fetal origin of disease” hypothesis, air pollution exposure in pregnancy may play an important role in stimulating the early programming of asthma and allergies. However, previous studies reported inconsistent findings. The aim of this meta-analysis was to provide higher grade evidence and quantitatively analyze the link between prenatal exposure to outdoor air pollutants and childhood asthma and wheezing. Databases (Web of Science and PubMed) were extensively searched for articles published from the start of the database to September 15, 2021. Either random-effect model or fixed-effect model was used to estimate the disease-specific relative risks (RR) with the corresponding 95% confidence intervals (CIs) to estimate the association. Newcastle-Ottawa Quality Score (NOS) was used to assess the quality of studies. This study finally included 13 cohort studies, and the findings showed that NO2 and SO2 exposure during entire pregnancy was significantly associated with wheezing (RR = 1.032, 95% CI: 1.000, 1.066) and asthma (RR = 1.114, 95% CI: 1.066, 1.164), respectively. Further analyses showed that PM2.5 were positively associated with asthma in the second (RR = 1.194, 95% CI: 1.143, 1.247) and third trimester (RR = 1.050, 95% CI: 1.007, 1.094), while NO2 (RR = 1.060, 95% CI: 1.021, 1.101) and SO2 (RR = 1.067, 95% CI: 1.013, 1.123) were shown positively associated with asthma only in the second trimester. The relationship between wheezing and outdoor air pollutants was not significant in any of the pregnancy subgroups. This study suggests that prenatal exposure of outdoor air pollution may increase the asthma and wheezing risk in the offspring and that the second trimester may be a sensitive period for air pollution exposure. But the interpretation of the causal association is hampered by limited number of studies on dose response.








Similar content being viewed by others
Data availability
Not applicable.
References
Aghapour M, Ubags ND, Bruder D et al (2022) Role of air pollutants in airway epithelial barrier dysfunction in asthma and COPD. Eur Respir Rev 31:210112. https://doi.org/10.1183/16000617.0112-2021
Aguilera I, Pedersen M, Garcia-Esteban R et al (2013) Early-life exposure to outdoor air pollution and respiratory health, ear infections, and eczema in infants from the INMA study. Environ Health Perspect 121:387–392. https://doi.org/10.1289/ehp.1205281
Aguilera J, Han X, Cao S et al (2022) Increases in ambient air pollutants during pregnancy are linked to increases in methylation of IL4, IL10, and IFNgamma. Clin Epigenetics 14:40. https://doi.org/10.1186/s13148-022-01254-2
Ahmad K, Kabir E, Ormsby GM et al (2021) Are wheezing, asthma and eczema in children associated with mother’s health during pregnancy? Evidence from an Australian birth cohort. Arch Public Health 79. https://doi.org/10.1186/s13690-021-00718-w
Asher MI, Rutter CE, Bissell K et al (2021) Worldwide trends in the burden of asthma symptoms in school-aged children: Global Asthma Network Phase I cross-sectional study. Lancet 398:1569–1580. https://doi.org/10.1016/S0140-6736(21)01450-1
Bettiol A, Gelain E, Milanesio E et al (2021) The first 1000 days of life: traffic-related air pollution and development of wheezing and asthma in childhood. A systematic review of birth cohort studies. Environmental Health 20. https://doi.org/10.1186/s12940-021-00728-9
Bosonea AM, Sharpe H, Wang T et al (2020) Developments in asthma incidence and prevalence in Alberta between 1995 and 2015. Allergy, Asthma Clin Immunol 16:87. https://doi.org/10.1186/s13223-020-00485-3
Chatkin J, Correa L, Santos U (2022) External environmental pollution as a risk factor for asthma. Clin Rev Allergy Immunol 62:72–89. https://doi.org/10.1007/s12016-020-08830-5
Clemente DB, Casas M, Vilahur N et al (2016) Prenatal ambient air pollution, placental mitochondrial DNA content, and birth weight in the INMA (Spain) and ENVIRONAGE (Belgium) Birth Cohorts. Environ Health Perspect 124:659–665. https://doi.org/10.1289/ehp.1408981
Cowie CT, Garden F, Jegasothy E et al (2019) Comparison of model estimates from an intra-city land use regression model with a national satellite-LUR and a regional Bayesian Maximum Entropy model, in estimating NO2 for a birth cohort in Sydney, Australia. Environ Res 174:24–34. https://doi.org/10.1016/j.envres.2019.03.068
Deng Q, Lu C, Norback D et al (2015) Early life exposure to ambient air pollution and childhood asthma in China. Environ Res 143:83–92. https://doi.org/10.1016/j.envres.2015.09.032
Deng Q, Lu C, Li Y et al (2016) Exposure to outdoor air pollution during trimesters of pregnancy and childhood asthma, allergic rhinitis, and eczema. Environ Res 150:119–127. https://doi.org/10.1016/j.envres.2016.05.050
Esplugues A, Ballester F, Estarlich M et al (2011) Outdoor, but not indoor, nitrogen dioxide exposure is associated with persistent cough during the first year of life. Sci Total Environ 409:4667–4673. https://doi.org/10.1016/j.scitotenv.2011.08.007
Global burden of diseases (2019) GBD cause and risk summaries. https://www.thelancet.com/gbd/summaries. Accessed 3 April 2022
Guarnieri M, Balmes JR (2014) Outdoor air pollution and asthma. Lancet 383:1581–1592. https://doi.org/10.1016/S0140-6736(14)60617-6
Guo X, Li Z, Ling W et al (2018) Epidemiology of childhood asthma in mainland China (1988-2014): a meta-analysis. Allergy Asthma Proc 39:15–29. https://doi.org/10.2500/aap.2018.39.4131
Han K, Ran Z, Wang X et al (2021) Traffic-related organic and inorganic air pollution and risk of development of childhood asthma: a meta-analysis. Environ Res 194:110493. https://doi.org/10.1016/j.envres.2020.110493
Han MS, Yuan QQ, Fahad S et al (2022) Dynamic evaluation of green development level of ASEAN region and its spatio-temporal patterns. J Clean Prod 362:132402. https://doi.org/10.1016/j.jclepro.2022.132402
Hazlehurst MF, Carroll KN, Loftus CT et al (2021) Maternal exposure to PM2.5 during pregnancy and asthma risk in early childhood: consideration of phases of fetal lung development. Environ Epidemiol 5:e130. https://doi.org/10.1097/ee9.0000000000000130
He BT, Huang JV, Kwok MK et al (2019) The association of early-life exposure to air pollution with lung function at similar to 17.5 years in the “Children of 1997” Hong Kong Chinese Birth Cohort. Environ Int 123:444–450. https://doi.org/10.1016/j.envint.2018.11.073
Herriges M, Morrisey EE (2014) Lung development: orchestrating the generation and regeneration of a complex organ. Development 141:502–513. https://doi.org/10.1242/dev.098186
Hu LH, Huang BQ, Bai SY et al (2021) SO2 derivatives induce dysfunction in human trophoblasts via inhibiting ROS/IL-6/STAT3 pathway. Ecotoxicol Environ Saf 210:111872. https://doi.org/10.1016/j.ecoenv.2020.111872
James SL, Abate D, Abate KH et al (2018) Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392:1789–1858. https://doi.org/10.1016/S0140-6736(18)32279-7
Ju L, Li C, Yang M et al (2021) Maternal air pollution exposure increases the risk of preterm birth: evidence from the meta-analysis of cohort studies. Environ Res 202:111654. https://doi.org/10.1016/j.envres.2021.111654
Kelley GA, Kelley KS (2012) Statistical models for meta-analysis: a brief tutorial. World J Methodol 2:27–32. https://doi.org/10.5662/wjm.v2.i4.27
Kingsley SL, Deyssenroth MA, Kelsey KT et al (2017) Maternal residential air pollution and placental imprinted gene expression. Environ Int 108:204–211. https://doi.org/10.1016/j.envint.2017.08.022
Ko EB, Hwang KA, Choi KC (2019) Prenatal toxicity of the environmental pollutants on neuronal and cardiac development derived from embryonic stem cells. Reprod Toxicol 90:15–23. https://doi.org/10.1016/j.reprotox.2019.08.006
Korten I, Ramsey K, Latzin P (2017) Air pollution during pregnancy and lung development in the child. Paediatr Respir Rev 21:38–46. https://doi.org/10.1016/j.prrv.2016.08.008
Lavigne E, Belair MA, Rodriguez Duque D et al (2018) Effect modification of perinatal exposure to air pollution and childhood asthma incidence. Eur Respir J. https://doi.org/10.1183/13993003.01884-2017
Lavigne E, Donelle J, Hatzopoulou M et al (2019) Spatiotemporal variations in ambient ultrafine particles and the incidence of childhood asthma. Am J Respir Crit Care Med 199:1487–1495. https://doi.org/10.1164/rccm.201810-1976OC
Lee A, Leon Hsu HH, Mathilda Chiu YH et al (2018) Prenatal fine particulate exposure and early childhood asthma: effect of maternal stress and fetal sex. J Allergy Clin Immunol 141:1880–1886. https://doi.org/10.1016/j.jaci.2017.07.017
Lewis JB, Bodine JS, Gassman JR et al (2018) Transgenic up-regulation of Claudin-6 decreases fine diesel particulate matter (DPM)-induced pulmonary inflammation. Environ Sci Pollut Res 25:18179–18188. https://doi.org/10.1007/s11356-018-1985-3
Litonjua AA, Carey VJ, Laranjo N et al (2016) Effect of prenatal supplementation with vitamin D on asthma or recurrent wheezing in offspring by age 3 years the VDAART Randomized clinical trial. Jama-J Am Med Assoc 315:362–370. https://doi.org/10.1001/jama.2015.18589
Liu W, Huang C, Cai J et al (2020) Prenatal and postnatal exposures to ambient air pollutants associated with allergies and airway diseases in childhood: a retrospective observational study. Environ Int 142:105853. https://doi.org/10.1016/j.envint.2020.105853
Liu NM, Miyashita L, Maher BA et al (2021) Evidence for the presence of air pollution nanoparticles in placental tissue cells. Sci Total Environ 751:142235. https://doi.org/10.1016/j.scitotenv.2020.142235
Ma XY, Longley I, Gao J et al (2019) A site-optimised multi-scale GIS based land use regression model for simulating local scale patterns in air pollution. Sci Total Environ 685:134–149. https://doi.org/10.1016/j.scitotenv.2019.05.408
Madsen C, Haberg SE, Magnus MC et al (2017) Pregnancy exposure to air pollution and early childhood respiratory health in the Norwegian Mother and Child Cohort Study (MoBa). BMJ Open 7:e015796. https://doi.org/10.1136/bmjopen-2016-015796
Martens DS, Cox B, Janssen BG et al (2017) Prenatal air pollution and newborns’ predisposition to accelerated biological aging. JAMA Pediatr 171:1160–1167. https://doi.org/10.1001/jamapediatrics.2017.3024
Masselot P, Sera F, Schneider R et al (2022) Differential mortality risks associated with PM2.5 components: a multi-country, multi-city study. Epidemiology 33:167–175. https://doi.org/10.1097/EDE.0000000000001455
Mattiuzzi C, Lippi G (2020) Worldwide asthma epidemiology: insights from the Global Health Data Exchange database. Int Forum Allergy Rhinol 10:75–80. https://doi.org/10.1002/alr.22464
Miller MD, Marty MA (2010) Impact of environmental chemicals on lung development. Environ Health Perspect 118:1155–1164. https://doi.org/10.1289/ehp.0901856
Mortimer K, Neugebauer R, Lurmann F et al (2008) Air pollution and pulmonary function in asthmatic children - effects of prenatal and lifetime exposures. Epidemiology 19:550–557. https://doi.org/10.1097/EDE.0b013e31816a9dcb
Murphy VE, Karmaus W, Mattes J et al (2021) Exposure to Stress and air pollution from bushfires during pregnancy: could epigenetic changes explain effects on the offspring? Int J Environ Res Public Health 18. https://doi.org/10.3390/ijerph18147465
Norback D, Lu C, Zhang Y et al (2019) Onset and remission of childhood wheeze and rhinitis across China - Associations with early life indoor and outdoor air pollution. Environ Int 123:61–69. https://doi.org/10.1016/j.envint.2018.11.033
Olson LM, Radecki L, Frintner MP et al (2007) At what age can children report dependably on their asthma health status? Pediatrics 119:E93–E102. https://doi.org/10.1542/peds.2005-3211
Olsson D, Forsberg B, Braback L et al (2021) Early childhood exposure to ambient air pollution is associated with increased risk of paediatric asthma: an administrative cohort study from Stockholm, Sweden. Environ Int 155. https://doi.org/10.1016/j.envint.2021.106667
Peden DB (2021) Prenatal exposure to particulate matter air pollution: a preventable risk for childhood asthma. J Allergy Clin Immunol 148:716–718. https://doi.org/10.1016/j.jaci.2021.07.007
Pesce G, Locatelli F, Cerveri I et al (2015) Seventy years of asthma in italy: age, period and cohort effects on incidence and remission of self-reported asthma from 1940 to 2010. PLoS One 10:e0138570. https://doi.org/10.1371/journal.pone.0138570
Radhakrishnan DK, Dell SD, Guttmann A et al (2014) Trends in the age of diagnosis of childhood asthma. J Allergy Clin Immunol 134:1057–1062. e1055. https://doi.org/10.1016/j.jaci.2014.05.012
Saenen ND, Martens DS, Neven KY et al (2019) Air pollution-induced placental alterations: an interplay of oxidative stress, epigenetics, and the aging phenotype? Clin Epigenetics 11:1–14. https://doi.org/10.1186/s13148-019-0688-z
Sbihi H, Tamburic L, Koehoorn M et al (2016) Perinatal air pollution exposure and development of asthma from birth to age 10 years. Eur Respir J 47:1062–1071. https://doi.org/10.1183/13993003.00746-2015
Scholten RH, Moller P, Andersen ZJ et al (2021) Telomere length in newborns is associated with exposure to low levels of air pollution during pregnancy. Environ Int 146:106202. https://doi.org/10.1016/j.envint.2020.106202
Serebrisky D, Wiznia A (2019) Pediatric asthma: a global epidemic. Ann Global Health 85:6. https://doi.org/10.5334/aogh.2416
Shah AS, Langrish JP, Nair H et al (2013) Global association of air pollution and heart failure: a systematic review and meta-analysis. Lancet 382:1039–1048. https://doi.org/10.1016/S0140-6736(13)60898-3
Shao J, Wheeler AJ, Zosky GR et al (2019) Long-term impacts of prenatal and infant exposure to fine particulate matter on wheezing and asthma: a systematic review and meta-analysis. Environ Epidemiol 3:e042. https://doi.org/10.1097/EE9.0000000000000042
Shu W, Li ML, Li ZA et al (2020) Meta-analysis of asthma prevalence of children aged 0-14 in surveillance cities of China. Zhonghua Yu Fang Yi Xue Za Zhi 54:875–883. https://doi.org/10.3760/cma.j.cn112150-20191015-00788
Simons E, To T, Moineddin R et al (2014) Maternal second-hand smoke exposure in pregnancy is associated with childhood asthma development. J Allergy Clin Immunol Pract 2:201–U232. https://doi.org/10.1016/j.jaip.2013.11.014
Tenero L, Tezza G, Cattazzo E et al (2013) Wheezing in preschool children. Early Hum Dev 89:S13–S17. https://doi.org/10.1016/j.earlhumdev.2013.07.017
Tsamou M, Vrijens K, Madhloum N et al (2018) Air pollution-induced placental epigenetic alterations in early life: a candidate miRNA approach. Epigenetics 13:135–146. https://doi.org/10.1080/15592294.2016.1155012
Vardavas CI, Hohmann C, Patelarou E et al (2016) The independent role of prenatal and postnatal exposure to active and passive smoking on the development of early wheeze in children. Eur Respir J 48:115–124. https://doi.org/10.1183/13993003.01016-2015
von Mutius E, Smits HH (2020) Primary prevention of asthma: from risk and protective factors to targeted strategies for prevention. Lancet 396:854–866. https://doi.org/10.1016/S0140-6736(20)31861-4
Wada T, Adachi Y, Murakami S et al (2021) Maternal exposure to smoking and infant’s wheeze and asthma: Japan Environment and Children's Study. Allergol Int 70:445–451. https://doi.org/10.1016/j.alit.2021.04.008
Wells GA, Shea B, O'Connell D et al (2018) The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 3 April 2022
WHO (2018) 9 out of 10 people worldwide breathe polluted air, but more countries are taking action. https://www.who.int/zh/news/item/02-05-2018-9-out-of-10-people-worldwide-breathe-polluted-air-but-morecountries-are-taking-action. Accessed 25 July 2022
WHO (2021) Ambient (outdoor) air pollution. https://www.who.int/zh/newsroom/fact-sheets/detail/ambient-(outdoor)-air-quality-and-health. Accessed 25 July 2022
Xie M, Liu XS, Cao XP et al (2020) Trends in prevalence and incidence of chronic respiratory diseases from 1990 to 2017. Respir Res 21:49. https://doi.org/10.1186/s12931-020-1291-8
Xu X, Qin N, Yang Z et al (2021) Potential for developing independent daytime/nighttime LUR models based on short-term mobile monitoring to improve model performance. Environ Pollut 268:115951. https://doi.org/10.1016/j.envpol.2020.115951
Yan W, Wang X, Dong T et al (2020) The impact of prenatal exposure to PM2.5 on childhood asthma and wheezing: a meta-analysis of observational studies. Environ Sci Pollut Res Int 27:29280–29290. https://doi.org/10.1007/s11356-020-09014-6
Yang K, Fahad S, He H (2022) Assessing the cooking oil fume exposure impacts on Chinese women health: an influential mechanism analysis. Environ Sci Pollut Res Int. https://doi.org/10.1007/s11356-022-19368-8
Yue HF, Yan W, Ji XT et al (2017) Maternal exposure of BALB/c mice to indoor NO2 and allergic asthma syndrome in offspring at adulthood with evaluation of DNA methylation associated Th2 polarization. Environ Health Perspect 125:097011. https://doi.org/10.1289/Ehp685
Zhang YP, Li BZ, Huang C et al (2013) Ten cities cross-sectional questionnaire survey of children asthma and other allergies in China. Chin Sci Bull 58:4182–4189. https://doi.org/10.1007/s11434-013-5914-z
Zhang MZ, Chu SS, Xia YK et al (2021a) Environmental exposure during pregnancy and the risk of childhood allergic diseases. World J Pediatr 17:467–475. https://doi.org/10.1007/s12519-021-00448-7
Zhang Y, Wei J, Shi Y et al (2021b) Early-life exposure to submicron particulate air pollution in relation to asthma development in Chinese preschool children. J Allergy Clin Immunol 148:771–782 e712. https://doi.org/10.1016/j.jaci.2021.02.030
Zou KX, Ding GL, Huang HF (2019) Advances in research into gamete and embryo-fetal origins of adult diseases. Sci China-Life Sci 62:360–368. https://doi.org/10.1007/s11427-018-9427-4
Acknowledgments
We also want to thank the Platform of Environmental Exposure and Life Health Research in Anhui Medical University.
Funding
This research was supported by the Natural Science Foundation of Anhui Province (No. 2108085MH304), the National Natural Science Foundation of China (No. 81872580), and the Student’s Platform for Innovation and Entrepreneurship Training Program of China (No.202110366005).
Author information
Authors and Affiliations
Contributions
Lei Hua: Conceptualization, Methodology, software, validation, formal analysis, data curation, writing—original draft. Liangliang Ju: Conceptualization, methodology, software, validation. Hanbing Xu: Validation, data curation, writing—original draft. Changlian Li: Validation, data curation, writing—original draft. Shu Sun: Data curation, writing—original draft. Qi Zhang: Data curation, writing—original draft. Jiyu Cao: Writing (review and editing), supervision. Rui Ding: Conceptualization, methodology, writing (review and editing), supervision, project administration, funding acquisition.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Probed the effect of maternal air pollution exposure on asthma/wheezing in progeny.
• NO2 and SO2 were respectively associated with wheezing and asthma in offspring.
• Both NO2 and SO2 can increase the risk of asthma by 6% in second trimester exposure.
• PM2.5 exposure increased the risk of asthma in the second and third trimester.
Supplementary Information
ESM 1
(DOCX 2437 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hua, L., Ju, L., Xu, H. et al. Outdoor air pollution exposure and the risk of asthma and wheezing in the offspring. Environ Sci Pollut Res 30, 14165–14189 (2023). https://doi.org/10.1007/s11356-022-23094-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23094-6