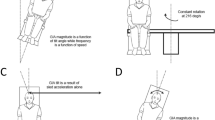
Summary
We studied the influence of static head position on the horizontal nystagmus produced by caloric, rotational and optokinetic stimulation in alert squirrel monkeys. Caloric nystagmus is stronger for nose up (NU) than for nose down (ND) pitches; so, for example, slow-phase eye velocity is four times larger in supine than in prone positions. A similarly directed asymmetry occurs in the horizontal vestibulo-ocular (HVOR) responses to longduration, constant angular-head accelerations, but not to midband (0.1 Hz) sinusoidal head rotations. Consistent with a first-order model of the HVOR, the low-frequency or acceleration gain of the reflex (GA) is equal to the product of the midband velocity gain (GV) and a time constant (TVOR). GV is proportional to the cosine of the angle between the horizontal-canal plane and the plane of rotation, from which it is concluded that signals from the horizontal, but not from the vertical canals contribute to the HVOR. TVOR can be as much as twice as large in NU than in ND positions. GA is proportional to TVOR and it, too, shows a NU-ND asymmetry. The time constant of optokinetic afternystagmus (TOKAN) was also studied. Since TVOR and TOKAN are modified in similar ways by static tilts, it is concluded that head position affects the time constants by way of velocity-storage mechanisms. Evidence is presented that the position-dependent modification of velocity storage is otolith-mediated. The results are used to analyze the mechanisms of caloric nystagmus. The caloric response consists of a convective component (CC), as originally envisioned by Bárány (1906), and a nonconvective component (NC). CC accounts for 75% of the caloric response in the conventional supine testing position. Both components can be affected by the position-dependent modification of TVOR or, equivalently, of GA. It has been suggested that two mechanisms might contribute to NC: 1) a direct thermal effect on hair cells or afferents; or 2) a thermal expansion of labyrinthine fluids that results in a cupular displacement. Both theoretical and experimental evidence indicates that only the first of these mechanisms could result in the steady-state caloric response that is observed in the absence of convection (e.g., in spaceflight and after canal plugging) and that contributes to the prone-supine asymmetry seen in caloric testing.
Similar content being viewed by others
References
Allum JHJ, Tole JR, Weiss AD (1975) MITNYS-II: a digital program for on-line analysis of nystagmus. IEEE Trans Biomed Engin 22: 196–202
Baker J, Wickland C, Goldberg J, Peterson B (1988) Motor output to lateral rectus in cats during the vestibulo-ocular reflex in threedimensional space. Neuroscience 25: 1–12
Bárány R (1906) Untersuchungen über den vom Vestibularapparat des Ohres reflektorisch ausgelösten rythmischen Nystagmus und seine Begleiterscheinungen. Mschr Ohrenheilkd 40: 193–297
Baumgarten R von, Benson A, Berthoz A, Brandt Th, Brand U, Bruzek W, Dichgans J, Kass J, Probst Th, Scherer H, Vieville T, Vogel H, Wetzig J (1984) Effects of rectilinear acceleration and optokinetic and caloric simulation in space. Science 225: 208–212
Behrman W von (1940) Über Indifferenzlagen und Nystagmusgebiete: Versuche mit Kreisbewegung des Kopfes in der Sagittalebene nach einseitiger karotischer Reizung. Acta Otolaryngol Suppl 40: 3–61
Benson AJ (1966) Modification of per- and post-rotational responses by the concomitant linear acceleration. In: Second symposium on the role of the vestibular organs in space exploration. US Government Printing Office, Washington DC, pp 199–211
Benson AJ (1974) Modification of the response to angular accelerations by linear accelerations. In: Kornhuber HH (eds) Handbook of sensory physiology, Vol. VI/2. Springer-Verlag, Berlin, pp 281–320
Benson AJ, Bodin MA (1966) Comparison of the effect of the direction of the gravitational acceleration on post-rotational responses in yaw, pitch and roll. Aerospace Med 37: 889–897
Blanks RHI, Curthoys IS, Bennett ML, Markham CH (1985) Planar relationships of the semicircular canals in rhesus and squirrel monkeys. Brain Res 340: 315–324
Böhmer A, Henn V, Suzuki J-I (1985) Vestibulo-ocular reflexes after selective plugging of the semicircular canals in the monkey: response plane determinations. Brain Res 326: 291–298
Clarke AH, Scherer H, Schleibinger J (1988) Body position and caloric nystagmus response. Acta Otolaryngol 106: 339–347
Coats AC, Smith SY (1967) Body position and the intensity of caloric nystagmus. Acta Otolaryngol 63: 515–532
Cohen B, Matsuo V, Raphan T (1977) Quantitative analysis of the velocity characteristics of optokinetic nystagmus and optokinetic after-nystagmus. J Physiol (Lond) 270: 321–344
Dickman JD, Correia MJ (1989) Responses of pigeon horizontal semicircular canal afferent fibers I. Step, trapezoid, and lowfrequency sinuosoid mechanical and rotational stimulation. J Neurophysiol 62: 1090–1101
DiZio P, Lackner JR (1988) The effects of gravitoinertial force level and head movements on post-rotational nystagmus and illusory after-rotation. Exp Brain Res 70: 485–495
Ezure K, Graf W (1984) A quantitative analysis of the spatial organization of the vestibulo-ocular reflexes in lateral- and frontal-eyed animals. I. Orientation of semicircular canals and extraocular muscles. Neuroscience 12: 85–93
Fernández C, Goldberg JM (1971) Physiology of peripheral neurons innervating semicircular canals of the squirrel monkey. II. Response to sinusoidal stimulation and dynamics of peripheral vestibular system. J Neurophysiol 34: 661–675
Fetter M, Hain TC, Zee DS (1986) Influence of eye and head position on the vestibulo-ocular reflex. Exp Brain Res 64: 208–216
Grossman GE, Leigh RJ, Abel LA, Lanska DJ, Thurston SE (1988) Frequency and velocity of rotational head perturbations during locomotion. Exp Brain Res 70: 470–476
Hain TC (1986) A model of the nystagmus induced by off vertical axis rotation. Biol Cybern 54: 337–350
Hille B (1984) Ionic channels of excitable membranes. Sinauer Associates, Sunderland, p 49
Hood JD (1989) Evidence of direct thermal action upon the vestibular receptors in the caloric test. Acta Otolaryngol 107: 161–165
Lansberg MP, Guedry FE Jr, Graybiel A (1965) Effect of changing resultant linear acceleration relative to the subject on nystagmus generated by angular acceleration. Aerospace Med 36: 456–460
McNally WJ, Stuart EA, Jamieson JS, Gaulton G (1948) Some experiments with caloric stimulation of the human labyrinth to study the relative values of ampullo-petal and ampullo-fugal endolymphatic flow (Ewald's laws). Trans Am Acad Ophthal Otolaryngol 52: 513–541
Minor LB, Goldberg JM (1985) Position-dependent asymmetries in horizontal nystagmus in the squirrel monkey. Soc Neurosci Abstr11: 694
Paige GD (1983a) Vestibuloocular reflex and its interactions with visual following mechanisms in the squirrel monkey. I. Response characteristics in normal animals. J Neurophysiol 49: 134–151
Paige GD (1983b) Vestibuloocular reflex and its interactions with visual following mechanisms in the squirrel monkey. II. Response characteristics and plasticity following unilateral inactivation of horizontal canal. J Neurophysiol 49: 152–168
Paige GD (1985) Caloric responses after horizontal canal inactivation. Acta Otolaryngol 100: 321–327
Raphan T, Cohen B (1988) Organizational principles of velocity storage in three dimensions: the effect of gravity on crosscoupling of optokinetic after-nystagmus. Ann NY Acad Sci 545: 74–92
Raphan T, Cohen B, Henn V (1981) Effects of gravity on rotatory nystagmus in monkeys. Ann NY Acad Sci 374: 44–55
Raphan T, Cohen B, Matsuo, V (1977) A velocity-storage mechanism responsible for optokinetic nystagmus (OKN), optokinetic afternystagmus (OKAN) and vestibular nystagmus. In: Baker R, Berthoz A (eds) Control of gaze by brain stem neurons. Elsevier-North Holland, Amsterdam, pp 37–47
Raphan T, Matsuo V, Cohen B (1979) Velocity storage in the vestibulo-ocular reflex arc (VOR). Exp Brain Res 35: 229–248
Robinson DA (1977) Vestibular and optokinetic symbiosis: an example of explaining by modeling. In: Baker R, Berthoz A (eds) Control of gaze by brain stem neurons. Elsevier-North Holland, Amsterdam, pp 49–58
Scherer H, Brandt U, Clarke AH, Merbold U, Parker R (1986) European vestibular experiments on the Spacelab-1 mission. 3. Caloric nystagmus in microgravity. Exp Brain Res 64: 255–263
Scherer H, Clarke AH (1985) The caloric vestibular reaction in space: physiological considerations. Acta Otolaryngol 100: 328–336
Simpson JI, Rudinger D, Reisine H, Henn V (1986) Geometry of extraocular muscles of the rhesus monkey. Soc Neurosci Abstr12: 1186
Steer, RW (1967) The influence of angular and linear acceleration and thermal stimulation on the human semicircular canal. ScD thesis. Mass Inst Technology, Cambridge MA
Wilpizeski C (1981) Temperature characteristics of squirrel monkey horizontal semicircular canals during caloric irrigation. Otolaryngol Head Neck Surg 89: 678–684
Young JH (1972) Analysis of vestibular system responses to thermal gradients induced in the temporal bone. PhD thesis. University of Michigan, Ann Arbor
Young JH, Anderson DJ (1974) Response patterns of primary vestibular neurons to thermal and rotational stimuli. Brain Res 79: 199–212
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Minor, L.B., Goldberg, J.M. Influence of static head position on the horizontal nystagmus evoked by caloric, rotational and optokinetic stimulation in the squirrel monkey. Exp Brain Res 82, 1–13 (1990). https://doi.org/10.1007/BF00230832
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00230832