Abstract
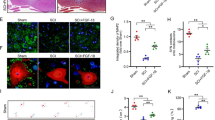
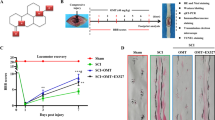
Autophagy is an intracellular catabolic mechanism for the degradation of cytoplasmic constituents in the autophagosomal–lysosomal pathway. This mechanism plays an important role in homeostasis and it is defective in certain diseases. Preceding studies have revealed that autophagy is developing as an important moderator of pathological responses associated to spinal cord injury (SCI) and plays a crucial role in secondary injury initiating a progressive degeneration of the spinal cord. Thus, based on this evidence in this study, we used two different selective inhibitors of mTOR activity to explore the functional role of autophagy in an in vivo model of SCI as well as to determine whether the autophagic process is involved in spinal cord tissue damage. We treated animals with a novel synthetic inhibitor temsirolimus and with a dual mTORC1 and mTORC2 inhibitor KU0063794 matched all with the well-known inhibitor of mTOR the rapamycin. Our results demonstrated that mTOR inhibitors could regulate the neuroinflammation associated to SCI and the results that we obtained evidently demonstrated that rapamycin and temsirolimus significantly diminished the expression of iNOS, COX2, GFAP, and re-established nNOS levels, but the administration of KU0063794 is able to blunt the neuroinflammation better than rapamycin and temsirolimus. In addition, neuronal loss and cell mortality in the spinal cord after injury were considerably reduced in the KU0063794-treated mice. Accordingly, taken together our results denote that the administration of KU0063794 produced a neuroprotective function at the lesion site following SCI, representing a novel therapeutic approach after SCI.





Similar content being viewed by others
References
Blesch A, Tuszynski MH (2009) Spinal cord injury: plasticity, regeneration and the challenge of translational drug development. Trends Neurosci 32(1):41–47. doi:10.1016/j.tins.2008.09.008
Wang W, Guo Z, Xu Z, Meng Q, Chen C, Zhang Y, Cao X (2015) Effect of pollen typhae on inhibiting autophagy in spinal cord injury of rats and its mechanisms. Int J Clin Exp Pathol 8(3):2375–2383
Wullschleger S, Loewith R, Hall MN (2006) TOR signaling in growth and metabolism. Cell 124(3):471–484. doi:10.1016/j.cell.2006.01.016
Betz C, Hall MN (2013) Where is mTOR and what is it doing there? J Cell Biol 203(4):563–574. doi:10.1083/jcb.201306041
Menon S, Manning BD (2008) Common corruption of the mTOR signaling network in human tumors. Oncogene 27(Suppl 2):S43–51. doi:10.1038/onc.2009.352
Foster KG, Fingar DC (2010) Mammalian target of rapamycin (mTOR): conducting the cellular signaling symphony. J Biol Chem 285(19):14071–14077. doi:10.1074/jbc.R109.094003
Sparks CA, Guertin DA (2010) Targeting mTOR: prospects for mTOR complex 2 inhibitors in cancer therapy. Oncogene 29(26):3733–3744. doi:10.1038/onc.2010.139
Phung TL, Ziv K, Dabydeen D, Eyiah-Mensah G, Riveros M, Perruzzi C, Sun J, Monahan-Earley RA et al (2006) Pathological angiogenesis is induced by sustained Akt signaling and inhibited by rapamycin. Cancer Cell 10(2):159–170. doi:10.1016/j.ccr.2006.07.003
Sarbassov DD, Ali SM, Sengupta S, Sheen JH, Hsu PP, Bagley AF, Markhard AL, Sabatini DM (2006) Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. Mol Cell 22(2):159–168. doi:10.1016/j.molcel.2006.03.029
Duran I, Siu LL, Oza AM, Chung TB, Sturgeon J, Townsley CA, Pond GR, Seymour L et al (2006) Characterisation of the lung toxicity of the cell cycle inhibitor temsirolimus. Eur J Cancer 42(12):1875–1880. doi:10.1016/j.ejca.2006.03.015
Garcia-Martinez JM, Moran J, Clarke RG, Gray A, Cosulich SC, Chresta CM, Alessi DR (2009) Ku-0063794 is a specific inhibitor of the mammalian target of rapamycin (mTOR). Biochem J 421(1):29–42. doi:10.1042/BJ20090489
Zhang H, Berel D, Wang Y, Li P, Bhowmick NA, Figlin RA, Kim HL (2013) A comparison of Ku0063794, a dual mTORC1 and mTORC2 inhibitor, and temsirolimus in preclinical renal cell carcinoma models. PLoS One 8(1):e54918. doi:10.1371/journal.pone.0054918
Syed F, Sanganee HJ, Singh S, Bahl A, Bayat A (2013) Potent dual inhibitors of TORC1 and TORC2 complexes (KU-0063794 and KU-0068650) demonstrate in vitro and ex vivo anti-keloid scar activity. J Invest Dermatol 133(5):1340–1350. doi:10.1038/jid.2012.483
Kanno H, Ozawa H, Sekiguchi A, Yamaya S, Tateda S, Yahata K, Itoi E (2012) The role of mTOR signaling pathway in spinal cord injury. Cell Cycle 11(17):3175–3179. doi:10.4161/cc.21262
Chen HC, Fong TH, Hsu PW, Chiu WT (2013) Multifaceted effects of rapamycin on functional recovery after spinal cord injury in rats through autophagy promotion, anti-inflammation, and neuroprotection. J Surg Res 179(1):e203–210. doi:10.1016/j.jss.2012.02.023
Lang-Lazdunski L, Blondeau N, Jarretou G, Lazdunski M, Heurteaux C (2003) Linolenic acid prevents neuronal cell death and paraplegia after transient spinal cord ischemia in rats. J Vasc Surg 38(3):564–575
Basso DM, Fisher LC, Anderson AJ, Jakeman LB, McTigue DM, Popovich PG (2006) Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains. J Neurotrauma 23(5):635–659. doi:10.1089/neu.2006.23.635
Shea TB (1994) Technical report. An inexpensive densitometric analysis system using a Macintosh computer and a desktop scanner. Biotechniques 16(6):1126–1128
Bethea JR, Castro M, Keane RW, Lee TT, Dietrich WD, Yezierski RP (1998) Traumatic spinal cord injury induces nuclear factor-kappaB activation. J Neurosci 18(9):3251–3260
Esposito E, Paterniti I, Meli R, Bramanti P, Cuzzocrea S (2012) GW0742, a high-affinity PPAR-delta agonist, mediates protection in an organotypic model of spinal cord damage. Spine (Phila Pa 1976) 37(2):E73–78. doi:10.1097/BRS.0b013e3182276d88
Abe K, Matsuki N (2000) Measurement of cellular 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction activity and lactate dehydrogenase release using MTT. Neurosci Res 38(4):325–329
Paterniti I, Impellizzeri D, Crupi R, Morabito R, Campolo M, Esposito E, Cuzzocrea S (2013) Molecular evidence for the involvement of PPAR-delta and PPAR-gamma in anti-inflammatory and neuroprotective activities of palmitoylethanolamide after spinal cord trauma. J Neuroinflammation 10:20. doi:10.1186/1742-2094-10-20
Kroemer G, Marino G, Levine B (2010) Autophagy and the integrated stress response. Mol Cell 40(2):280–293. doi:10.1016/j.molcel.2010.09.023
Saitoh T, Akira S (2010) Regulation of innate immune responses by autophagy-related proteins. J Cell Biol 189(6):925–935. doi:10.1083/jcb.201002021
Lapaquette P, Guzzo J, Bretillon L, Bringer MA (2015) Cellular and molecular connections between autophagy and inflammation. Mediators Inflamm 2015:398483. doi:10.1155/2015/398483
Xu J, Kim GM, Chen S, Yan P, Ahmed SH, Ku G, Beckman JS, Xu XM et al (2001) iNOS and nitrotyrosine expression after spinal cord injury. J Neurotrauma 18(5):523–532. doi:10.1089/089771501300227323
Bal-Price A, Brown GC (2001) Inflammatory neurodegeneration mediated by nitric oxide from activated glia-inhibiting neuronal respiration, causing glutamate release and excitotoxicity. J Neurosci 21(17):6480–6491
Hausmann ON (2003) Post-traumatic inflammation following spinal cord injury. Spinal Cord 41(7):369–378. doi:10.1038/sj.sc.3101483
Esposito E, Cuzzocrea S (2011) Anti-TNF therapy in the injured spinal cord. Trends Pharmacol Sci 32(2):107–115. doi:10.1016/j.tips.2010.11.009
Nesic O, Xu GY, McAdoo D, High KW, Hulsebosch C, Perez-Pol R (2001) IL-1 receptor antagonist prevents apoptosis and caspase-3 activation after spinal cord injury. J Neurotrauma 18(9):947–956. doi:10.1089/089771501750451857
Dong H, Fazzaro A, Xiang C, Korsmeyer SJ, Jacquin MF, McDonald JW (2003) Enhanced oligodendrocyte survival after spinal cord injury in Bax-deficient mice and mice with delayed Wallerian degeneration. J Neurosci 23(25):8682–8691
Ackery A, Robins S, Fehlings MG (2006) Inhibition of Fas-mediated apoptosis through administration of soluble Fas receptor improves functional outcome and reduces posttraumatic axonal degeneration after acute spinal cord injury. J Neurotrauma 23(5):604–616. doi:10.1089/neu.2006.23.604
Acknowledgments
The authors would like to thank Maria Antonietta Medici for her excellent technical assistance during this study and Mr Francesco Soraci for his secretarial and administrative assistance and Miss Valentina Malvagni for her editorial assistance with the manuscript.
This work was supported by L’Oréal award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was permitted by the University of Messina Review Board for the care of animals. All animal experiments complied with regulations in Italy (D.M. 116192) as well as the EU regulations (O.J. of E.C. L 358/1 12/18/1986).
Competing Interests
The authors declare that they have no competing interests.
Additional information
Marika Cordaro and Irene Paterniti contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cordaro, M., Paterniti, I., Siracusa, R. et al. KU0063794, a Dual mTORC1 and mTORC2 Inhibitor, Reduces Neural Tissue Damage and Locomotor Impairment After Spinal Cord Injury in Mice. Mol Neurobiol 54, 2415–2427 (2017). https://doi.org/10.1007/s12035-016-9827-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-9827-0