Abstract
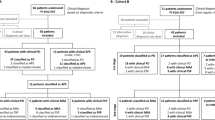
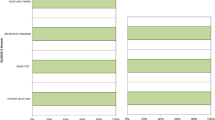
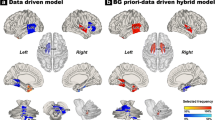
Age and gender are the important factors for brain metabolic declines in both normal aging and neurodegeneration, and the confounding effects may influence early and differential diagnosis of neurodegenerative diseases based on the [18F]fluorodeoxyglucose positron emission tomography ([18F]FDG PET). We aimed to explore the potential of the adjustment of age- and gender-related confounding factors on [18F]FDG PET images in differentiation of Parkinson’s disease (PD), multiple system atrophy (MSA) and progressive supra-nuclear palsy (PSP). Eight hundred and seventy-seven clinically definitely diagnosed Parkinsonian patients from a benchmark Huashan Parkinsonian PET imaging database were included. An age- and gender-adjusted Z (AGAZ) score was established based on the gender-specific longitudinal metabolic changes on healthy subjects. AGAZ scores and standardized uptake value ratio (SUVR) values were quantified at regional-level and support vector machine-based error-correcting output codes method was applied for classification. Additional references of the classifications based on metabolic pattern scores were included. The feature-based AGAZ score showed the best performance in classification (accuracy for PD, MSA, PSP: 93.1%, 96.3%, 94.8%). In both genders, the AGAZ score consistently achieved the best efficiency, and the improvements compared to the conventional SUVR value for PD, MSA, and PSP mainly laid in specificity (Male: 5.7%; Female: 11.1%), sensitivity (Male: 7.2%; Female: 7.3%), and sensitivity (Male: 7.3%; Female: 17.2%). Female patients benefited more from the adjustment on [18F]FDG PET in MSA and PSP groups (absolute net reclassification index, p < 0.001). Collectively, the adjustment of age- and gender-related confounding factors may improve the differential diagnosis of Parkinsonism. Particularly, the diagnosis of female Parkinsonian population has the best improvement from this correction.








Similar content being viewed by others
Availability of Data and Material
All data included in this study will be available to the scientific community upon completion of the non-disclosure agreement (NDA) with the corresponding authors according to international data protection regulations.
References
Alba AC, Agoritsas T, Walsh M, Hanna S, Iorio A, Devereaux PJ et al (2017) Discrimination and calibration of clinical prediction models: Users’ guides to the medical literature. JAMA 318(14):1377–1384. https://doi.org/10.1001/jama.2017.12126
Baba Y, Putzke JD, Whaley NR, Wszolek ZK, Uitti RJ (2006) Progressive supranuclear palsy: phenotypic sex differences in a clinical cohort. Mov Disord 21(5):689–692. https://doi.org/10.1002/mds.20769
Bonte S, Vandemaele P, Verleden S, Audenaert K, Deblaere K, Goethals I et al (2017) Healthy brain ageing assessed with 18F-FDG PET and age-dependent recovery factors after partial volume effect correction. Eur J Nucl Med Mol Imaging 44(5):838–849. https://doi.org/10.1007/s00259-016-3569-0
Boxer AL, Yu JT, Golbe LI, Litvan I, Lang AE, Höglinger GU (2017) Advances in progressive supranuclear palsy: new diagnostic criteria, biomarkers, and therapeutic approaches. Lancet Neurol 16(7):552–563. https://doi.org/10.1016/S1474-4422(17)30157-6
Brugnolo A, Morbelli S, Arnaldi D, De Carli F, Accardo J, Bossert I et al (2014) Metabolic correlates of rey auditory verbal learning test in elderly subjects with memory complaints. J Alzheimer’s Dis 39(1):103–113. https://doi.org/10.3233/JAD-121684
Cavedo E, Chiesa PA, Houot M, Ferretti MT, Grothe MJ, Teipel SJ et al (2018) Sex differences in functional and molecular neuroimaging biomarkers of Alzheimer’s disease in cognitively normal older adults with subjective memory complaints. Alzheimer’s Dement 14(9):1204–1215. https://doi.org/10.1016/j.jalz.2018.05.014
Cerri S, Mus L, Blandini F (2019) Parkinson’s disease in women and men: what’s the difference? J Parkinsons Dis 9(3):501–515. https://doi.org/10.3233/JPD-191683
Chmielnicki W, Stąkapor K (2011) A New Approach to Multi-class SVM-Based Classification Using Error Correcting Output Codes. In: Burduk, R., Kurzyński, M., Woźniak, M., Żołnierek, A. (eds) Computer Recognition Systems 4. Advances in Intelligent and Soft Computing, vol 95. Springer, Berlin, Heidelberg, pp 499–506. https://doi.org/10.1007/978-3-642-20320-6_52
Cosgrove KP, Mazure CM, Staley JK (2007) Evolving knowledge of sex differences in brain structure, function, and chemistry. Biol Psychiatry 62(8):847–855. https://doi.org/10.1016/j.biopsych.2007.03.001
De Micco R, Esposito F, di Nardo F, Caiazzo G, Siciliano M, Russo A et al (2019) Sex-related pattern of intrinsic brain connectivity in drug-naïve Parkinson’s disease patients. Mov Disord 34(7):997–1005. https://doi.org/10.1002/mds.27725
Dukart J, Schroeter ML, Mueller K (2011) Age Correction in dementia—matching to a healthy brain. PLoS ONE 6(7):e22193. https://doi.org/10.1371/journal.pone.0022193
Ewers M, Brendel M, Rizk-Jackson A, Rominger A, Bartenstein P, Schuff N et al (2014) Reduced FDG-PET brain metabolism and executive function predict clinical progression in elderly healthy subjects. NeuroImage Clin 4:45–52. https://doi.org/10.1016/j.nicl.2013.10.018
Ferretti MT, Iulita MF, Cavedo E, Chiesa PA, Dimech AS, Chadha AS et al (2018) Sex differences in Alzheimer disease—the gateway to precision medicine. Nat Rev Neurol 14(8):457–469. https://doi.org/10.1038/s41582-018-0032-9
Franke K, Ziegler G, Klöppel S, Gaser C (2010) Estimating the age of healthy subjects from T1-weighted MRI scans using kernel methods: exploring the influence of various parameters. Neuroimage 50(3):883–892. https://doi.org/10.1016/j.neuroimage.2010.01.005
Ge J, Wu J, Peng S, Wu P, Wang J, Zhang H et al (2018) Reproducible network and regional topographies of abnormal glucose metabolism associated with progressive supranuclear palsy: multivariate and univariate analyses in American and Chinese patient cohorts. Hum Brain Mapp 39(7):2842–2858. https://doi.org/10.1002/hbm.24044
Gillies GE, Pienaar IS, Vohra S, Qamhawi Z (2014) Sex differences in Parkinson’s disease. Front Neuroendocrinol 35(3):370–384. https://doi.org/10.1016/j.yfrne.2014.02.002
Good CD, Johnsrude IS, Ashburner J, Henson RNA, Friston KJ, Frackowiak RSJ (2001) A voxel-based morphometric study of ageing in 465 normal adult human brains. Neuroimage 14(1 Pt 1):21–36. https://doi.org/10.1006/nimg.2001.0786
Goyal MS, Blazey TM, Su Y, Couture LE, Durbin TJ, Bateman RJ et al (2019) Persistent metabolic youth in the aging female brain. Proc Natl Acad Sci USA 116(8):3251–3255. https://doi.org/10.1073/pnas.1815917116
Greve DN, Svarer C, Fisher PM, Feng L, Hansen AE, Baare W et al (2014) Cortical surface-based analysis reduces bias and variance in kinetic modeling of brain PET data. Neuroimage 92:225–236. https://doi.org/10.1016/j.neuroimage.2013.12.021
Haaxma CA, Bloem BR, Borm GF, Oyen WJG, Leenders KL, Eshuis S et al (2007) Gender differences in Parkinson’s disease. J Neurol Neurosurg Psychiatry 78(8):819–824. https://doi.org/10.1136/jnnp.2006.103788
Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL et al (2019) Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol 15(10):565–581. https://doi.org/10.1038/s41582-019-0244-7
Ishibashi K, Onishi A, Fujiwara Y, Oda K, Ishiwata K, Ishii K (2018) Longitudinal effects of aging on 18F-FDG distribution in cognitively normal elderly individuals. Sci Rep 8(1):11557. https://doi.org/10.1038/s41598-018-29937-y
Jiang J, Sun Y, Zhou H, Li S, Huang Z, Wu P et al (2018) Study of the influence of age in 18F-FDG PET images using a data-driven approach and its evaluation in Alzheimer’s disease. Contrast Media Mol Imaging 2018:3786083. https://doi.org/10.1155/2018/3786083
Kaczkurkin AN, Raznahan A, Satterthwaite TD (2019) Sex differences in the developing brain: insights from multimodal neuroimaging. Neuropsychopharmacology 44(1):71–85. https://doi.org/10.1038/s41386-018-0111-z
Kakimoto A, Ito S, Okada H, Nishizawa S, Minoshima S, Ouchi Y (2016) Age-related sex-specific changes in brain metabolism and morphology. J Nucl Med 57(2):221–225. https://doi.org/10.2967/jnumed.115.166439
Kato T, Inui Y, Nakamura A, Ito K (2016) Brain fluorodeoxyglucose (FDG) PET in dementia. Ageing Res Rev 30:73–84. https://doi.org/10.1016/j.arr.2016.02.003
Knopman DS, Jack CR, Wiste HJ, Lundt ES, Weigand SD, Vemuri P et al (2014) 18F-fluorodeoxyglucose positron emission tomography, aging, and apolipoprotein E genotype in cognitively normal persons. Neurobiol Aging 35(9):2096–2106. https://doi.org/10.1016/j.neurobiolaging.2014.03.006
Li B, Jang I, Riphagen J, Almaktoum R, Yochim KM, Ances BM et al (2021) Identifying individuals with Alzheimer’s disease-like brains based on structural imaging in the Human Connectome Project Aging cohort. Hum Brain Mapp 42(17):5535–5546. https://doi.org/10.1002/hbm.25626
Li TR, Dong QY, Jiang XY, Kang GX, Li X, Xie YY et al (2022) Exploring brain glucose metabolic patterns in cognitively normal adults at risk of Alzheimer’s disease: a cross-validation study with Chinese and ADNI cohorts. NeuroImage Clin 33:102900. https://doi.org/10.1016/j.nicl.2021.102900
Meles SK, Teune LK, De Jong BM, Dierckx RA, Leenders KL (2017) Metabolic imaging in Parkinson disease. J Nucl Med 58(1):23–28. https://doi.org/10.2967/jnumed.116.183152
Meyer PT, Frings L, Rücker G, Hellwig S (2017) 18F-FDG PET in Parkinsonism: differential diagnosis and evaluation of cognitive impairment. J Nucl Med 58(12):1888–1898. https://doi.org/10.2967/jnumed.116.186403
Mosconi L, Berti V, Quinn C, McHugh P, Petrongolo G, Varsavsky I et al (2017) Sex differences in Alzheimer risk: brain imaging of endocrine vs chronologic aging. Neurology 89(13):1382–1390. https://doi.org/10.1212/WNL.0000000000004425
Nobili F, Arbizu J, Bouwman F, Drzezga A, Agosta F, Nestor P et al (2018) European Association of Nuclear Medicine and European Academy of Neurology recommendations for the use of brain 18 F-fluorodeoxyglucose positron emission tomography in neurodegenerative cognitive impairment and dementia: Delphi consensus. Eur J Neurol 25(10):1201–1217. https://doi.org/10.1111/ene.13728
Petit-Taboue MC, Landeau B, Desson JF, Dary M, Baron JC (1998) Effects of healthy aging on the regional cerebral metabolic rate of glucose assessed with statistical parametric mapping. Neuroimage 7(3):176–184. https://doi.org/10.1006/nimg.1997.0318
Shen B, Wei S, Ge J, Peng S, Liu F, Li L et al (2020) Reproducible metabolic topographies associated with multiple system atrophy: network and regional analyses in Chinese and American patient cohorts. NeuroImage Clin 28:102416. https://doi.org/10.1016/j.nicl.2020.102416
Takahashi R, Ishii K, Kakigi T, Yokoyama K (2011) Gender and age differences in normal adult human brain: voxel-based morphometric study. Hum Brain Mapp 32(7):1050–1058. https://doi.org/10.1002/hbm.21088
Tang CC, Poston KL, Eckert T, Feigin A, Frucht S, Gudesblatt M et al (2010) Differential diagnosis of parkinsonism: a metabolic imaging study using pattern analysis. Lancet Neurol 9(2):149–158. https://doi.org/10.1016/S1474-4422(10)70002-8
Tremblay C, Abbasi N, Zeighami Y, Yau Y, Dadar M, Rahayel S et al (2020) Sex effects on brain structure in de novo Parkinson’s disease: a multimodal neuroimaging study. Brain 143(10):3052–3066. https://doi.org/10.1093/brain/awaa234
van Aalst J, Devrome M, Van Weehaeghe D, Rezaei A, Radwan A, Schramm G et al (2022) Regional glucose metabolic decreases with ageing are associated with microstructural white matter changes: a simultaneous PET/MR study. Eur J Nucl Med Mol Imaging 49(2):664–680. https://doi.org/10.1007/s00259-021-05518-6
Wu P, Wang J, Peng S, Ma Y, Zhang H, Guan Y et al (2013) Metabolic brain network in the Chinese patients with Parkinson’s disease based on 18F-FDG PET imaging. Parkinsonism Relat Disord 19(6):622–627. https://doi.org/10.1016/j.parkreldis.2013.02.013
Wu P, Zhao Y, Wu J, Brendel M, Lu J, Ge J et al (2022) Differential diagnosis of parkinsonism based on deep metabolic imaging indices. J Nucl Med. https://doi.org/10.2967/jnumed.121.263029
Yoshizawa H, Gazes Y, Stern Y, Miyata Y, Uchiyama S (2014) Characterizing the normative profile of 18F-FDG PET brain imaging: sex difference, aging effect, and cognitive reserve. Psychiatry Res 221(1):78–85. https://doi.org/10.1016/j.pscychresns.2013.10.009
Zhang H, Wu P, Ziegler SI, Guan Y, Wang Y, Ge J et al (2017) Data-driven identification of intensity normalization region based on longitudinal coherency of 18F-FDG metabolism in the healthy brain. Neuroimage 146:589–599. https://doi.org/10.1016/j.neuroimage.2016.09.031
Acknowledgements
The authors thank the patients and family members who participated in the research.
Funding
This work was supported by National Natural Science Foundation of China (81671239, 81361120393, 82171252, 81701250, 81401135, 81971641, 91949118, 81771372, 82021002), the Ministry of Science and Technology of China (2016YFC1306504), Shanghai Municipal Science and Technology Major Project (2017SHZDZX01, 2018SHZDZX03) and ZJ Lab, Shanghai Aging and Maternal and Child Health Research Special Project (2020YJZX0111), Clinical Research Plan of Shanghai Hospital Development Center (SHDC2020CR1038B), Science and Technology Innovation 2030 Major Projects (2022ZD0211600), Youth Medical Talents—Medical Imaging Practitioner Program by Shanghai Municipal Health Commission and Shanghai Medical and Health Development Foundation (SHWRS(2020)_087), the Swiss National Science Foundation (188350), and Jacques & Gloria Gossweiler Foundation and Siemens Healthineers.
Author information
Authors and Affiliations
Contributions
CZ, FL, and KS conceived and designed the clinical study; MW and KS conceived and developed the algorithms. MW conducted the computational experiments. JL, PW, and HZ collected the imaging data and inspected images together with IY, SZ, JJ, SF, MS, AR, and SCH. JW, FL, JL, and PW collected clinical data and defined the standards of subject inclusion and evaluations. MW, JL, and KS contributed to the analysis of the data. JL, MW, KS, CZ, and FL wrote the manuscript with the advice, input and proof of other co-authors.
Corresponding authors
Ethics declarations
Conflict of interest
Axel Rominger and Kuangyu Shi received research support from Novartis and Siemens Healthineers. Other authors report no commercial interests or potential conflicts of interest.
Ethics Approval
This study obtained ethics permission from the Institutional Review Board of Huashan Hospital. All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee, and the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to Participate
The written informed consent was obtained from subjects included in this study.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lu, J., Wang, M., Wu, P. et al. Adjustment for the Age- and Gender-Related Metabolic Changes Improves the Differential Diagnosis of Parkinsonism. Phenomics 3, 50–63 (2023). https://doi.org/10.1007/s43657-022-00079-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43657-022-00079-6