Abstract
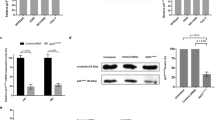
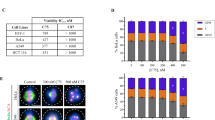
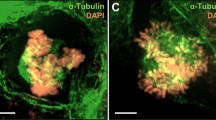
Paclitaxel is a widely used microtubule drug and cancer medicine. Here we report that by short exposure to paclitaxel at a low dose, multipolar spindles were induced in mitotic cells without centrosome amplification. Both TPX2 depletion and Aurora-A overexpression antagonized the multipolarity. Live cell imaging showed that some paclitaxel-treated cells accomplished multipolar cell division and a portion of the daughter cells went on to the next round of mitosis. The surviving cells grew into clones with varied genome content. The results indicated that an aneuploidy population could be induced by short exposure to paclitaxel at a low dose, implicating potential side effects of paclitaxel.
Similar content being viewed by others
References
Boveri T B. Zur Frage der Entstehung maligner Tumoren. Jena: Gustav Fischer, 1914
Brinkley B R. Managing the centrosome numbers game: from chaos to stability in cancer cell division. Trends Cell Biol, 2001, 11: 18–21 1:CAS:528:DC%2BD3MXnvFWn, 10.1016/S0962-8924(00)01872-9, 11146294
Saunders W S, Shuster M, Huang X, et al. Chromosomal instability and cytoskeletal defects in oral cancer cells. Proc Natl Acad Sci USA, 2000, 97: 303–308 1:CAS:528:DC%2BD3cXjvVGisA%3D%3D, 10.1073/pnas.97.1.303, 10618413
Quintyne N J, Reing J E, Hoffelder D R, et al. Spindle multipolarity is prevented by centrosomal clustering. Science, 2005, 307: 127–129 1:CAS:528:DC%2BD2MXnvFWk, 10.1126/science.1104905, 15637283
Pihan G A, Purohit A, Wallace J, et al. Centrosome defects and genetic instability in malignant tumors. Cancer Res, 1998, 58: 3974–3985 1:CAS:528:DyaK1cXlvVyqsrc%3D, 9731511
Pihan G A, Purohit A, Wallace J, et al. Centrosome defects can account for cellular and genetic changes that characterize prostate cancer progression. Cancer Res, 2001, 61: 2212–2219 1:CAS:528:DC%2BD3MXit1Ogtro%3D, 11280789
Pihan G A, Wallace J, Zhou Y, et al. Centrosome abnormalities and chromosome instability occur together in pre-invasive carcinomas. Cancer Res, 2003, 63: 1398–1404 1:CAS:528:DC%2BD3sXit1SgurY%3D, 12649205
Sluder G, Nordberg J J. The good, the bad and the ugly: the practical consequences of centrosome amplification. Curr Opin Cell Biol, 2004, 16: 49–54 1:CAS:528:DC%2BD2cXitlSms7w%3D, 10.1016/j.ceb.2003.11.006, 15037304
Doxsey S, Zimmerman W, Mikule K. Centrosome control of the cell cycle. Trends Cell Biol, 2005, 15: 303–311 1:CAS:528:DC%2BD2MXlt1Srtrk%3D, 10.1016/j.tcb.2005.04.008, 15953548
Hinchcliffe E H, Sluder G. “It takes two to tango”: understanding how centrosome duplication is regulated throughout the cell cycle. Genes Dev, 2001, 15: 1167–1181 1:CAS:528:DC%2BD3MXktVansrY%3D, 10.1101/gad.894001, 11358861
Scholey J M, Brust-Mascher I, Mogilner A. Cell division. Nature, 2003, 422: 746–752 1:CAS:528:DC%2BD3sXivFGhsbw%3D, 10.1038/nature01599, 12700768
Fu J, Bian M, Jiang Q, et al. Roles of Aurora kinases in mitosis and tumorigenesis. Mol Cancer Res, 2007, 5: 1–10 1:CAS:528:DC%2BD2sXptFeqtw%3D%3D, 10.1158/1541-7786.MCR-06-0208, 17259342
Cleveland D W. NuMA: a protein involved in nuclear structure, spindle assembly, and nuclear re-formation. Trends Cell Biol, 1995, 5: 60–64 1:CAS:528:DyaK2MXktFOisrw%3D, 10.1016/S0962-8924(00)88947-3, 14731413
Garrett S, Auer K, Compton D A, et al. hTPX2 is required for normal spindle morphology and centrosome integrity during vertebrate cell division. Curr Biol, 2002, 12: 2055–2059 1:CAS:528:DC%2BD38XpsFehurk%3D, 10.1016/S0960-9822(02)01277-0, 12477396
Karsenti E, Vernos I. The mitotic spindle: a self-made machine. Science, 2001, 294: 543–547 1:CAS:528:DC%2BD3MXnslOntbw%3D, 10.1126/science.1063488, 11641489
Paoletti A, Giocanti N, Favaudon V, et al. Pulse treatment of interphasic HeLa cells with nanomolar doses of docetaxel affects centrosome organization and leads to catastrophic exit of mitosis. J Cell Sci, 1997, 110: 2403–2415 1:CAS:528:DyaK2sXnt1Giur8%3D, 9410879
De Brabander M, Geuens G, Nuydens R, et al. Taxol induces the assembly of free microtubules in living cells and blocks the organizing capacity of the centrosomes and kinetochores. Proc Natl Acad Sci USA, 1981, 78: 5608–5612 10.1073/pnas.78.9.5608, 6117858
Jordan M A, Toso R J, Thrower D, et al. Mechanism of mitotic block and inhibition of cell proliferation by taxol at low concentrations. Proc Natl Acad Sci USA, 1993, 90: 9552–9556 1:CAS:528:DyaK3sXms1aisrg%3D, 10.1073/pnas.90.20.9552, 8105478
Jordan M A, Wendell K, Gardiner S, et al. Mitotic block induced in HeLa cells by low concentrations of paclitaxel (taxol) results in abnormal mitotic exit and apoptotic cell death. Cancer Res, 1996, 56: 816–825 1:CAS:528:DyaK28XhtVClsbo%3D, 8631019
Chen J G, Horwitz S B. Differential mitotic responses to microtubule-stabilizing and -destabilizing drugs. Cancer Res, 2002, 62: 1935–1938 1:CAS:528:DC%2BD38XivVyltLg%3D, 11929805
Ikui A E, Yang C P, Matsumoto T, et al. Low concentrations of taxol cause mitotic delay followed by premature dissociation of p55CDC from Mad2 and BubR1 and abrogation of the spindle checkpoint, leading to aneuploidy. Cell Cycle, 2005, 4: 1385–1388 1:CAS:528:DC%2BD28XjvFOmsbc%3D, 16138009
Xiong Y, Huo Y, Chen C, et al. Vascular endothelial growth factor (VEGF) receptor-2 tyrosine 1175 signaling controls VEGF-induced von Willebrand factor release from endothelial cells via phospholipase C-gamma 1- and protein kinase A-dependent pathways. J Biol Chem, 2009, 284: 23217–23224 1:CAS:528:DC%2BD1MXhtVWqtrnK, 10.1074/jbc.M109.019679, 19570985
Shi Q, and King R W. Chromosome nondisjunction yields tetraploid rather than aneuploid cells in human cell lines. Nature, 2005, 437: 1038–1042 1:CAS:528:DC%2BD2MXhtV2ktrnO, 10.1038/nature03958, 16222248
Schatz C A, Santarella R, Hoenger A, et al. Importin alpha-regulated nucleation of microtubules by TPX2. Embo J, 2003, 22: 2060–2070 1:CAS:528:DC%2BD3sXjsVKmur8%3D, 10.1093/emboj/cdg195, 12727873
Anand S, Penrhyn-Lowe S, Venkitaraman A R. AURORA-A amplification overrides the mitotic spindle assembly checkpoint, inducing resistance to Taxol. Cancer Cell, 2003, 3: 51–62 1:CAS:528:DC%2BD3sXhtFamt7Y%3D, 10.1016/S1535-6108(02)00235-0, 12559175
Kufer T A, Sillje H H, Korner R, et al. Human TPX2 is required for targeting Aurora-A kinase to the spindle. J Cell Biol, 2002, 158: 617–623 1:CAS:528:DC%2BD38XmsVSntbo%3D, 10.1083/jcb.200204155, 12177045
Marumoto T, Zhang D, Saya H. Aurora-A—a guardian of poles. Nature reviews, 2005, 5: 42–50 1:CAS:528:DC%2BD2MXlvVen, 10.1038/nrc1526, 15630414
Torres K, Horwitz S B. Mechanisms of Taxol-induced cell death are concentration dependent. Cancer Res, 1998, 58: 3620–3626 1:CAS:528:DyaK1cXlsFajt7o%3D, 9721870
Hornick J E, Bader J R, Tribble E K, et al. Live-cell analysis of mitotic spindle formation in taxol-treated cells. Cell Motil Cytoskeleton, 2008, 65: 595–613 10.1002/cm.20283, 18481305
Sotillo R, Hernando E, Diaz-Rodriguez E, et al. Mad2 overexpression promotes aneuploidy and tumorigenesis in mice. Cancer Cell, 2007, 11: 9–23 1:CAS:528:DC%2BD2sXhtFKmt74%3D, 10.1016/j.ccr.2006.10.019, 17189715
Weaver B A, Silk A D, Montagna C, et al. Aneuploidy acts both oncogenically and as a tumor suppressor. Cancer Cell, 2007, 11: 25–36 1:CAS:528:DC%2BD2sXhtFClsLs%3D, 10.1016/j.ccr.2006.12.003, 17189716
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bian, M., Fu, J., Yan, Y. et al. Short exposure to paclitaxel induces multipolar spindle formation and aneuploidy through promotion of acentrosomal pole assembly. Sci. China Life Sci. 53, 1322–1329 (2010). https://doi.org/10.1007/s11427-010-4086-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-010-4086-1