Abstract
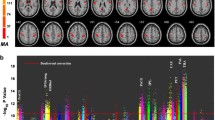
Schizophrenia is considered a disorder of abnormal brain connectivity. Although whole brain maps of averaged bivariate voxel correlations have been successfully applied to study connectivity abnormalities in schizophrenia these maps do not adequately explore the multivariate nature of brain connectivity. Here we adapt a novel method for high-dimensional regression (supervised principal component regression) to estimate brain maps of multivariate non redundant connectivity (NRC) from resting functional Magnetic Resonance Imaging (fMRI) data of 116 patients with schizophrenia and 122 matched controls. Disorder related differences in NRC involved caudate hyper-connectivity and hypo-connectivity of several cortical areas such as the dorsal cingulate, the cuneus and the right postcentral cortex. These abnormalities were coupled with abnormalities in the amplitude of signal fluctuations and, to a minor extent, with differences in the dimensionality of connectivity patterns as quantified by the number of supervised principal components. Second level seed correlation analyses linked the observed abnormalities to an additional set of brain regions relevant to schizophrenia such as the thalamus and the temporal cortex. The non redundant connectivity maps proposed here are a new tool that will complement the information provided by other already available voxel based whole brain connectivity measures.






Similar content being viewed by others
References
Alderson-Day, B., McCarthy-Jones, S., & Fernyhough, C. (2015). Hearing voices in the resting brain: a review of intrinsic functional connectivity research on auditory verbal hallucinations. Neuroscience & Biobehavioral Reviews, 55, 78–87. doi:10.1016/j.neubiorev.2015.04.016.
Anticevic, A., Brumbaugh, M. S., Winkler, A. M., Lombardo, L. E., Barrett, J., Corlett, P. R., et al. (2013). Global prefrontal and fronto-amygdala dysconnectivity in bipolar I disorder with psychosis history. Biological Psychiatry, 73(6), 565–573. doi:10.1016/j.biopsych.2012.07.031.
Anticevic, A., Cole, M. W., Repovs, G., Murray, J. D., Brumbaugh, M. S., Winkler, A. M., et al. (2014a). Characterizing thalamo-cortical disturbances in schizophrenia and bipolar illness. Cerebral Cortex, 24(12), 3116–3130. doi:10.1093/cercor/bht165.
Anticevic, A., Hu, S., Zhang, S., Savic, A., Billingslea, E., Wasylink, S., et al. (2014b). Global resting-state functional magnetic resonance imaging analysis identifies frontal cortex, striatal, and cerebellar dysconnectivity in obsessive-compulsive disorder. Biological Psychiatry, 75(8), 595–605. doi:10.1016/j.biopsych.2013.10.021.
Anticevic, A., Hu, X., Xiao, Y., Hu, J., Li, F., Bi, F., et al. (2015). Early-course unmedicated schizophrenia patients exhibit elevated prefrontal connectivity associated with longitudinal change. Journal of Neuroscience, 35(1), 267–286. doi:10.1523/JNEUROSCI.2310-14.2015.
Bair, E., Hastie, T., Debashis, P., & Tibshirani, R. (2006). Prediction by supervised principal components. Journal of the American Statistical Association, 101, 119–137.
Biswal, B., Yetkin, F. Z., Haughton, V. M., & Hyde, J. S. (1995). Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magnetic Resonance in Medicine, 34(4), 537–541.
Buckner, R. L., Sepulcre, J., Talukdar, T., Krienen, F. M., Liu, H., Hedden, T., et al. (2009). Cortical hubs revealed by intrinsic functional connectivity: mapping, assessment of stability, and relation to Alzheimer’s disease. Journal of Neuroscience, 29(6), 1860–1873. doi:10.1523/JNEUROSCI.5062-08.2009.
Bullmore, E., & Sporns, O. (2009). Complex brain networks: graph theoretical analysis of structural and functional systems. Nature Reviews Neuroscience, 10(3), 186–198. doi:10.1038/nrn2575.
Cole, M. W., Pathak, S., & Schneider, W. (2010). Identifying the brain’s most globally connected regions. NeuroImage, 49(4), 3132–3148. doi:10.1016/j.neuroimage.2009.11.001.
Cole, M. W., Anticevic, A., Repovs, G., & Barch, D. (2011). Variable global dysconnectivity and individual differences in schizophrenia. Biological Psychiatry, 70(1), 43–50. doi:10.1016/j.biopsych.2011.02.010.
Di, X., Kim, E. H., Huang, C. C., Tsai, S. J., Lin, C. P., & Biswal, B. B. (2013). The influence of the amplitude of low-frequency fluctuations on resting-state functional connectivity. Frontiers in Human Neuroscience, 7, 118. doi:10.3389/fnhum.2013.00118.
Fornito, A., Zalesky, A., Pantelis, C., & Bullmore, E. T. (2012). Schizophrenia, neuroimaging and connectomics. NeuroImage, 62(4), 2296–2314. doi:10.1016/j.neuroimage.2011.12.090.
Gottlich, M., Kramer, U. M., Kordon, A., Hohagen, F., & Zurowski, B. (2015). Resting-state connectivity of the amygdala predicts response to cognitive behavioral therapy in obsessive compulsive disorder. Biological Psychology, 111, 100–109. doi:10.1016/j.biopsycho.2015.09.004.
Guo, W. B., Liu, F., Xue, Z. M., Xu, X. J., Wu, R. R., Ma, C. Q., et al. (2012). Alterations of the amplitude of low-frequency fluctuations in treatment-resistant and treatment-response depression: a resting-state fMRI study. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 37(1), 153–160. doi:10.1016/j.pnpbp.2012.01.011.
Hastie, T., Debashis, P., & Friedman, J. K. (2009). The elements of statistical learning. Data mining, inference and prediction (2nd ed.). New York: Springer.
Hoptman, M. J., Zuo, X. N., Butler, P. D., Javitt, D. C., D’Angelo, D., Mauro, C. J., et al. (2010). Amplitude of low-frequency oscillations in schizophrenia: a resting state fMRI study. Schizophrenia Research, 117(1), 13–20. doi:10.1016/j.schres.2009.09.030.
Hou, J., Wu, W., Lin, Y., Wang, J., Zhou, D., Guo, J., et al. (2012). Localization of cerebral functional deficits in patients with obsessive-compulsive disorder: a resting-state fMRI study. Journal of Affective Disorders, 138(3), 313–321. doi:10.1016/j.jad.2012.01.022.
Hou, J. M., Zhao, M., Zhang, W., Song, L. H., Wu, W. J., Wang, J., et al. (2014). Resting-state functional connectivity abnormalities in patients with obsessive-compulsive disorder and their healthy first-degree relatives. Journal of Psychiatry & Neuroscience, 39(5), 304–311.
Jo, H. J., Gotts, S. J., Reynolds, R. C., Bandettini, P. A., Martin, A., Cox, R. W., et al. (2013). Effective preprocessing procedures virtually eliminate distance-dependent motion artifacts in resting state FMRI. Journal of Applied Mathematics. doi:10.1155/2013/935154.
Kaufmann, T., Skatun, K. C., Alnaes, D., Doan, N. T., Duff, E. P., Tonnesen, S., et al. (2015). Disintegration of sensorimotor brain networks in Schizophrenia. Schizophrenia Bulletin, 41(6), 1326–1335. doi:10.1093/schbul/sbv060.
Kay, S. R., Fiszbein, A., & Opler, L. A. (1987). The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophrenia Bulletin, 13(2), 261–276.
Liddle, P. F. (1987). The symptoms of chronic schizophrenia. a re-examination of the positive–negative dichotomy. British Journal of Psychiatry, 151, 145–151.
Modinos, G., Costafreda, S. G., van Tol, M. J., McGuire, P. K., Aleman, A., & Allen, P. (2013). Neuroanatomy of auditory verbal hallucinations in schizophrenia: a quantitative meta-analysis of voxel-based morphometry studies. Cortex, 49(4), 1046–1055. doi:10.1016/j.cortex.2012.01.009.
Pettersson-Yeo, W., Allen, P., Benetti, S., McGuire, P., & Mechelli, A. (2011). Dysconnectivity in schizophrenia: where are we now? Neuroscience & Biobehavioral Reviews, 35(5), 1110–1124. doi:10.1016/j.neubiorev.2010.11.004.
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L., & Petersen, S. E. (2012). Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. NeuroImage, 59(3), 2142–2154. doi:10.1016/j.neuroimage.2011.10.018.
R_Development_Core_Team. (2011). R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing.
Raichle, M. E. (2011). The restless brain. Brain Connectivity, 1(1), 3–12. doi:10.1089/brain.2011.0019.
Rubinov, M., & Bullmore, E. (2013). Schizophrenia and abnormal brain network hubs. Dialogues in Clinical Neuroscience, 15(3), 339–349.
Salvador, R., Suckling, J., Schwarzbauer, C., & Bullmore, E. (2005). Undirected graphs of frequency-dependent functional connectivity in whole brain networks. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 360(1457), 937–946. doi:10.1098/rstb.2005.1645.
Salvador, R., Vega, D., Pascual, J. C., Marco, J., Canales-Rodriguez, E. J., Aguilar, S., et al. (2014). Converging medial frontal resting state and diffusion-based abnormalities in borderline personality disorder. Biological Psychiatry. doi:10.1016/j.biopsych.2014.08.026.
Satterthwaite, T. D., Wolf, D. H., Loughead, J., Ruparel, K., Elliott, M. A., Hakonarson, H., et al. (2012). Impact of in-scanner head motion on multiple measures of functional connectivity: relevance for studies of neurodevelopment in youth. NeuroImage, 60(1), 623–632. doi:10.1016/j.neuroimage.2011.12.063.
Satterthwaite, T. D., Elliott, M. A., Gerraty, R. T., Ruparel, K., Loughead, J., Calkins, M. E., et al. (2013). An improved framework for confound regression and filtering for control of motion artifact in the preprocessing of resting-state functional connectivity data. NeuroImage, 64, 240–256. doi:10.1016/j.neuroimage.2012.08.052.
Sepulcre, J., Liu, H., Talukdar, T., Martincorena, I., Yeo, B. T., & Buckner, R. L. (2010). The organization of local and distant functional connectivity in the human brain. PLoS Computational Biology, 6(6), e1000808. doi:10.1371/journal.pcbi.1000808.
Shen, Y., Yao, J., Jiang, X., Zhang, L., Xu, L., Feng, R., et al. (2015). Sub-hubs of baseline functional brain networks are related to early improvement following two-week pharmacological therapy for major depressive disorder. Human Brain Mapping, 36(8), 2915–2927. doi:10.1002/hbm.22817.
Smith, S. M., Jenkinson, M., Woolrich, M. W., Beckmann, C. F., Behrens, T. E., Johansen-Berg, H., et al. (2004). Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage, 23(Suppl 1), S208–219. doi:10.1016/j.neuroimage.2004.07.051.
Stephan, K. E., Friston, K. J., & Frith, C. D. (2009). Dysconnection in schizophrenia: from abnormal synaptic plasticity to failures of self-monitoring. Schizophrenia Bulletin, 35(3), 509–527. doi:10.1093/schbul/sbn176.
Steriade, M., & Llinas, R. R. (1988). The functional states of the thalamus and the associated neuronal interplay. Physiological Reviews, 68(3), 649–742.
Turner, J. A., Damaraju, E., van Erp, T. G., Mathalon, D. H., Ford, J. M., Voyvodic, J., et al. (2013). A multi-site resting state fMRI study on the amplitude of low frequency fluctuations in schizophrenia. Frontiers in Neuroscience, 7, 137. doi:10.3389/fnins.2013.00137.
Wechsler, D. (1997). Wechsler memory scale (3rd ed.). San Antonio: The Psychological Corporation.
Wilson, B. A. N., & Burgess, P. (1996). Behavioural assessment of the Dysexecutive Syndrome(BADS). London: Harcourt Assessment.
Woodward, N. D., Karbasforoushan, H., & Heckers, S. (2012). Thalamocortical dysconnectivity in schizophrenia. The American Journal of Psychiatry, 169(10), 1092–1099. doi:10.1176/appi.ajp.2012.12010056.
Xu, Y., Zhuo, C., Qin, W., Zhu, J., & Yu, C. (2015). Altered spontaneous brain activity in schizophrenia: a meta-analysis and a large-sample study. Biomedical Research International, 2015, 204628. doi:10.1155/2015/204628.
Yu, R., Chien, Y. L., Wang, H. L., Liu, C. M., Liu, C. C., Hwang, T. J., et al. (2014). Frequency-specific alternations in the amplitude of low-frequency fluctuations in schizophrenia. Human Brain Mapping, 35(2), 627–637. doi:10.1002/hbm.22203.
Zang, Y. F., He, Y., Zhu, C. Z., Cao, Q. J., Sui, M. Q., Liang, M., et al. (2007). Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain Dev, 29(2), 83–91. doi:10.1016/j.braindev.2006.07.002.
Acknowledgments
This work was supported by the Catalonian Government (2014SGR1573), several grants from the Plan Nacional de I + D + i and co-funded by the Instituto de Salud Carlos III-Subdirección General de Evaluación y Fomento de la Investigación and the European Regional Development Fund (FEDER): Miguel Servet Research Contracts (CP10/00596 to EP-C, CP13/00018 to RS and CP14/00041 to JR) and Research Project Grants (PI14/00292, PI14/01691, PI14/01148 and PI14/01151).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Author Raymond Salvador, Author Ramón Landín-Romero, Author Maria Anguera, Author Erick J. Canales-Rodríguez, Author Joaquim Raduà, Author Amalia Guerrero-Pedraza, Author Salvador Sarró, Author Teresa Maristany, Author Peter J. McKenna and Author Edith Pomarol-Clotet declare that they have no conflict of interest.
Informed consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, and the applicable revisions at the time of the investigation. Informed consent was obtained from all patients for being included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 115 kb)
Appendix 1 Development of a computationally efficient formula to calculate NRC values
Appendix 1 Development of a computationally efficient formula to calculate NRC values
Equation 2 of the main text involves a strong reduction in the dimensionality of the independent variable set from p to m variables (with m < N) making the calculation of a multiple correlation feasible. Still, the computational burden of fitting Eq. 2 for each one of the grey matter voxels is very high. Luckily, as shown below Eq. 2 can be estimated without the need to obtain the PCs or their correlations with Y at each single voxel.
We start with the definition of
where L T is the matrix of m orthogonal and normalized eigenvectors related to the m components (given in rows). Each one of the components PC i has a different variance given by its respective eigenvalue Λ i (and Λ is the diagonal covariance matrix of PC).
Now, due to the scale invariance of correlations, we can consider an alternative formula for Eq. 2 based on normalized components \( NPC=\left\{P{C}_1/\sqrt{\varLambda_1},P{C}_2/\sqrt{\varLambda_2},\dots, P{C}_m/\sqrt{\varLambda_m}\right\} \)
and since Y and NPC have unit variance
which, by considering Eq. A1, becomes \( \mathrm{c}\mathrm{o}\mathrm{r}\;\left(Y,PC\right)=\sqrt{\varLambda^{-1}}\;\mathrm{c}\mathrm{o}\mathrm{v}\;\left(Y,{L}^TX\right)=\sqrt{\varLambda^{-1}}\;{L}^T\mathrm{c}\mathrm{o}\mathrm{r}\;\left(Y,X\right) \).
Finally, Eq. 2 (main text) may be reduced to
And since Λ − 1 is diagonal, it may be further simplified to a sum
in which every term is the squared correlation between each component and Y.
In summary, since both eigenvectors (L) and eigenvalues (Λ) are obtained from the diagonalization of correlation matrices, once the matrix containing correlations between each pair of voxels is initially calculated there is no need to operate with X anymore.
Finally, computational time can be further reduced by implementing sub-sampling strategies, which are acceptable considering the high levels of spatial autocorrelation in fMRI images. In this study a sub-sample of randomly chosen voxels was used for the computation of NRC at each voxel.
Rights and permissions
About this article
Cite this article
Salvador, R., Landin-Romero, R., Anguera, M. et al. Non redundant functional brain connectivity in schizophrenia. Brain Imaging and Behavior 11, 552–564 (2017). https://doi.org/10.1007/s11682-016-9535-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-016-9535-4