Abstract
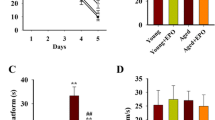
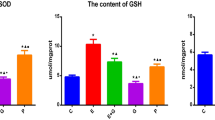
Erythropoietin (EPO) has a neuroprotective effect and can resist aging, which most likely occur through EPO increasing the activity of antioxidant enzymes and scavenging free radicals. In this study, we verified the anti-aging function of EPO and discussed the mechanism occurring through the extracellular signal-regulated kinase (ERK)/NF-E2-related factor 2 (Nrf2)-ARE pathway. A rat model of aging was induced by the continuous subcutaneous injection of 5 % d-galactose for 6 weeks. At the beginning of the sixth week, physiological saline or EPO was administered twice per day through a lateral ventricle system for a total of 7 days. In one group, 2 μl PD98059 was administered 30 min before EPO. Learning and memory ability were analyzed with the Morris water maze system. HE staining was used to observe the morphological changes in the neurons in the hippocampus, and immunohistochemical staining as well as Western blots were carried out to detect the expression of ERK for each group of rats and the expression of phosphorylated-ERK (P-ERK), Nrf2, and superoxide dismutase (SOD). Real-Time PCR was carried out to detect the amount of Nrf2 mRNA and the KEAP1 mRNA expression. EPO can significantly improve learning and memory ability in aging rats and can provide protection against aging by improving the hippocampus morphology. Immunohistochemical staining and Western blots showed P-ERK, Nrf2, and Cu-Zn SOD decreases in aging rats compared to the normal group, while the expression for those proteins increased after EPO intervention. PD98059 inhibited the enhanced expression of P-ERK, Nrf2, and Cu-Zn SOD induced by EPO. Real-Time PCR results suggested that the trend of Nrf2mRNA expression was the same as that for the proteins, which confirmed that the enhancement occurred at the gene level. As such, EPO can significantly resist or delay aging and protect the brain by reducing oxidative stress. The most likely mechanism is that EPO can promote the ERK/Nrf2-ARE pathway in aging rats and that PD98059 can inhibit that process. These findings may facilitate further studies on the mechanism of aging and applications for the neuroprotective properties of EPO for clinical treatments.








Similar content being viewed by others
References
Buendia I, Michalska P, Navarro E, Gameiro I, Egea J, Leon R (2016) Nrf2-ARE pathway: an emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol Ther 157:84–104
Bundy JD, He J (2016) Hypertension and related cardiovascular disease burden in China. Ann Glob Health 82:227–233
Butler RN, Miller RA, Perry D, Carnes BA, Williams TF, Cassel C, Brody J, Bernard MA, Partridge L, Kirkwood T, Martin GM, Olshansky SJ (2008) New model of health promotion and disease prevention for the 21st century. BMJ 337:a399
Chetelat G, Ossenkoppele R, Villemagne VL, Perrotin A, Landeau B, Mezenge F, Jagust WJ, Dore V, Miller BL, Egret S, Seeley WW, van der Flier WM, La Joie R, Ames D, van Berckel BN, Scheltens P, Barkhof F, Rowe CC, Masters CL, de La Sayette V, Bouwman F, Rabinovici GD (2016) Atrophy, hypometabolism and clinical trajectories in patients with amyloid-negative Alzheimer’s disease. Brain 139:2528–2539
Costa LG, Garrick JM, Roque PJ, Pellacani C (2016) Mechanisms of neuroprotection by quercetin: counteracting oxidative stress and more. Oxidative Med Cell Longev 2016:2986796
Digicaylioglu M, Lipton SA (2001) Erythropoietin-mediated neuroprotection involves cross-talk between Jak2 and NF-kappaB signalling cascades. Nature 412:641–647
Engel PA (2016) Is age-related failure of metabolic reprogramming a principal mediator in idiopathic Parkinson’s disease? Implications for treatment and inverse cancer risk. Med Hypotheses 93:154–160
Gassen NC, Chrousos GP, Binder EB, Zannas AS (2016) Life stress, glucocorticoid signaling, and the aging epigenome: implications for aging-related diseases. Neurosci Biobehav Rev
Genc K, Egrilmez MY, Genc S (2010) Erythropoietin induces nuclear translocation of Nrf2 and heme oxygenase-1 expression in SH-SY5Y cells. Cell Biochem Funct 28:197–201
Herrera-Arozamena C, Marti-Mari O, Estrada M, de la Fuente Revenga M, Rodriguez-Franco MI (2016) Recent advances in neurogenic small molecules as innovative treatments for neurodegenerative diseases. Molecules 21
Jin W, Kong J, Lu T, Wang H, Ni H, Wu J, Dai Y, Jiang J, Liang W (2011) Erythropoietin prevents secondary brain injury induced by cortical lesion in mice: possible involvement of Nrf2 signaling pathway. Ann Clin Lab Sci 41:25–32
Juul S (2012) Neuroprotective role of erythropoietin in neonates. J Matern Fetal Neonatal Med 25(Suppl 4):105–107
Juul SE, Pet GC (2015) Erythropoietin and neonatal neuroprotection. Clin Perinatol 42:469–481
Katsuoka F, Yamamoto M (2016) Small Maf proteins (MafF, MafG, MafK): history, structure and function. Gene 586:197–205
Katsuoka F, Motohashi H, Engel JD, Yamamoto M (2005) Nrf2 transcriptionally activates the mafG gene through an antioxidant response element. J Biol Chem 280:4483–4490
Khalil SK, Amer HA, El Behairy AM, Warda M (2016) Oxidative stress during erythropoietin hyporesponsiveness anemia at end stage renal disease: molecular and biochemical studies. J Adv Res 7:348–358
Kilic E, Kilic U, Soliz J, Bassetti CL, Gassmann M, Hermann DM (2005) Brain-derived erythropoietin protects from focal cerebral ischemia by dual activation of ERK-1/-2 and Akt pathways. FASEB J 19:2026–2028
Koltover VK (2016) Free radical timer of aging: from chemistry of free radicals to systems theory of reliability. Curr Aging Sci
Koskenkorva-Frank TS, Weiss G, Koppenol WH, Burckhardt S (2013) The complex interplay of iron metabolism, reactive oxygen species, and reactive nitrogen species: insights into the potential of various iron therapies to induce oxidative and nitrosative stress. Free Radic Biol Med 65:1174–1194
Kume T, Suenaga A, Izumi Y, Akaike A (2016) Protective effect of dimethyl fumarate on an oxidative stress model induced by sodium nitroprusside in mice. Biol Pharm Bull 39:1055–1059
Li YP, Yang GJ, Jin L, Yang HM, Chen J, Chai GS, Wang L (2015) Erythropoietin attenuates Alzheimer-like memory impairments and pathological changes induced by amyloid beta42 in mice. Brain Res 1618:159–167
Liochev SI (2015) Which is the most significant cause of aging? Antioxidants (Basel) 4:793–810
Lu MC, Ji JA, Jiang ZY, You QD (2016) The Keap1-Nrf2-ARE pathway as a potential preventive and therapeutic target: an update. Med Res Rev 36:924–963
Merelli A, Czornyj L, Lazarowski A (2013) Erythropoietin: a neuroprotective agent in cerebral hypoxia, neurodegeneration, and epilepsy. Curr Pharm Des 19:6791–6801
Merelli A, Czornyj L, Lazarowski A (2015) Erythropoietin as a new therapeutic opportunity in brain inflammation and neurodegenerative diseases. Int J Neurosci 125:793–797
Morishita E, Masuda S, Nagao M, Yasuda Y, Sasaki R (1997) Erythropoietin receptor is expressed in rat hippocampal and cerebral cortical neurons, and erythropoietin prevents in vitro glutamate-induced neuronal death. Neuroscience 76:105–116
Sanz A (2016) Mitochondrial reactive oxygen species: do they extend or shorten animal lifespan? Biochim Biophys Acta 1857:1116–1126
Sargin D, Friedrichs H, El-Kordi A, Ehrenreich H (2010) Erythropoietin as neuroprotective and neuroregenerative treatment strategy: comprehensive overview of 12 years of preclinical and clinical research. Best Pract Res Clin Anaesthesiol 24:573–594
Wu H, Wang H, Zhang W, Wei X, Zhao J, Yan P, Liu C (2015) rhEPO affects apoptosis in hippocampus of aging rats by upregulating SIRT1. Int J Clin Exp Pathol 8:6870–6880
Zhang DD (2006) Mechanistic studies of the Nrf2-Keap1 signaling pathway. Drug Metab Rev 38:769–789
Acknowledgements
This study was supported by the National Natural Science Foundation of China (81170300).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, H., Zhao, J., Chen, M. et al. The Anti-Aging Effect of Erythropoietin via the ERK/Nrf2-ARE Pathway in Aging Rats. J Mol Neurosci 61, 449–458 (2017). https://doi.org/10.1007/s12031-017-0885-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-017-0885-1