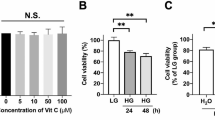
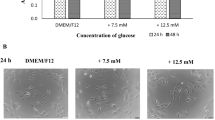
Abstract
Diabetic retinopathy (DR) is a major cause of acquired blindness among working adults. The retinal pigment epithelium (RPE), constitutes an outer blood-retinal barrier, is vastly affected in diabetic humans and animals. Lower levels of lutein in the serum and retina of diabetic population, and beneficial effects of carotenoids supplementation in diabetic retinopathy patients created an interest to examine the protective effect of lutein on hyperglycemia-mediated changes in oxidative stress and antioxidant defense system in ARPE-19 cells. The WST-1 assay was performed to analyze the impact of glucose, and lutein on the viability of ARPE-19. The intracellular oxidative stress was measured by a DCF (dichlorofluorescein) assay, mitochondrial membrane potential (MMP) was monitored using a JC-10 MMP assay kit and GSH level was examined using GSH/GSSG ratio detection kit. The oxidative stress markers, protein carbonyl and malondialdehyde were spectrophotometrically measured using 2,4-dinitrophenylhydrazine and 2-thiobarbituric acid, respectively. The expression of endogenous antioxidant enzymes and regulatory proteins in ARPE-19 was quantified by western blotting. The localization of Nrf2 protein was examined by immunofluorescent staining. The results show that lutein (up to 1.0 μM) did not affect the viability of ARPE-19 grown in both normal and high-glucose conditions. Lutein treatment blocked high glucose-mediated elevation of intracellular ROS, protein carbonyl and malondialdehyde content in ARPE-19 cells. The decreased MMP and GSH levels observed in ARPE-19 grown under high-glucose condition were rescued by lutein treatment. Further, lutein protected high glucose-mediated down-regulation of a redox-sensitive transcription factor, Nrf2, and antioxidant enzymes, SOD2, HO-1, and catalase. This protective effect of lutein was linked with activated nuclear translocation of Nrf2, which was associated with increased activation of regulatory proteins such as Erk and AKT. Our study indicates that improving the concentration of lutein in the retina could protect RPE from diabetes-associated damage.








Similar content being viewed by others
References
Bai Y, Cui W, Xin Y, Miao X, Barati MT, Zhang C, Chen Q, Tan Y, Cui T, Zheng Y, Cai L (2013) Prevention by sulforaphane of diabetic cardiomyopathy is associated with up-regulation of Nrf2 expression and transcription activation. J Mol Cell Cardiol 57:82–95. https://doi.org/10.1016/j.yjmcc.2013.01.008
Ben-Dor A, Steiner M, Gheber L, Danilenko M, Dubi N, Linnewiel K, Zick A, Sharoni Y, Levy J (2005) Carotenoids activate the antioxidant response element transcription system. Mol Cancer Ther 4:177–186
Bernstein PS, Law WC, Rando RR (1987) Isomerization of all-trans-retinoids to 11-cis-retinoids in vitro. Proc Natl Acad Sci U S A 84:1849–1853. https://doi.org/10.1073/pnas.84.7.1849
Bone RA, Landrum JT, Guerra LH, Ruiz CA (2003) Lutein and zeaxanthin dietary supplements raise macular pigment density and serum concentrations of these carotenoids in humans. J Nutr 133:992–998
Boulton M, Dayhaw-Barker P (2001) The role of the retinal pigment epithelium: topographical variation and ageing changes. Eye 15:384–389. https://doi.org/10.1038/eye.2001.141
Cai J, Nelson KC, Wu M, Sternberg P, Jones DP (2000) Oxidative damage and protection of the RPE. Prog Retin Eye Res 19:205–221
Choe E, Min DB (2006) Chemistry and reactions of reactive oxygen species in foods. Crit Rev Food Sci Nutr 46:1–22. https://doi.org/10.1080/10408390500455474
Coyne T, Ibiebele TI, Baade PD, Dobson A, McClintock C, Dunn S, Leonard D, Shaw J (2005) Diabetes mellitus and serum carotenoids: findings of a population-based study in Queensland, Australia. Am J Clin Nutr 82:685–693. https://doi.org/10.1093/ajcn/82.3.685
Dalle-Donne I, Rossi R, Giustarini D, Milzani A, Colombo R (2003) Protein carbonyl groups as biomarkers of oxidative stress. Clin Chim Acta 329:23–38
Das BN, Kim YW, Keum YS (2013, 2013) Mechanisms of Nrf2/keap1-dependent phase II cytoprotective and detoxifying gene expression and potential cellular targets of chemopreventive isothiocyanates. Oxidative Med Cell Longev. https://doi.org/10.1155/2013/839409
David JA, Rifkin WJ, Rabbani PS, Ceradini DJ (2017) The Nrf2/Keap1/ARE pathway and oxidative stress as a therapeutic target in type II diabetes mellitus. J Diabetes Res 2017:1–15. https://doi.org/10.1155/2017/4826724
Decanini A, Karunadharma PR, Nordgaard CL, Feng X, Olsen TW, Ferrington DA (2008) Human retinal pigment epithelium proteome changes in early diabetes. Diabetologia 51:1051–1061. https://doi.org/10.1007/s00125-008-0991-2
Desjardins DM, Yates PW, Dahrouj M, Liu Y, Crosson CE, Ablonczy Z (2016) Progressive early breakdown of retinal pigment epithelium function in hyperglycemic rats. Investig Opthalmol Vis Sci 57:2706–2713. https://doi.org/10.1167/iovs.15-18397
Dieter BP (2015) Dysregulation of Nrf2 signaling in diabetes: an opportunity for a multitarget approach. J Diabetes Metab 06:1–12. https://doi.org/10.4172/2155-6156.1000475
Filomeni G, Piccirillo S, Rotilio G, Ciriolo MR (2012) P38 MAPK and ERK1/2 dictate cell death/survival response to different pro-oxidant stimuli via p53 and Nrf2 in neuroblastoma cells SH-SY5Y. Biochem Pharmacol 83:1349–1357. https://doi.org/10.1016/j.bcp.2012.02.003
Frede K, Ebert F, Kipp AP, Schwerdtle T, Baldermann S (2017) Lutein activates the transcription factor Nrf2 in human retinal pigment epithelial cells. J Agric Food Chem 65:5944–5952. https://doi.org/10.1021/acs.jafc.7b01929
Garg TK, Chang JY (2004) 15-deoxy-delta 12,14-prostaglandin J2 prevents reactive oxygen species generation and mitochondrial membrane depolarization induced by oxidative stress. BMC Pharmacol 4:6. https://doi.org/10.1186/1471-2210-4-6
Goodwin T (1980) Functions of carotenoids. Biochem Carotenoids 1:77–95
Hamann S (2002) Molecular mechanisms of water transport in the eye. Int Rev Cytol 215:395–431
Hu B-J, Hu Y-N, Lin S, Ma W-J, Li X-R (2011) Application of lutein and Zeaxanthin in nonproliferative diabetic retinopathy. Int J Ophthalmol 4:303–306. https://doi.org/10.3980/j.issn.2222-3959.2011.03.19
Ishida K, Panjwani N, Cao Z, Streilein JW (2003) Participation of pigment epithelium in ocular immune privilege. 3. Epithelia cultured from iris, ciliary body, and retina suppress T-cell activation by partially non-overlapping mechanisms. Ocul Immunol Inflamm 11:91–105. https://doi.org/10.1076/ocii.11.2.91.15914
Jiménez-Osorio AS, Picazo A, González-Reyes S, Barrera-Oviedo D, Rodríguez-Arellano ME, Pedraza-Chaverri J (2014) Nrf2 and redox status in prediabetic and diabetic patients. Int J Mol Sci 15:20290–20305. https://doi.org/10.3390/ijms151120290
Johnson J, Maher P, Hanneken A (2009) The flavonoid, eriodictyol, induces long-term protection in arpe-19 cells through its effects on Nrf2 activation and phase 2 gene expression. Investig Ophthalmol Vis Sci 50:2398–2406. https://doi.org/10.1167/iovs.08-2088
Kamoshita M, Toda E, Osada H, Narimatsu T, Kobayashi S, Tsubota K, Ozawa Y (2016) Lutein acts via multiple antioxidant pathways in the photo-stressed retina. Sci Rep 6:–10. https://doi.org/10.1038/srep30226
Kannan R, Zhang N, Sreekumar PG, Spee CK, Rodriguez A, Barron E, Hinton DR (2006) Stimulation of apical and basolateral VEGF-A and VEGF-C secretion by oxidative stress in polarized retinal pigment epithelial cells. Mol Vis 12:1649–1659
Kavalappa YP, Udayawara Rudresh D, Gopal SS, Haranahalli Shivarudrappa A, Stephen NM, Rangiah K, Ponesakki G (2019) β-Carotene isolated from the marine red alga, Gracillaria sp. potently attenuates the growth of human hepatocellular carcinoma (HepG2) cells by modulating multiple molecular pathways. J Funct Foods 52:165–176. https://doi.org/10.1016/j.jff.2018.11.015
Koushan K, Rusovici R, Li W, Ferguson LR, Chalam KV (2013) The role of lutein in eye-related disease. Nutrients 5:1823–1839. https://doi.org/10.3390/nu5051823
Kowluru RA, Mishra M (2015) Oxidative stress, mitochondrial damage and diabetic retinopathy. Biochim Biophys Acta Mol basis Dis 1852:2474–2483
Li Z, Dong X, Liu H, Chen X, Shi H, Fan Y, Hou D, Zhang X (2013) Astaxanthin protects ARPE-19 cells from oxidative stress via upregulation of Nrf2-regulated phase II enzymes through activation of PI3K/AKT. Mol Vis 19:1656–1666
Liu H, Liu W, Zhou X, Long C, Kuang X, Hu J, Tang Y, Liu L, He J, Huang Z, Fan Y, Jin G, Zhang Q, Shen H (2017) Protective effect of lutein on ARPE-19 cells upon H2O2-induced G2/M arrest. Mol Med Rep 16:2069–2074. https://doi.org/10.3892/mmr.2017.6838
Lu L, Hackett SF, Mincey A, Lai H, Campochiaro PA (2006) Effects of different types of oxidative stress in RPE cells. J Cell Physiol 206:119–125. https://doi.org/10.1002/jcp.20439
Madsen-Bouterse SA, Kowluru RA (2008) Oxidative stress and diabetic retinopathy: pathophysiological mechanisms and treatment perspectives. Rev Endocr Metab Disord 9:315–327
Maminishkis A, Chen S, Jalickee S, Banzon T, Shi G, Wang FE, Ehalt T, Hammer JA, Miller SS (2006) Confluent monolayers of cultured human fetal retinal pigment epithelium exhibit morphology and physiology of native tissue. Investig Ophthalmol Vis Sci 47:3612–3624. https://doi.org/10.1167/iovs.05-1622
Matzinger M, Fischhuber K, Heiss EH (2018) Activation of Nrf2 signaling by natural products-can it alleviate diabetes? Biotechnol Adv 36:1738–1767
Min KJ, Lee JT, Hye JE, Kwon TK (2011) An IκBα phosphorylation inhibitor induces heme oxygenase-1(HO-1) expression through the activation of reactive oxygen species (ROS)-Nrf2-ARE signaling and ROS-PI3K/AKT signaling in an NF-κB-independent mechanism. Cell Signal 23:1505–1513. https://doi.org/10.1016/j.cellsig.2011.05.013
Moschos MM, Dettoraki M, Tsatsos M, Kitsos G, Kalogeropoulos C (2017) Effect of carotenoids dietary supplementation on macular function in diabetic patients. Eye Vis 4:23. https://doi.org/10.1186/s40662-017-0088-4
Ndisang JF, Jadhav A, Mishra M (2014) The heme oxygenase system suppresses perirenal visceral adiposity, abates renal inflammation and ameliorates diabetic nephropathy in zucker diabetic fatty rats. PLoS One 9:e87936. https://doi.org/10.1371/journal.pone.0087936
Nguyen T, Sherratt PJ, Huang H-C, Yang CS, Pickett CB (2003) Increased protein stability as a mechanism that enhances Nrf2-mediated transcriptional activation of the antioxidant response element. Degradation of Nrf2 by the 26 S proteasome. J Biol Chem 278:4536–4541. https://doi.org/10.1074/jbc.M207293200
Nishimoto S, Koike S, Inoue N, Suzuki T, Ogasawara Y (2017) Activation of Nrf2 attenuates carbonyl stress induced by methylglyoxal in human neuroblastoma cells: increase in GSH levels is a critical event for the detoxification mechanism. Biochem Biophys Res Commun 483:874–879. https://doi.org/10.1016/j.bbrc.2017.01.024
Nita M, Grzybowski A (2016) The role of the reactive oxygen species and oxidative stress in the Pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxidative Med Cell Longev 2016:1–23. https://doi.org/10.1155/2016/3164734
Pitha-Rowe I, Liby K, Royce D, Sporn M (2009) Synthetic Triterpenoids attenuate cytotoxic retinal injury: cross-talk between Nrf2 and PI3K/AKT signaling through inhibition of the lipid phosphatase PTEN. Investig Opthalmology Vis Sci 50:5339. https://doi.org/10.1167/iovs.09-3648
Prabhakar PV, Reddy UA, Singh SP, Balasubramanyam A, Rahman MF, Indu Kumari S, Agawane SB, Murty USN, Grover P, Mahboob M (2012) Oxidative stress induced by aluminum oxide nanomaterials after acute oral treatment in Wistar rats. J Appl Toxicol 32:436–445. https://doi.org/10.1002/jat.1775
Samuels IS, Bell BA, Pereira A, Saxon J, Peachey NS (2014) Early retinal pigment epithelium dysfunction is concomitant with hyperglycemia in mouse models of type 1 and type 2 diabetes. J Neurophysiol 113:1085–1099. https://doi.org/10.1152/jn.00761.2014
Santos JM, Mishra M, Kowluru RA (2014) Posttranslational modification of mitochondrial transcription factor A in impaired mitochondria biogenesis: implications in diabetic retinopathy and metabolic memory phenomenon. Exp Eye Res 121:168–177. https://doi.org/10.1016/J.EXER.2014.02.010
Shi X, Zhou B (2010) The role of Nrf2 and MAPK pathways in PFOS-induced oxidative stress in zebrafish embryos. Toxicol Sci 115:391–400. https://doi.org/10.1093/toxsci/kfq066
Shivarudrappa A, Gopal SS, Ponesakki G (2019) An in vitro protocol to study the effect of hyperglycemia on intracellular redox signaling in human retinal pigment epithelial (ARPE-19) cells. Mol Biol Rep 46:1263–1274
Siewert S, González I, Santillán L, Lucero R, Ojeda MS, Gimenez MS (2013) Downregulation of Nrf2 and HO-1 expression contributes to oxidative stress in type 2 diabetes mellitus: a study in Juana Koslay City, San Luis, Argentina. J Diabetes Mellit 03:71–78. https://doi.org/10.4236/jdm.2013.32011
Sowmya Shree G, Yogendra Prasad K, Arpitha HS, Deepika UR, Nawneet Kumar K, Mondal P, Ganesan P (2017) β-Carotene at physiologically attainable concentration induces apoptosis and down-regulates cell survival and antioxidant markers in human breast cancer (MCF-7) cells. Mol Cell Biochem 436:1–12. https://doi.org/10.1007/s11010-017-3071-4
Sparrow JR, Fishkin N, Zhou J, Cai B, Jang YP, Krane S, Itagaki Y, Nakanishi K (2003) A2E, a byproduct of the visual cycle. Vis Res 43:2983–2990. https://doi.org/10.1016/S0042-6989(03)00475-9
Sugasawa K, Deguchi J, Okami T, Yamamoto A, Omori K, Uyama M, Tashiro Y (1994) Immunocytochemical analyses of distributions of Na, K-ATPase and GLUT1, insulin and transferrin receptors in the developing retinal pigment epithelial cells. Cell Struct Funct 19:21–28. https://doi.org/10.1247/csf.19.21
Thomas SE, Harrison EH (2016) Mechanisms of selective delivery of xanthophylls to retinal pigment epithelial cells by human lipoproteins. J Lipid Res 57:1865–1878. https://doi.org/10.1194/jlr.M070193
van Reyk DM, Gillies MC, Davies MJ (2003) The retina: oxidative stress and diabetes. Redox Rep 8:187–192. https://doi.org/10.1179/135100003225002673
Varì R, D’Archivio M, Filesi C, Carotenuto S, Scazzocchio B, Santangelo C, Giovannini C, Masella R (2011) Protocatechuic acid induces antioxidant/detoxifying enzyme expression through JNK-mediated Nrf2 activation in murine macrophages. J Nutr Biochem 22:409–417. https://doi.org/10.1016/j.jnutbio.2010.03.008
Vinores SA, Derevjanik NL, Ozaki H, Okamoto N, Campochiaro PA (2000) Cellular mechanisms of blood-retinal barrier dysfunction in macular edema. In: Macular Edema. Springer Netherlands, Dordrecht, pp 13–24
Wang J, Fields J, Zhao C, Langer J, Thimmulappa RK, Kensler TW, Yamamoto M, Biswal S, Doré S (2007) Role of Nrf2 in protection against intracerebral hemorrhage injury in mice. Free Radic Biol Med 43:408–414. https://doi.org/10.1016/j.freeradbiomed.2007.04.020
Wang L, Chen Y, Sternberg P, Cai J (2008) Essential roles of the PI3 kinase/AKT pathway in regulating Nrf2-dependent antioxidant functions in the RPE. Investig Opthalmology Vis Sci 49:1671–1678. https://doi.org/10.1167/iovs.07-1099
Wang J, Xu E, Elliott MH, Zhu M, Le YZ (2010) Müller cell-derived VEGF is essential for diabetes-induced retinal inflammation and vascular leakage. Diabetes 59:2297–2305. https://doi.org/10.2337/db09-1420
Whitehead AJ, Mares JA, Danis RP (2006) Macular pigment: a review of current knowledge. Arch Ophthalmol 124:1038–1045
Xu HZ, Le YZ (2011) Significance of outer blood-retina barrier breakdown in diabetes and ischemia. Investig Ophthalmol Vis Sci 52:2160–2164. https://doi.org/10.1167/iovs.10-6518
Young RW, Bok D (1969) Participation of the retinal pigment epithelium in the rod outer segment renewal process. J Cell Biol 42:392–403. https://doi.org/10.1083/jcb.42.2.392
Yu W, Simmons-Menchaca M, Gapor A, Sanders BG, Kline K (1999) Induction of apoptosis in human breast cancer cells by tocopherols and tocotrienols. Nutr Cancer 33:26–32. https://doi.org/10.1080/01635589909514744
Yu R, Mandlekar S, Lei W, Fahl WE, Tan TH, Kong AN (2000) p38 mitogen-activated protein kinase negatively regulates the induction of phase II drug-metabolizing enzymes that detoxify carcinogens. J Biol Chem 275:2322–2327. https://doi.org/10.1074/JBC.275.4.2322
Zhong Q, Kowluru RA (2011) Epigenetic changes in mitochondrial superoxide dismutase in the retina and the development of diabetic retinopathy. Diabetes 60:1304–1313. https://doi.org/10.2337/db10-0133
Zou X, Gao J, Zheng Y, Wang X, Chen C, Cao K, Xu J, Li Y, Lu W, Liu J, Feng Z (2014) Zeaxanthin induces Nrf2-mediated phase II enzymes in protection of cell death. Cell Death Dis 5:e1218–e1218. https://doi.org/10.1038/cddis.2014.190
Acknowledgments
The author, Arpitha H S, acknowledges the University Grants Commission (UGC), New Delhi, India for granting Research Fellowship. The authors thank the Director, CSIR-CFTRI for the constant support.
Funding
We would like to thank the Council for Scientific and Industrial Research (CSIR), New Delhi for granting financial support under the 12th Five Year Plan Project (BSC-0404).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shivarudrappa, A.H., Ponesakki, G. Lutein reverses hyperglycemia-mediated blockage of Nrf2 translocation by modulating the activation of intracellular protein kinases in retinal pigment epithelial (ARPE-19) cells. J. Cell Commun. Signal. 14, 207–221 (2020). https://doi.org/10.1007/s12079-019-00539-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-019-00539-1