Summary
Metastasis is temperature dependent in the renal adenocarcinoma of the North American leopard frog, Rana pipiens. Widespread, multiple, metastatic colonies occur in tumor-bearing frogs kept at 28°C for 50 days while tumor-bearing frogs kept at 7°C for 98 days or more have either no secondary deposits or they have only an occasional small metastatic nodule. An attractive aspect of the frog tumor is that invasion and metastasis can be permitted or inhibited by the manipulation of temperature alone-no exogenous chemicals or drugs are required for the effect. Because of this, biological variables which reproducibly and specifically associate with metastasis permissive conditions when ambient temperature is cycled between permissive and inhibitory values are strong candidates for being causal elements in the multistep process leading to metastasis.
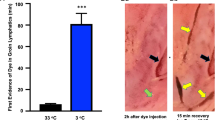
Intravascularly injected labelled renal tumor cells reached all organs studied in as little as 15 minutes at both metastasis restrictive and permissive temperature. The results with tumor cell inoculation dispose of the possibility that failure of metastasis in chilled animals is due to cold-induced changes in blood flow. Histologically typical metastatic colonies developed in frogs, kept at the permissive temperature, after injection with disaggregated tumor cells which were previously cryopreserved.
Frog tumors elaborate type I collagenase in a temperature dependent manner. Type IV collagenase has been demonstrated as well. Tumor cell detachment in vitro, assembly and disassembly of tumor cell cytoplasmic microtubules, and invasion in vitro, are all temperature dependent.
Similar content being viewed by others
References
Lucke B, Schlumberger H: Induction of metastasis of frog carcinoma by increase of environmental temperature. J Exp Med 89: 269–278, 1949.
Smallwood WM: Adrenal tumors in the kidney of the frog. Anat Anz 26: 652–658, 1905.
Downs AW: An epithelial tumor of the intestine of the frog, Nature 130: 778, 1932.
Murray JA: The zoological distribution of cancer. Imperial Cancer Research Fund, 3rd Scientific Report: 41–60, 1908.
Lucke B: A neoplastic disease of the frog, Rana pipiens. Am J Cancer 20: 352–379, 1934.
McKinnell RG: Incidence and histology of renal tumors of leopard frogs from the north central states. Ann NY Acad Sci 126: 85–98, 1965.
Tweedell KS: Simulated transmission of renal tumors in oocytes and embryos of Rana pipiens. In: Mizell M (ed) Biology of amphibian tumors. Springer-Verlag, New York, 1969, pp 229–239.
Rafferty KAJr: Kidney tumors of the leopard frog: A review. Cancer Res 24: 169–185, 1964.
Duryee WR: Precancer cells in amphibian adenocarcinoma. Ann NY Acad Sci 63: 1280–1302, 1956.
Duryee WR, Long ME, Taylor HCJr, McKelway WP, Ehrmann RL: Human and amphibian neoplasms compared. Science 131: 276–280, 1960.
Lucke B, Schlumberger H: Common neoplasms in fish, amphibians, and reptiles. J Tech Methods Bull Int Ass Med Museums 22: 4–17, 1942.
Lucke B: Carcinoma of the leopard frog: Its probable causation by a virus. J Exp Med 68: 457–468, 1938.
Lucke B: Kidney carcinoma of the leopard frog: A virus tumor. Ann NY Acad Sci 54: 1093–1109, 1952.
Fawcett DW: Electron microscope observations on intracellular virus-like particles associated with the cells of the Lucke renal adenocarcinoma. J Cell Sci 2: 725–741, 1956.
Zambernard J, Vatter AE: The fine structural cytochemistry of virus particles found in renal tumors of leopard frogs. I. An enzymatic study of the viral nucleoid. J Virol 28: 318–324, 1966.
Lunger PO: The isolation and morphology of the Lucke frog kidney tumor virus. J Virol 24: 138–145, 1964.
Zambernard J, McKinnell RG: ‘Virus-free’ renal tumors obtained from prehibernating leopard frogs of known geographic origin. Cancer Res 29: 653–657, 1969.
McKinnell RG, Zambernard J: Virus particles in renal tumors obtained from spring Rana pipiens of known geographic origin. Cancer Res 28: 684–688, 1968.
McKinnell RG, Ellis VL: Herpesviruses in tumors of postspawning Rana pipiens. Cancer Res 32: 1154–1159, 1972.
McKinnell RG, Ellis VL, Dapkus DC, Steven LMJr: Early replication of herpesviruses in naturally occurring frog tumors. Cancer Res 32: 1729–1732, 1972.
McKinnell RG, Ellis VL: Epidemiology of the frog renal tumour and the significance of tumour nuclear transplantation studies to a viral aetiology of the tumour — a review. Int Agency Res Cancer Sci Publ 2: 187–197.
McKinnell RG: The Lucke frog kidney tumor and its herpesvirus. Am Zool 13: 97–114, 1973.
McKinnell RG: The Lucke tumor: A neoplastic disease of frog kidneys. In: Hoff GL, Frye FL, Jacobson ER (eds) Diseases of amphibians and reptiles, Plenum, New York, 1984, pp 581–605.
Tweedell KS: Induced oncogenesis in developing frog kidney cells. Cancer Res 27: 2042–2052, 1967.
Tweedell KS, Wong WY: Frog kidney tumors induced by herpesvirus cultured in pronephric cells. J Nat Cancer Inst 52: 621–624, 1974.
Tweedell KS: Pronephric tumour cell lines from herpesvirus-transformed cells. Int Agency Res Cancer Sci Publ 24: II 609–616, 1978.
DiBerardino MA: The karyotype of Rana pipiens and investigation of its stability during embryonic differentiation. Dev Biol 5: 101–126, 1962.
McKinnell RG, Tweedell KS: Induction of renal tumors in triploid leopard frogs. J Nat Cancer Inst 44: 1161–1166, 1970.
Naegele RF, Granoff A, Darlington RW: The presence of the Lucke tumor herpesvirus genome in induced tadpole tumors and its oncogenicity: Koch-Henle postulates fulfilled. Proc Nat Acad Sci USA 71: 830–834, 1974.
Conant R: A field guide to reptiles and amphibians. 2nd edn. Houghton Mifflin, Boston, 1975.
Wright AH, Wright AA: Handbook of frogs and toads of the United States and Canada. Cornell Univ Press, Ithaca, 1949.
McKinnell RG: Lucke renal adenocarcinoma: Epidemiological aspects. In: Mizell M (ed) Biology of amphibian tumors. Springer-Verlag, New York, 1969, pp 254–260.
Nace GW, Richards CM: Development of biologically defined strains of amphibians. In: Mizell M (ed) Biology of amphibian tumors. Springer-Verlag, New York, 1969, pp 409–418.
Auclair W: Monolayer culture of Rana pipiens kidney and ecological factors. In: Duryee WR, Warner L (eds) Proceedings of frog kidney adenocarcinoma conference. National Institutes of Health, Bethesda, 1961, pp 107–113.
McKinnell RG, McKinnell BK: Seasonal fluctuation of frog renal adenocarcinoma prevalence in natural populations. Cancer Res 28: 440–444, 1968.
McKinnell RG, Gorham E, Martin FB, SchaadIV JW: Reduced prevalence of the Lucke renal adenocarcinoma in populations of Rana pipiens in Minnesota. J Nat Cancer Inst 63: 821–824, 1979.
McKinnell RG, Gorham E, Martin FB: Continued diminished prevalence of the Lucke renal adenocarcinoma in Minnesota leopard frogs. Am Midl Nat 104: 402–404, 1980.
McKinnell RG, Gorham E, Binger DG: Variation in the incidence of renal adenocarcinoma among Rana pipiens held under tumor-promoting conditions. Am Midl Nat 108: 111–116, 1982.
DiBerardino MA, King TJ, McKinnell RG: Chromosome studies of a frog renal adenocarcinoma line carried by serial intraocular transplantation. J Nat Cancer Inst 31: 769–789, 1963.
Rollins LA, McKinnell RG: The influence of glucocorticoids on survival and growth of allografted tumors in the anterior eye chamber of leopard frogs. Develop Comp Immunol 4: 283–294, 1980.
Schmid WD: Survival of frogs in low temperature. Science 215: 697–698, 1982.
Merrell DJ: Life history of the leopard frog, Rana pipiens, in Minnesota. Bell Museum Natural History, University of Minnesota, Occ Paper 15, 1977.
Haddow A, Blake I: Neoplasms in fish: A report of six cases with a summary of the literature. J Pathol Bacteriol 36: 41–47, 1933.
Lucke B: A neoplastic disease of the kidney of the frog, Rana pipiens. II. On the occurrence of metastasis. Am J Cancer 22: 326–334, 1934.
Schlumberger HG, Lucke B: Tumors of fishes, amphibians, and reptiles. Cancer Res 8: 657–754, 1948.
Willis RA: The spread of tumors in the human body. 3rd edn. Butterworths, London, 1973.
McKinnell RG, Cunningham WP: Herpesviruses in metastatic Lucke renal adenocarcinoma. Differentiation 22: 41–46, 1982.
Tarin D, McKinnell RG, Nace GW: Artifically induced metastasis by cells from spontaneous Lucke renal adenocarcinoma. Invasion Metastasis 4: 198–208, 1984.
Schwartz PE, Naftolin F: Type 2 herpes simplex virus and vulvar carcinoma in situ. N Engl J Med 305: 517–518, 1981.
Langille BL, Crisp B: Temperature dependence of blood viscosity in frogs and turtles: effect of heat exchange with environment. Am J Physiol 239: R248-R253, 1980.
Potter KM, Juacaba SF, Price JE, Tarin D: Observations on organ distribution of fluorescein-labelled tumours cells released intravascularly. Invasion Metastasis 3: 221–233, 1983.
McKinnell RG: The Lucke renal adenocarcinoma: Environmental influences on the biology of the tumor with an appendix concerning chemical mutagenesis. In: Dawe CJ, Harshbarger JC, Kondo S, Sugimura T, Takayama S (eds) Phyletic approaches to cancer. Japan Scientific Societies Press, Tokyo, 1981, pp 101–110.
Jaucaba SF, McKinnell RG, Hanson WJ, Tarin D: Vascular distribution of injected Lucke renal adenocarcinoma cells in warm and cold frogs. Ms. submitted.
Tarin D, Price JE: Metastatic colonization potential of primary tumour cells in mice. Br J Cancer 39: 740–754, 1979.
Price JE, Tarin D: Retention of ‘metastatic’ colonization potential by cells of spontaneous primary tumours after cryopreservation. Br J Cancer 45: 790–793, 1982.
Rotman B, Papermaster BW: Membrane properties of living mammalian cells as studied by enzymatic hydrolysis of flurogenic esters. Proc Nat Acad Sci USA 55: 131–141, 1966.
Bodmer W, Tripp M, Bodmer J: Application of a fluorochromatic cytotoxicity assay to human leukocyte typing. Histocompatibility testing 3: 341–350, 1967.
Zeidman I: Metastasis: a review of recent advances. Cancer Res 17: 157–162, 1957.
Zeidman I: Metastasis: an overview. In: Marchalonis JJ, Hanna MGJr, Fidler IF (eds) Cancer Biology Reviews, 2. Marcel Dekker, New York, 1981, pp 1–27.
Nicholson GL: Cancer metastasis. Sci Am 240: 66–76, 1979.
Poste G, Fidler IJ: The pathogenesis of cancer metastasis. Nature 283: 139–146, 1980.
Tarin D: Morphological studies on the mechanisms of carcinogenesis. In: Tarin D (ed) Tissue interactions in carcinogenesis. Academic Press, London, 1972, pp 227–290.
Tarin D: Cellular interactions in neoplasia. In: Weiss L (ed) Fundamental aspects of metastasis. North Holland, Amsterdam, 1976, pp 151–187.
Ogilvie DJ, Hailey JA, Juacaba SF, Lee EG, Tarin D: 1984. Ms. submitted.
Wooley DE, Tetlow LC, Mooney CJ, Evanson JM: Human collagenase and its extracellular inhibitors in relation to tumor invasiveness. In: Strauli P, Barrett AJ, Baici A (eds) Proteinases and tumor invasion. Raven Press, New York, 1980, pp 97–113.
Tarin D, Hoyt BJ, Evans DJ: Correlation of collagenase secretion with metastatic-colonization potential in naturally occurring murine mammary tumours. Br J Cancer 46: 266–278, 1982.
Gross J: An essay on biological degradation of collagen. In: Hay ED (ed) Cell biology of extracellular matrix. Plenum Press, New York, 1981, pp 217–258.
Ogilvie DJ, McKinnell RG, Tarin D: Temperature-dependent elaboration of collagenase by the renal adenocarcinoma of the leopard frog, Rana pipiens. Cancer Res 44: 3438–3441, 1984.
Woolley DE, Glanville RW, Roberts DR, Evanson JM: Purification, characterization and inhibition of human skin collagenase. Biochem J 169: 265–276, 1978.
Liotta L, Abe S, Robey PG, Martin GR: Preferential digestion of basement membrane collagen by an enzyme derived from a metastatic murine tumor. Proc Nat Acad Sci USA 76: 2268–2272, 1979.
Shields SE, Ogilvie DJ, McKinnell RG, Tarin D: Degradation of basement membrane collagens by metalloproteases released by human, murine, and amphibian tumours. J Pathol 143: 193–197, 1984.
Bailey AJ, Sims TJ, Duance VC, Light ND: Partial characterization of a second basement membrane collagen in human placenta. FEBS Letters 99: 361, 1979.
Trelstad RL, Lawley KR: Isolation and initial characterization of human basement membrane collagens. Biochem Biophys Res Comm 76: 376–384, 1977.
Tarin D: Sequential electron microscopical study of experimental mouse skin carcinogenesis. Int J Cancer 2: 195–211, 1967.
Wolf K, Quimby MC: Amphibian cell culture: permanent cell line from the bullfrog (Rana catesbeiana). Science 144: 1578–1580, 1964.
Sweat F, Puchtler H, Rosentahl SI: Sirius Red F3BA as a stain for connective tissue. Arch Pathol 78: 69–72, 1964.
Junqueira LCU, Bignolas G, Brentani RR: Picrosiruis staining plus polarization microscopy, a specific method for collagen detection in tissue sections. Histochem J 11: 447–455, 1979.
Salsbury AJ: The significance of the circulating cancer cell. Cancer Treat Rev 2: 55–72, 1975.
Coman DR: Decreased mutual adhesiveness, a property of cells from squamous cell carcinomas. Cancer Res 4: 625–629, 1944.
Coman DR: Mechanisms responsible for the origin and distribution of blood-borne tumor metastases: a review. Cancer Res 13: 397–404, 1953.
Seppanen ED, McKinnell RG, Tarin D, Rollins-Smith LA, Hanson W: Temperature-dependent dissociation of Lucke renal adenocarcinoma cells. Differentiation 26: 227–230, 1984.
Tweedell KS: Cytopathology of a frog renal adenocarcinoma in vitro with fluorescence microscopy. Ann NY Acad Sci 126: 170–187, 1965.
Harrison FW, Zambernard J, Cowden RR: Fluorescent cytochemistry of calid and algid normal and Lucke tumor-bearing kidneys. Acta Histochem (Jena) 54: 295–306, 1975.
McKinnell RG, Steven LMJr, Labat DD: Frog renal tumors are composed of stroma, vascular elements, and epithelial cells: What type nucleus programs for tadpoles with the cloning procedure? In: Muller-Berat N, Rosenfeld C, Tarin D, Viza D (eds) Progress in differentiation research. North Holland, Amsterdam, 1976, pp 319–330.
Mareel MMK, De Brabander M: Effect of microtubule inhibitors on malignant invasion in vitro. J Nat Cancer Inst 61: 787–792, 1978.
Mareel M, Storme G, De Bruyne G, Van Cauwenberge R: Anti-invasive effect of microtubule inhibitors in vitro. De Brabander M, DeMay J (eds) Microtubules and microtubule inhibitors. Elsevier/North Holland, Amsterdam, 1980, pp 535–544.
Mareel MM, Storme GA, DeBruyne GK, Van Cauwenberge RM: Vinblastine, vincristine and vindesine: antiinvasive effect on MO4 mouse fibrosarcoma cells in vitro. Eur J Cancer Clin Oncol 18: 199–210, 1982.
Brinkley BR, Fuller GM, Highfield DP: Cytoplasmic microtubules in normal and transformed cells in culture: Analysis by tubulin antibody immunofluorescence. Proc Nat Acad Sci USA 72: 4981–4985, 1975.
McKinnell RG, DeBruyne GK, Mareel MM, Tarin D, Tweedell KS: Cytoplasmic microtubules of normal and tumor cells of the leopard frog: temperature effects. Differentiation 26: 231–234, 1984.
Przybelski RJ, Tweedell KS: Karyotype analysis of a frog pronephric tumor cell line. Exp Cell Biol 46: 289–297, 1978.
Tweedell KS: Localization of glycoconjugates on the surfaces of pronephric tumor cells in vitro. Histochemistry 75: 507–521, 1982.
De Mey J, Hoebeke J, DeBrabander M, Geuens G, Joniau M: Immunoperoxidase visualization of microtubules and microtubular proteins. Nature 264: 273–275, 1976.
Asch BB, Medina D, Brinkley BR: Microtubules and actin-containing filaments of normal, preneoplastic, and neoplastic mouse mammary epithelial cells. Cancer Res 39: 893–907, 1979.
Brinkley BR, Fistel SA, Marcum JM, Pardue RL: Microtubules in cultured cells; indirect immunofluorescent staining with tubulin antibody. Int Rev Cytol 63: 59–95, 1980.
Osborn M, Weber K: The display of microtubules in transformed cells. Cell 12: 561–571, 1977.
DeBrabander MJ, Van de Veire RML, Aerts FEM, Borgers M, Janssen PAJ: The effects of methyl [5-(2-thienylcarbonyl)-1H-benzimidazol-2-yl] carbamate, (R 17934; NSC 238159), a new synthetic antitumoral drug interfering with microtubules, on mammalian cells cultured in vitro. Cancer Res 36: 950–916, 1976.
Mareel MM: Invasion in vitro. Cancer Metastasis Rev 2: 201–218, 1983.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McKinnell, R.G., Tarin, D. Temperature-dependent metastasis of the Lucke renal carcinoma and its significance for studies on mechanisms of metastasis. Cancer Metast Rev 3, 373–386 (1984). https://doi.org/10.1007/BF00051461
Issue Date:
DOI: https://doi.org/10.1007/BF00051461