Summary
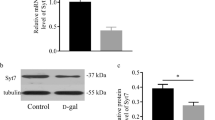
Mitochondrial DNA (mtDNA) common deletion (CD) plays a significant role in aging and age-related diseases. In this study, we used D-galactose (D-gal) to generate an animal model of aging and the involvement and causative mechanisms of mitochondrial damage in such a model were investigated. Twenty 5-week-old male Sprague-Dawley rats were randomly divided into two groups: D-gal group (n=10) and control group (n=10). The quantity of the mtDNA CD in the hippocampus was determined using a TaqMan real-time PCR assay. Transmission electron microscopy was used to observe the mitochondrial ultrastructure in the hippocampus. Western blot was used to detect the protein levels of NADPH oxidase (NOX) and uncoupling protein 2 (UCP2). We found that the level of mtDNA CD was significantly higher in the hippocampus of D-gal-induced aging rats than in control rats. In comparison with the control group, the mitochondrial ultrastructure in the hippocampus of D-gal-treated rats was damaged, and the protein levels of NOX and UCP2 were significantly increased in the hippocampus of D-gal-induced aging rats. This study demonstrated that the levels of mtDNA CD and NOX protein expression were significantly increased in the hippocampus of D-gal-induced aging rats. These findings indicate that NOX-dependent reactive oxygen species generation may contribute to D-gal-induced mitochondrial damage.
Similar content being viewed by others
References
Frisard M, Ravussin E. Energy metabolism and oxidative stress: impact on the metabolic syndrome and the aging process. Endocrine, 2006,29(1):27–32
Choi JH, Won MH. Microglia in the normally aged hippocampus. Lab Anim Res, 2011,27(3):181–187
Harman D. Aging: overview. Ann N Y Acad Sci, 2001,928:1–21
Reynolds A, Laurie C, Mosley RL, et al. Oxidative stress and the pathogenesis of neurodegenerative disorders. Int Rev Neurobiol, 2007,82:297–325
Ames A 3rd. CNS energy metabolism as related to function. Brain Res Brain Res Rev, 2000,34(1–2):42–68
Markaryan A, Nelson EG, Hinojosa R. Quantification of the mitochondrial DNA common deletion in presbycusis. Laryngoscope, 2009,119(6):1184–1189
Meissner C, Bruse P, Mohamed SA, et al. The 4977 bp deletion of mitochondrial DNA in human skeletal muscle, heart and different areas of the brain: a useful biomarker or more. Exp Gerontol, 2008,43(7):645–652
Chen B, Zhong Y, Peng W, et al. Age-related changes in the central auditory system: comparison of D-galactose-induced aging rats and naturally aging rats. Brain Res, 2010,1344:43–53
Kong WJ, Wang Y, Wang Q, et al. The relation between D-galactose injection and mitochondrial DNA 4834 bp deletion mutation. Exp Gerontol, 2006,41(6):628–634
Kong WJ, Hu YJ, Wang Q, et al. The effect of the mtDNA4834 deletion on hearing. Biochem Biophys Res Commun, 2006,344(1):425–430
Zhong Y, Hu YJ, Yang Y, et al. Contribution of common deletion to total deletion burden in mitochondrial DNA from inner ear of d-galactose-induced aging rats. Mutat Res, 2011,712(1–2):11–19
Zhong Y, Hu YJ, Chen B, et al. Mitochondrial transcription factor A overexpression and base excision repair deficiency in the inner ear of rats with D-galactose-induced aging. FEBS J, 2011,278(14):2500–2510
Chen B, Zhong Y, Peng W, et al. Increased mitochondrial DNA damage and decreased base excision repair in the auditory cortex of D-galactose-induced aging rats. Mol Biol Rep, 2011,38(6):3635–3642
Filburn CR, Edris W, Tamatani M, et al. Mitochondrial electron transport chain activities and DNA deletions in regions of the rat brain. Mech Ageing Dev, 1996,87(1): 35–46
Chen TF, Chiu MJ, Huang CT, et al. Changes in dietary folate intake differentially affect oxidised lipid and mitochondrial DNA damage in various brain regions of rats in the absence/presence of intracerebroventricularly injected amyloid beta-peptide challenge. Br J Nutr, 2011,105(9): 1294–1302
Nicolle MM, Gonzalez J, Sugaya K, et al. Signatures of hippocampal oxidative stress in aged spatial learning-impaired rodents. Neuroscience, 2001,107(3):415–431
Cottrell DA, Blakely EL, Johnson MA, et al. Cytochrome c oxidase deficient cells accumulate in the hippocampus and choroid plexus with age. Neurobiol Aging, 2001,22(2):265–272
Ho SC, Liu JH, Wu RY. Establishment of the mimetic aging effect in mice caused by D-galactose. Biogerontology, 2003,4(1):15–18
Cui X, Zuo P, Zhang Q, et al. Chronic systemic D-galactose exposure induces memory loss, neurodegen eration, and oxidative damage in mice: protective effects of R-alpha-lipoic acid. J Neurosci Res, 2006,83(8):1584–1590
Hua X, Lei M, Zhang Y, et al. Long-term D-galactose injection combined with ovariectomy serves as a new rodent model for Alzheimer’s disease. Life Sci, 2007,80(20): 1897–1905
Hsieh HM, Wu WM, Hu ML. Soy isoflavones attenuate oxidative stress and improve parameters related to aging and Alzheimer’s disease in C57BL/6J mice treated with D-galactose. Food Chem Toxicol, 2009,47(3):625–632
Lu J, Wu DM, Zheng YL, et al. Purple sweet potato color alleviates D-galactose-induced brain aging in old mice by promoting survival of neurons via PI3K pathway and inhibiting cytochrome C-mediated apoptosis. Brain Pathol, 2010,20(3):598–612
Wei H, Li L, Song Q, et al. Behavioural study of the D-galactose induced aging model in C57BL/6J mice. Behav Brain Res, 2005,157(2):245–251
Zhang D, Liu G, Shi J, et al. Coeloglossum viride var. bracteatum extract attenuates D-galactose and NaNO2 induced memory impairment in mice. J Ethnopharmacol, 2006,104(1–2):250–256
Chen CF, Lang SY, Zuo PP, et al. Effects of D-galactose on the expression of hippocampal peripheral-type benzodiazepine receptor and spatial memory performances in rats. Psychoneuroendocrinology, 2006,31(7):805–811
Kumar A, Prakash A, Dogra S. Naringin alleviates cognitive impairment, mitochondrial dysfunction and oxidative stress induced by D-galactose in mice. Food Chem Toxicol, 2010,48(2):626–632
Bedard K, Krause KH. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev, 2007,87(1):245–313
Pamenter ME, Ali SS, Tang Q, et al. An in vitro ischemic penumbral mimic perfusate increases NADPH oxidase-mediated superoxide production in cultured hippocampal neurons. Brain Res, 2012,1452:165–172
Kleinschnitz C, Grund H, Wingler K, et al. Post-stroke inhibition of induced NADPH oxidase type 4 prevents oxidative stress and neurodegeneration. PLoS Biol, 2010,8(9):e1000479
Chen H, Yoshioka H, Kim GS, et al. Oxidative stress in ischemic brain damage: mechanisms of cell death and potential molecular targets for neuroprotection. Antioxid Redox Signal, 2011,14(8):1505–1517
Walder CE, Green SP, Darbonne WC, et al. Ischemic stroke injury is reduced in mice lacking a functional NADPH oxidase. Stroke, 1997,28(11):2252–2258
Nair D, Dayyat EA, Zhang SX, et al. Intermittent hypoxia-induced cognitive deficits are mediated by NADPH oxidase activity in a murine model of sleep apnea. PLoS One, 2011,6(5):e19847
Raz L, Zhang QG, Zhou CF, et al. Role of Rac1 GTPase in NADPH oxidase activation and cognitive impairment following cerebral ischemia in the rat. PLoS One, 2010,5(9):e12606
Bruce-Keller AJ, Gupta S, Knight AG, et al. Cognitive impairment in humanized APPxPS1 mice is linked to Abeta(1–42) and NOX activation. Neurobiol Dis, 2011,44(3):317–326
Raha S, Robinson BH. Mitochondria, oxygen free radicals, disease and ageing. Trends Biochem Sci, 2000,25(10):502–508
Brand MD, Esteves TC. Physiological functions of the mitochondrial uncoupling proteins UCP2 and UCP3. Cell Metab, 2005,2(2):85–93
Barazzoni R, Nair KS. Changes in uncoupling protein-2 and -3 expression in aging rat skeletal muscle, liver, and heart. Am J Physiol Endocrinol Metab, 2001,280(3): E413–419
Amaral S, Mota P, Rodrigues AS, et al. Testicular aging involves mitochondrial dysfunction as well as an increase in UCP2 levels and proton leak. FEBS Lett, 2008,582(30): 4191–4196
Nicklas JA, Brooks EM, Hunter TC, et al. Development of a quantitative PCR (TaqMan) assay for relative mitochondrial DNA copy number and the common mitochondrial DNA deletion in the rat. Environ Mol Mutagen, 2004,44(4):313–320
Hiona A, Leeuwenburgh C. The role of mitochondrial DNA mutations in aging and sarcopenia: implications for the mitochondrial vicious cycle theory of aging. Exp Gerontol, 2008,43(1):24–33
Lee HC, Pang CY, Hsu HS, et al. Differential accumulations of 4,977 bp deletion in mitochondrial DNA of various tissues in human ageing. Biochim Biophys Acta, 1994,1226(1):37–43
Mohamed SA, Hanke T, Erasmi AW, et al. Mitochondrial DNA deletions and the aging heart. Exp Gerontol, 2006,41(5):508–517
Corral-Debrinski M, Horton T, Lott MT, et al. Mitochondrial DNA deletions in human brain: regional variability and increase with advanced age. Nat Genet, 1992,2(4): 324–329
Soong NW, Hinton DR, Cortopassi G, et al. Mosaicism for a specific somatic mitochondrial DNA mutation in adult human brain. Nat Genet, 1992,2(4):318–323
Kraytsberg Y, Kudryavtseva E, McKee AC, et al. Mitochondrial DNA deletions are abundant and cause functional impairment in aged human substantia nigra neurons. Nat Genet, 2006,38(5):518–520
Bender A, Krishnan KJ, Morris CM, et al. High levels of mitochondrial DNA deletions in substantia nigra neurons in aging and Parkinson disease. Nat Genet, 2006,38(5): 515–517
Tian Y, Zou B, Yang L, et al. High molecular weight persimmon tannin ameliorates cognition deficits and attenuates oxidative damage in senescent mice induced by D-galactose. Food Chem Toxicol, 2011,49(8):1728–1736
Zhang XL, An LJ, Bao YM, et al. d-galactose administration induces memory loss and energy metabolism disturbance in mice: protective effects of catalpol. Food Chem Toxicol, 2008,46(8):2888–2894
Cui X, Zuo P, Zhang Q, et al. Chronic systemic D-galactose exposure induces memory loss, neurodegeneration, and oxidative damage in mice: protective effects of R-alpha-lipoic acid. J Neurosci Res, 2006,84(3):647–654
Hamilton ML, Van Remmen H, Drake JA, et al. Does oxidative damage to DNA increase with age. Proc Natl Acad Sci USA, 2001,98(18):10469–10474
Fukui H, Moraes CT. Mechanisms of formation and accumulation of mitochondrial DNA deletions in aging neurons. Hum Mol Genet, 2009,18(6):1028–1036
Krishnan KJ, Reeve AK, Samuels DC, et al. What causes mitochondrial DNA deletions in human cells. Nat Genet, 2008,40(3):275–279
Brand MD, Affourtit C, Esteves TC, et al. Mitochondrial superoxide: production, biological effects, and activation of uncoupling proteins. Free Radic Biol Med, 2004,37(6): 755–767
Zhang CY, Baffy G, Perret P, et al. Uncoupling protein-2 negatively regulates insulin secretion and is a major link between obesity, beta cell dysfunction, and type 2 diabetes. Cell, 2001,105(6):745–755
Mizuno T, Miura-Suzuki T, Yamashita H, et al. Distinct regulation of brain mitochondrial carrier protein-1 and uncoupling protein-2 genes in the rat brain during cold exposure and aging. Biochem Biophys Res Commun, 2000,278(3):691–697
Deng S, Yang Y, Han Y, et al. UCP2 inhibits ROS-mediated apoptosis in A549 under hypoxic conditions. PLoS One, 2012,7(1):e30714
Author information
Authors and Affiliations
Corresponding authors
Additional information
This project was supported by grants from the Natural Science Foundation of Hubei province (No. 2010CDB08005), the National Natural Science Foundation of China (No. 30730094 and 81000409) and Special Funds for State Key Development Program for Basic Research of China (973 Program) (No. 2011CB504504).
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Du, Z., Hu, Y., Yang, Y. et al. NADPH oxidase-dependent oxidative stress and mitochondrial damage in hippocampus of D-galactose-induced aging rats. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 32, 466–472 (2012). https://doi.org/10.1007/s11596-012-0081-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-012-0081-z