Abstract
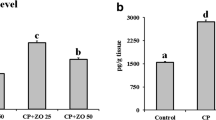
Cisplatin (CP) is one of the most important anticancer compounds with its therapeutic usefulness in diverse types of solid cancer. However, its use is limited due to nephrotoxicity induced by it. Oxidative stress is an effective participant which contributes actively to pathogenesis of CP-induced nephrotoxicity. Nanoparticle form of a rare earth metal cerium, also known as nanoceria (NC), has come up as a potential antioxidant and anti-inflammatory agent. In the present study, administration of CP in Swiss mice resulted in reduction of body weight, increased oxidative stress and pro-inflammatory cytokine levels including IL-6 and TNF-α along with alteration in normal histological architecture of kidney. On the contrary, NC (0.2 and 2 mg/kg i.p.) ameliorated nephrotoxicity of CP which was evident by reduction in levels of renal injury markers in plasma, i.e., creatinine and blood urea nitrogen. NC ameliorated oxidative stress by showing a reduction in levels of malondialdehyde and increased levels of endogenous antioxidants reduced glutathione and catalase. Further, NC treatment also reduced the levels of pro-inflammatory cytokines. Furthermore, protective effect of NC was also corroborated by histopathological studies wherein, kidneys from CP group showed altered tissue structure after acute as well as chronic exposure of CP while the tissues from treated groups showed absence of alterations in kidney histology. The results from present study suggested that oxidative stress and pro-inflammatory cytokines play a central role in pathogenesis of CP-induced nephrotoxicity and NC provides protection from CP-induced nephrotoxicity due to its antioxidant and anti-inflammatory properties.






Similar content being viewed by others
References
Wang D, Lippard SJ (2005) Cellular processing of platinum anticancer drugs. Nat Rev Drug Discov 4(4):307–320
Cohen SM, Lippard SJ (2001) Cisplatin: from DNA damage to cancer chemotherapy. Prog Nucleic Acid Res Mol Biol 67:93–130
Siddik ZH (2003) Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene 22(47):7265–7279
Heiger-Bernays WJ, Essigmann JM, Lippard SJ (1990) Effect of the antitumor drug cis-diamminedichloroplatinum (II) and related platinum complexes on eukaryotic DNA replication. Biochemistry 29(36):8461–8466
Jamieson ER, Lippard SJ (1999) Structure, recognition, and processing of cisplatin− DNA adducts. Chem Rev 99(9):2467–2498
Pabla N, Dong Z (2008) Cisplatin nephrotoxicity: mechanisms and renoprotective strategies. Kidney Int 73(9):994–1007
Ciarimboli G, Ludwig T, Lang D, Pavenstädt H, Koepsell H, Piechota H-J, Haier J, Jaehde U, Zisowsky J, Schlatter E (2005) Cisplatin nephrotoxicity is critically mediated via the human organic cation transporter 2. Am J Pathol 167(6):1477–1484
Kuhlmann M, Burkhardt G, Köhler H (1997) Insights into potential cellular mechanisms of cisplatin nephrotoxicity and their clinical application. Nephrol Dial Transplant 12(12):2478–2480
Chirino YI, Pedraza-Chaverri J (2009) Role of oxidative and nitrosative stress in cisplatin-induced nephrotoxicity. Exp Toxicol Pathol 61(3):223–242
Atessahin A, Yilmaz S, Karahan I, Ceribasi AO, Karaoglu A (2005) Effects of lycopene against cisplatin-induced nephrotoxicity and oxidative stress in rats. Toxicology 212(2–3):116–123
de Oliveira Mora L, Antunes LMG, Bianchi MdLP (2003) The effects of oral glutamine on cisplatin-induced nephrotoxicity in rats. Pharmacol Res 47(6):517–522
Bompart G (1989) Cisplatin-induced changes in cytochrome P-450, lipid peroxidation and drug-metabolizing enzyme activities in rat kidney cortex. Toxicol Lett 48(2):193–199
Jin-Gang Z, Lindup WE (1993) Role of mitochondria in cisplatin-induced oxidative damage exhibited by rat renal cortical slices. Biochem Pharmacol 45(11):2215–2222
Hannemann J, Baumann K (1988) Cisplatin-induced lipid peroxidation and decrease of gluconeogenesis in rat kidney cortex: different effects of antioxidants and radical scavengers. Toxicology 51(2–3):119–132
Kemp G, Rose P, Lurain J, Berman M, Manetta A, Roullet B, Homesley H, Belpomme D, Glick J (1996) Amifostine pretreatment for protection against cyclophosphamide-induced and cisplatin-induced toxicities: results of a randomized control trial in patients with advanced ovarian cancer. J Clin Oncol 14(7):2101–2112
Foster-Nora JA, Siden R (1997) Amifostine for protection from antineoplastic drug toxicity. Am J Health Syst Pharm 54(7):787–800
Hartmann JT, Fels LM, Knop S, Stolte H, Kanz L, Bokemeyer C (2000) A randomized trial comparing the nephrotoxicity of cisplatin/ifosfamide-based combination chemotherapy with or without amifostine in patients with solid tumors. Investig New Drugs 18(3):281–289
Castiglione F, Dalla AM, Porcile G (1999) Protection of normal tissues from radiation and cytotoxic therapy: the development of amifostine. Tumori 85(2):85–91
Ramnath N, LoRusso P, Simon M, Martino S (1997) Phase II evaluation of cisplatin and WR2721 for refractory metastatic breast cancer. Am J Clin Oncol 20(4):368–372
Rick O, Beyer J, Schwella N, Schubart H, Schleicher J, Siegert W (2001) Assessment of amifostine as protection from chemotherapy-induced toxicities after conventional-dose and high-dose chemotherapy in patients with germ cell tumor. Ann Oncol 12(8):1151–1155
Kyosseva SV, Chen L, Seal S, McGinnis JF (2013) Nanoceria inhibit expression of genes associated with inflammation and angiogenesis in the retina of Vldlr null mice. Exp Eye Res 116:63–74
Hirst SM, Karakoti AS, Tyler RD, Sriranganathan N, Seal S, Reilly CM (2009) Anti-inflammatory properties of cerium oxide nanoparticles. Small 5(24):2848–2856
Celardo I, Traversa E, Ghibelli L (2011) Cerium oxide nanoparticles: a promise for applications in therapy. J Exp Ther Oncol 9(1):47–51
Das M, Patil S, Bhargava N, Kang J-F, Riedel LM, Seal S, Hickman JJ (2007) Auto-catalytic ceria nanoparticles offer neuroprotection to adult rat spinal cord neurons. Biomaterials 28(10):1918–1925
González-Flores D, De Nicola M, Bruni E, Caputo F, Rodríguez AB, Pariente JA, Ghibelli L (2014) Nanoceria protects from alterations in oxidative metabolism and calcium overloads induced by TNFα and cycloheximide in U937 cells: pharmacological potential of nanoparticles. Mol Cell Biochem 397(1–2):245–253
Pirmohamed T, Dowding JM, Singh S, Wasserman B, Heckert E, Karakoti AS, King JE, Seal S, Self WT (2010) Nanoceria exhibit redox state-dependent catalase mimetic activity. Chem Commun 46(16):2736–2738
Korsvik C, Patil S, Seal S, Self WT (2007) Superoxide dismutase mimetic properties exhibited by vacancy engineered ceria nanoparticles. Chem Commun 10:1056–1058
Dowding JM, Dosani T, Kumar A, Seal S, Self WT (2012) Cerium oxide nanoparticles scavenge nitric oxide radical (˙ NO). Chem Commun 48(40):4896–4898
Cimini A, D’Angelo B, Das S, Gentile R, Benedetti E, Singh V, Monaco AM, Santucci S, Seal S (2012) Antibody-conjugated PEGylated cerium oxide nanoparticles for specific targeting of Aβ aggregates modulate neuronal survival pathways. Acta Biomater 8(6):2056–2067
Estevez A, Pritchard S, Harper K, Aston J, Lynch A, Lucky J, Ludington J, Chatani P, Mosenthal W, Leiter J (2011) Neuroprotective mechanisms of cerium oxide nanoparticles in a mouse hippocampal brain slice model of ischemia. Free Radic Biol Med 51(6):1155–1163
Heckman KL, DeCoteau W, Estevez A, Reed KJ, Costanzo W, Sanford D, Leiter JC, Clauss J, Knapp K, Gomez C (2013) Custom cerium oxide nanoparticles protect against a free radical mediated autoimmune degenerative disease in the brain. ACS Nano 7(12):10582–10596
Sangomla S, Saifi MA, Khurana A, Godugu C (2018) Nanoceria ameliorates doxorubicin induced cardiotoxicity: possible mitigation via reduction of oxidative stress and inflammation. J Trace Elem Med Biol 47:53–62
Niu J, Azfer A, Rogers LM, Wang X, Kolattukudy PE (2007) Cardioprotective effects of cerium oxide nanoparticles in a transgenic murine model of cardiomyopathy. Cardiovasc Res 73(3):549–559
Pourkhalili N, Hosseini A, Nili-Ahmadabadi A, Rahimifard M, Navaei-Nigjeh M, Hassani S, Baeeri M, Abdollahi M (2012) Improvement of isolated rat pancreatic islets function by combination of cerium oxide nanoparticles/sodium selenite through reduction of oxidative stress. Toxicol Mech Methods 22(6):476–482
Khurana A, Saifi M, Godugu C (2017) Nanoceria reduces oxidative stress, inflammation and display anti-fibrotic properties in animal models of chronic pancreatitis. Pancreas, Lippincott Williams & Wilkins Two Commerce SQ, 2001 Market St, Philadelphia, PA 19103 USA, pp 1411–1412
Asati A, Santra S, Kaittanis C, Perez JM (2010) Surface-charge-dependent cell localization and cytotoxicity of cerium oxide nanoparticles. ACS Nano 4(9):5321–5331
Sack M, Alili L, Karaman E, Das S, Gupta A, Seal S, Brenneisen P (2014) Combination of conventional chemotherapeutics with redox-active cerium oxide nanoparticles—a novel aspect in cancer therapy. Mol Cancer Ther 13(7):1740–1749
Tarnuzzer RW, Colon J, Patil S, Seal S (2005) Vacancy engineered ceria nanostructures for protection from radiation-induced cellular damage. Nano Lett 5(12):2573–2577
Xu M-X, Zhu Y-F, Chang H-F, Liang Y (2016) Nanoceria restrains PM2. 5-induced metabolic disorder and hypothalamus inflammation by inhibition of astrocytes activation related NF-κB pathway in Nrf2 deficient mice. Free Radic Biol Med 99:259–272
Reddy DR, Khurana A, Bale S, Ravirala R, Reddy VSS, Mohankumar M, Godugu C (2016) Natural flavonoids silymarin and quercetin improve the brain distribution of co-administered P-gp substrate drugs. SpringerPlus 5(1):1618
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim Biophys Acta Gen Subj 582(1):67–78
Aebi H (1984) [13] Catalase in vitro. Methods Enzymol 105:121–126
Kumar GS, Kulkarni A, Khurana A, Kaur J, Tikoo K (2014) Selenium nanoparticles involve HSP-70 and SIRT1 in preventing the progression of type 1 diabetic nephropathy. Chem Biol Interact 223:125–133
Ries F, Klastersky J (1986) Nephrotoxicity induced by cancer chemotherapy with special emphasis on cisplatin toxicity. Am J Kidney Dis 8(5):368–379
Gong J, Costanzo A, Yang H-Q, Melino G, Kaelin WG, Levrero M, Wang JY (1999) The tyrosine kinase c-Abl regulates p73 in apoptotic response to cisplatin-induced DNA damage. Nature 399(6738):806–809
Leibbrandt ME, Wolfgang GH, Metz AL, Ozobia AA, Haskins JR (1995) Critical subcellular targets of cisplatin and related platinum analogs in rat renal proximal tubule cells. Kidney Int 48(3):761–770
Antunes LMG, Darin JDAC, Maria de Lourdes PB (2001) Effects of the antioxidants curcumin or selenium on cisplatin-induced nephrotoxicity and lipid peroxidation in rats. Pharmacol Res 43(2):145–150
Santos N, Catao C, Martins N, Curti C, Bianchi M, Santos A (2007) Cisplatin-induced nephrotoxicity is associated with oxidative stress, redox state unbalance, impairment of energetic metabolism and apoptosis in rat kidney mitochondria. Arch Toxicol 81(7):495–504
Cummings BS, Schnellmann RG (2002) Cisplatin-induced renal cell apoptosis: caspase 3-dependent and-independent pathways. J Pharmacol Exp Ther 302(1):8–17
Zhang B, Ramesh G, Norbury C, Reeves W (2007) Cisplatin-induced nephrotoxicity is mediated by tumor necrosis factor-α produced by renal parenchymal cells. Kidney Int 72(1):37–44
Baliga R, Ueda N, Walker PD, Shah SV (1999) Oxidant mechanisms in toxic acute renal failure. Drug Metab Rev 31(4):971–997
Ramesh G, Reeves WB (2004) Inflammatory cytokines in acute renal failure. Kidney Int 66:S56–S61
Ramesh G, Reeves WB (2004) Salicylate reduces cisplatin nephrotoxicity by inhibition of tumor necrosis factor-α. Kidney Int 65(2):490–498
Antunes LMG, Darin JDAC, Bianchi MDLP (2000) Protective effects of vitamin C against cisplatin-induced nephrotoxicity and lipid peroxidation in adult rats: a dose-dependent study. Pharmacol Res 41(4):405–411
Alili L, Sack M, Karakoti AS, Teuber S, Puschmann K, Hirst SM, Reilly CM, Zanger K, Stahl W, Das S (2011) Combined cytotoxic and anti-invasive properties of redox-active nanoparticles in tumor–stroma interactions. Biomaterials 32(11):2918–2929
Lin W, Huang Y-w, Zhou X-D, Ma Y (2006) Toxicity of cerium oxide nanoparticles in human lung cancer cells. Int J Toxicol 25(6):451–457
Alili L, Sack M, von Montfort C, Giri S, Das S, Carroll KS, Zanger K, Seal S, Brenneisen P (2013) Downregulation of tumor growth and invasion by redox-active nanoparticles. Antioxid Redox Signal 19(8):765–778
Cooke MS, Evans MD, Dizdaroglu M, Lunec J (2003) Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J 17(10):1195–1214
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408(6809):239–247
Kumari P, Saifi MA, Khurana A, Godugu C (2018) Cardioprotective effects of nanoceria in a murine model of cardiac remodeling. J Trace Elem Med Biol 50:198–208
Ramesh G, Reeves WB (2002) TNF-α mediates chemokine and cytokine expression and renal injury in cisplatin nephrotoxicity. J Clin Invest 110(6):835–842
Kouvaris JR, Kouloulias VE, Vlahos LJ (2007) Amifostine: the first selective-target and broad-spectrum radioprotector. Oncologist 12(6):738–747
Saifi MA, Khurana A, Godugu C (2018) Nanomaterials in chromatography: current trends in chromatographic research technology and techniques. In: Nanotoxicology: toxicity and risk assessment of nanomaterials, nanomaterials in chromatography. Elsevier, Amsterdam, pp 437–465
Saifi M, Khan W, Godugu C (2018) Cytotoxicity of nanomaterials: nanotoxicology to address the safety concerns of nanoparticles. Pharm Nanotechnol 6(1):3–16
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saifi, M.A., Sangomla, S., Khurana, A. et al. Protective Effect of Nanoceria on Cisplatin-Induced Nephrotoxicity by Amelioration of Oxidative Stress and Pro-inflammatory Mechanisms. Biol Trace Elem Res 189, 145–156 (2019). https://doi.org/10.1007/s12011-018-1457-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-018-1457-0