Abstract
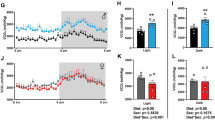
Gender-specific differences in longevity are reported across species and are mediated by mechanisms not entirely understood. In C57Bl/6 mice, commonly used in aging research, males typically outlive females. Since in these animals modest but prolonged reduction of core body (Tc) increased life span, we hypothesized that differential Tc may contribute to sex-specific longevity. Here, we compared the circadian profiles of Tc and locomotor activity (LMA) of male and female C57Bl/6 mice. Since Tc and LMA normally change with age, measurements were carried out in young (3 months) as well as in old (24 months) mice. In young females, Tc was influenced by estrous but was overall higher than in males. This difference was larger in old animals after age eliminated the variations associated with estrous. Although temperature homeostasis is regulated centrally by the sexually dimorphic hypothalamic preoptic area, these differences were uniquely dependent on the gonads. In fact, bilateral gonadectomy abolished the effects of estrous and increased resting Tc in males eliminating all sex-specific differences in Tc and LMA. These effects were only partially mimicked by hormonal replacement as Tc was affected by progesterone and to a lesser extent by estrogen but not by testosterone. Thus, gonadal-dependent modulation of Tc may be one of the physiological parameters contributing to gender-specific differences in longevity.



Similar content being viewed by others
References
Abe J, Okazawa M et al (2003) Primary cold-sensitive neurons in acutely dissociated cells of rat hypothalamus. Neurosci Lett 342(1–2):29–32
Ali SS, Xiong C et al (2006) Gender differences in free radical homeostasis during aging: shorter-lived female C57BL6 mice have increased oxidative stress. Aging Cell 5(6):565–574
Boulant JA (2006) Neuronal basis of Hammel's model for set-point thermoregulation. J Appl Physiol 100(4):1347–1354
Brown RE, Gander GW et al (1970) Estrogen and cortisone: effects on thermoregulation in the female rabbit. Proc Soc Exp Biol Med 134(1):83–86
Brown-Borg HM, Borg KE et al (1996) Dwarf mice and the ageing process. Nature 384(6604):33
Carandente F, De Matteis MA et al (1982) Circadian temperature rhythm in intact, sham operated, gonadectomized rats. Chronobiologia 9(2):211–221
Cargill SL, Carey JR et al (2003) Age of ovary determines remaining life expectancy in old ovariectomized mice. Aging Cell 2(3):185–190
Conti B, Sugama S et al (2000) Modulation of IL-18 production in the adrenal cortex following acute ACTH or chronic corticosterone treatment. Neuroimmunomodulation 8(1):1–7
Conti B, Sanchez-Alavez M et al (2006) Transgenic mice with a reduced core body temperature have an increased life span. Science 314(5800):825–828
Coomber P, Crews D et al (1997) Independent effects of incubation temperature and gonadal sex on the volume and metabolic capacity of brain nuclei in the leopard gecko (Eublepharis macularius), a lizard with temperature-dependent sex determination. J Comp Neurol 380(3):409–421
Coschigano KT, Clemmons D et al (2000) Assessment of growth parameters and life span of GHR/BP gene-disrupted mice. Endocrinology 141(7):2608–2613
Coschigano KT, Holland AN et al (2003) Deletion, but not antagonism, of the mouse growth hormone receptor results in severely decreased body weights, insulin, and insulin-like growth factor I levels and increased life span. Endocrinology 144(9):3799–3810
Crews D, Coomber P et al (1996) Brain organization in a reptile lacking sex chromosomes: effects of gonadectomy and exogenous testosterone. Horm Behav 30(4):474–486
Dalal SJ, Estep JS et al (2001) Standardization of the Whitten effect to induce susceptibility to Neisseria gonorrhoeae in female mice. Contemp Top Lab Anim Sci 40(2):13–17
Drori D, Folman Y (1986) Interactive environmental and genetic effects on longevity in the male rat: litter size, exercise, electric shocks and castration. Exp Aging Res 12(2):59–64
Flurkey K, Papaconstantinou J et al (2001) Lifespan extension and delayed immune and collagen aging in mutant mice with defects in growth hormone production. Proc Natl Acad Sci USA 98(12):6736–6741
Flurkey K, Papaconstantinou J et al (2002) The Snell dwarf mutation Pit1(dw) can increase life span in mice. Mech Ageing Dev 123(2–3):121–130
Fregly MJ, Kelleher DL et al (1979) Tolerance of estrogen-treated rats to acute cold exposure. J Appl Physiol 47(1):59–66
Gangwar PC (1982) Effects of climate on the thermoregulatory responses of male buffalo (Bubalus-bubalis) Calves supplemented with different levels of testosterone and TDN. Int J Biometeorol 26(1):73–79
Goldman JM, Murr AS et al (2007) The rodent estrous cycle: characterization of vaginal cytology and its utility in toxicological studies. Birth Defects Res B Dev Reprod Toxicol 80(2):84–97
Gordon CJ (1993) Temperature regulation in laboratory rodents. Cambridge University Press, New York
Gordon CJ (2008) Cardiac and thermal homeostasis in the aging Brown Norway rat. J Gerontol A Biol Sci Med Sci 63(12):1307–1313
Griffin JD, Saper CB et al (2001) Synaptic and morphological characteristics of temperature-sensitive and -insensitive rat hypothalamic neurones. J Physiol 537(Pt 2):521–535
Hammel H (1965) Neurons and temperature regulation. In: Yamamoto WS, Brobeck JR (eds) Physiological controls and regulations. Saunders, Philadelphia, pp 71–97
Holzenberger M, Dupont J et al (2003) IGF-1 receptor regulates lifespan and resistance to oxidative stress in mice. Nature 421(6919):182–187
Hori A, Minato K et al (1999) Warming-activated channels of warm-sensitive neurons in rat hypothalamic slices. Neurosci Lett 275(2):93–96
Kruijver FP, Swaab DF (2002) Sex hormone receptors are present in the human suprachiasmatic nucleus. Neuroendocrinology 75(5):296–305
Kurosu H, Yamamoto M et al (2005) Suppression of aging in mice by the hormone Klotho. Science 309(5742):1829–1833
Ladiges W, Van Remmen H et al (2009) Lifespan extension in genetically modified mice. Aging Cell 8(4):346–352
Liu RK, Walford RL (1966) Increased growth and life-span with lowered ambient temperature in the annual fish, Cynolebias adloffi. Nature 212:1277–1278
Marrone BL, Gentry RT et al (1976) Gonadal hormones and body temperature in rats: effects of estrous cycles, castration and steroid replacement. Physiol Behav 17(3):419–425
Mason JB, Cargill SL et al (2009) Transplantation of young ovaries to old mice increased life span in transplant recipients. J Gerontol A Biol Sci Med Sci 64(12):1207–1211
Mobbs CV, Gee DM et al (1984) Reproductive senescence in female C57BL/6J mice: ovarian impairments and neuroendocrine impairments that are partially reversible and delayable by ovariectomy. Endocrinology 115(5):1653–1662
Morrison SF, Nakamura K et al (2008) Central control of thermogenesis in mammals. Exp Physiol 93(7):773–797
Nakamura K, Morrison SF (2008) A thermosensory pathway that controls body temperature. Nat Neurosci 11(1):62–71
Nieschlag E, Nieschlag S et al (1993) Lifespan and testosterone. Nature 366(6452):215
Paul KN, Dugovic C et al (2006) Diurnal sex differences in the sleep–wake cycle of mice are dependent on gonadal function. Sleep 29(9):1211–1223
Refinetti R (2003) Metabolic heat production, heat loss and the circadian rhythm of body temperature in the rat. Exp Physiol 88(3):423–429
Reynolds MA, Ingram DK et al (1985) Relationship of body temperature stability to mortality in aging mice. Mech Ageing Dev 30(2):143–152
Rikke BA, Johnson TE (2004) Lower body temperature as a potential mechanism of life extension in homeotherms. Exp Gerontol 39(6):927–930
Rikke BA, Johnson TE (2007) Physiological genetics of dietary restriction: uncoupling the body temperature and body weight responses. Am J Physiol Regul Integr Comp Physiol 293(4):R1522–R1527
Schriner SE, Linford NJ et al (2005) Extension of murine life span by overexpression of catalase targeted to mitochondria. Science 308(5730):1909–1911
Selman C, Lingard S et al (2008) Evidence for lifespan extension and delayed age-related biomarkers in insulin receptor substrate 1 null mice. FASEB J 22(3):807–818
Silberberg SD, Magleby KL (1999) Beating the odds with Big K. Science 285(5435):1859–1860
Silva NL, Boulant JA (1986) Effects of testosterone, estradiol, and temperature on neurons in preoptic tissue slices. Am J Physiol 250(4 Pt 2):R625–R632
Sokolove PG, Bushell WN (1978) The chi square periodogram: its utility for analysis of circadian rhythms. J Theor Biol 72(1):131–160
Sugama S, Kim Y et al (2000) Tissue-specific expression of rat IL-18 gene and response to adrenocorticotropic hormone treatment. J Immunol 165(11):6287–6292
Tabarean IV, Conti B et al (2005) Electrophysiological properties and thermosensitivity of mouse preoptic and anterior hypothalamic neurons in culture. Neuroscience 135(2):433–449
Talan MI, Engel BT (1986) Temporal decrease of body temperature in middle-aged C57BL/6J mice. J Gerontol 41(1):8–12
Tower J, Arbeitman M (2009) The genetics of gender and life span. J Biol 8(4):38
Tsai CL, Matsumura K et al (1988) Effects of progesterone on thermosensitive neurons in preoptic slice preparations. Neurosci Lett 86(1):56–60
Tsai CL, Kanosue K et al (1992) Effects of estradiol treatment on responses of rat preoptic warm sensitive neurons to progesterone in vitro. Neurosci Lett 136(1):23–26
Turturro A, Witt WW et al (1999) Growth curves and survival characteristics of the animals used in the biomarkers of aging program. J Gerontol A Biol Sci Med Sci 54(11):B492–B501
van Baak MA (2008) Meal-induced activation of the sympathetic nervous system and its cardiovascular and thermogenic effects in man. Physiol Behav 94(2):178–186
Vina J, Borras C et al (2005a) Why females live longer than males: control of longevity by sex hormones. Sci Aging Knowledge Environ 23:pe17
Vina J, Borras C et al (2005b) Why females live longer than males? Importance of the upregulation of longevity-associated genes by oestrogenic compounds. FEBS Lett 579(12):2541–2545
Weinert D (1994) Lower variability in female as compared to male laboratory mice: investigations on circadian rhythms. J Exp Anim Sci 37:121–137
Weinert D (2010) Circadian temperature variation and ageing. Ageing Res Rev 9(1):51–60
Weinert D, Waterhouse J et al (2004) Changes of body temperature and thermoregulation in the course of the ovarian cycle in laboratory mice. Biol Rhythm Res 35(3):171–185
Yang JN, Tiselius C et al (2007) Sex differences in mouse heart rate and body temperature and in their regulation by adenosine A1 receptors. Acta Physiol Oxf 190(1):63–75
Acknowledgments
Supported by The Ellison Medical Foundation and NIH AG028040.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11357_2010_9164_MOESM1_ESM.pdf
Supplemental Table 1 Cosinor analysis of circadian Tc and LMA profile female and male mice during hormone replacement. DHT, 5α-dihydrotestosterone; 17beta E, 17β-estradiol; TX, treated. (PDF 49 kb)
Fig4
Supplemental Figure 1 Profile of Tc and LMA of 3-month-old female mice indicating (arrows) the time of vaginal swabs collected for histological analysis for determination of estrous phases. A profile comprising 1 h before and 1 h after sample collection is shown in the lower panel. (GIF 171 kb)
Fig5
Supplemental Figure 2 Tc and LMA profiles of castrated male and ovariectomized female treated with placebo or 5α-dihydrotestosterone (5αHDT) testosterone, progesterone, or 17β-estradiol (17βE) recorded over 24 h. (n = 6, 6; *p < 0.05). (GIF 311 kb)
Fig6
Supplemental Figure 3 Survival curves of C57Bl/6 mice maintained in the same dietary and environmental conditions used for the mice used in the present study, showing that males outlived females. Vertical lines correspond to the age investigated in this study: 3 and 24 months. The curves were extrapolated from the study previously published by us (Conti et al. 2006). (EPS 781 kb) (GIF 37 kb)
About this article
Cite this article
Sanchez-Alavez, M., Alboni, S. & Conti, B. Sex- and age-specific differences in core body temperature of C57Bl/6 mice. AGE 33, 89–99 (2011). https://doi.org/10.1007/s11357-010-9164-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-010-9164-6