Abstract
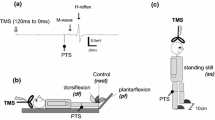
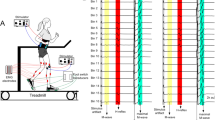
The surface-recorded electromyographic (EMG) responses evoked in the ankle musculature by focal, transcranial, magnetic stimulation of the motor cortex were studied in healthy human subjects. Such soleus evoked motor responses (EMRs) were characterised over a wide range of background levels of motor activity and using different stimulus intensities. EMRs were recorded during predominantly (1) volitional and (2) postural tasks. In the former task subjects were seated and voluntarily produced prescribed levels of soleus activation by reference to a visual monitor of EMG. In the latter task subjects assumed standing postures without EMG feedback. Comparison of the EMRs of soleus, traditionally considered a slow anti-gravity extensor muscle, during these tasks was used to evaluate its cortical control in primarily volitional versus primarily postural activities. The form of soleus EMRs produced by single magnetic cortical stimuli comprised an initial (approx. 30 ms) increase and subsequent (approx. 50 ms) depression of EMG. Cortical stimulation could elicit substantial excitatory soleus EMG responses; for example, responses evoked by mild, magnetic stimuli (125% threshold for inducing a response in the relaxed muscle) as subjects exerted full voluntary plantarflexor effort averaged almost 20% of the maximum M-wave which could be elicited by an electrical stimulus to the posterior tibial nerve. Excitatory EMRs could be elicited in the voluntarily relaxed soleus muscle of the majority of subjects during sitting. The amplitude of soleus responses, induced by threshold stimuli for the relaxed state or approximately 125% threshold intensity, increased approximately linearly with background EMG over a wide range of volitional contraction levels. By contrast, there was no systematic change in the latency of excitatory soleus EMRs with increasing voluntary effort. The excitatory responses evoked in the voluntarily relaxed soleus of seated subjects by magnetic stimulation were regularly facilitated by incremental, voluntary contraction of the contralateral ankle extensors in a graded manner. However, such facilitation of responses was not observed when subjects voluntarily activated the muscle in which EMRs were elicited. The pattern of the responses elicited in soleus by magnetic stimulation during the postural task generally resembled that found during the volitional task. The amplitudes of excitatory soleus EMRs at a given stimulus intensity, obtained when subjects stood quietly, leaned forwards or stood on their toes to produce differing levels of ankle extensor contraction, increased with background EMG. Overall, the relationship between the size of cortically evoked soleus responses and the tonic level of motor activity, observed in individual subjects at matched stimulus intensities, did not consistently differ between postural and volitional tasks. The present results suggest that the motor cortex is potentially capable of exerting rapid regulation of the soleus muscle, and presumably other ankle extensors, not only when the muscle participates in volitional tasks but also when it is engaged in postural maintenance.
Similar content being viewed by others
References
Ackermann H, Scholz E, Koehler W, Dichgans J (1991) Influence of posture and voluntary background contraction upon compound muscle action potentials from anterior tibial and soleus muscle following transcranial magnetic stimulation. Electroencephalogr Clin Neurophysiol 81: 71–80
Advani A, Ashby P (1990) Corticospinal control of soleus motoneurons in man. Can J Physiol Pharm 68: 1231–1235
Baker SN, Olivier E, Lemon RN (1994) Recording an identified pyramidal volley evoked by trancranial magnetic stimulation in a conscius macaque monkey. Exp Brain Res 99: 529–532
Barker AT, Freeston IL, Jalinous R, Merton PA, Morton HB (1985) Magnetic stimulation of the human brain (abstract). J Physiol (Lond) 369: 3P
Brouwer R, Ashby P (1992) Corticospinal projections to lower limb motoneurons in man. Exp Brain Res 89: 649–654
Burke D, Hicks R, Gandevia SC, Stephen J, Woodforth I, Crawford M (1993) Direct comparison of corticospinal volleys in human subjects to transcranial magnetic and electrical stimulation. J Physiol (Lond) 470: 383–393
Capaday C, Stein RB (1986) Amplitude modulation of the soleus H-reflex in the human during walking and standing. J Neurosci 6: 1308–1313
Capaday C, Stein RB (1987) Difference in the amplitude of the human soleus H-reflex during walking and running. J Physiol (Lond) 392: 513–522
Clough JFM, Kernell D, Phillips CG (1968) The distribution of monosynaptic excitation from the pyramidal tract and from primary spindle afferents to motoneurones of the baboon's hand and forearm. J Physiol (Lond) 198: 145–166
Cowan JMA, Day BL, Marsden CD, Rothwell JC (1986) The effects of percutaneous motor cortex stimulation on H reflexes in muscles of the arm and leg in intact man. J Physiol (Lond) 377: 333–347
Datta AK, Harrison LM, Stephens JA (1989) Task-dependent changes in the size of response to magnetic brain stimulation in human first dorsal interosseous muscle. J Physiol (Lond) 418: 13–23
Dimitrijevic MR, Kofler M, McKay WB, Sherwood AM, Van der Linden C, Lissens MA (1992) Early and late lower limb motor evoked potentials elicited by transcranial magnetic motor cortex stimulation. Electroencephalogr Clin Neurophysiol 85: 365–373
Edgley SA, Eyre JA, Lemon RN, Miller S (1990) Excitation of the cortico-spinal tract by electromagnetic and electrical stimulation of the scalp in the macaque monkey. J Physiol (Lond) 425: 301–320
Flament D, Goldsmith P, Buckley CJ, Lemon RN (1993) Tak dependence of responses in first dorsal interosseous muscle to magnetic brain stimulation in man. J Physiol (Lond) 464: 361–378
Grillner S, Udo M (1971) Recruitment in the tonic stretch reflex. Acta Physiol Scand 81: 571–573
Gottlieb GL, Agarwal GC, Stark L (1970) Interactions between voluntary and postural mechanisms of the human motor system. J Neurophysiol 33: 365–381
Hess CW, Mills KR, Murray NMF (1987) Responses in small hand muscles from magnetic stimulation of the human brain. J Physiol (Lond) 388: 397–419
Iles JF, Pisini JV (1992) Cortical modulation of transmission in spinal reflex pathways of man. J Physiol (Lond) 455: 425–446
Jackson JH (1931) Selected writings of John Hughlings Jackson. In: Taylor J (eds) Hodder and Stoughton, London
Jankowska E, Padel Y, Tanaka R (1975) Projections of pyramidal tract cells to alpha-motoneurones innervating hindlimb muscles in the monkey. J Physiol (Lond) 249: 637–667
Kischka U, Fajfr R, Fellenberg T, Hess CW (1993) Facilitation of motor evoked potentials from magnetic brain stimulation in man: a comparitive study of different target muscles. J Clin Neurophysiol 10: 505–512
Kujirai T, Caramia MD, Rothwell JC, Day BL, Thompson PD, Ferbert A, Wroe S, Asselman P, Marsden CD (1993) Corticocortical inhibition in human motor cortex. J Physiol (Lond) 471: 501–519
Lacour M, Vidai PP, Xerri C (1978) Dynamic characteristics of vestibular and visual control of rapid postural adjustments. In: Desmedt JE (ed) Motor control mechanisms in health and disease. Raven, New York, pp 589–606
Lavoie BA, Cody FWJ, Capaday C (1994) Cortical control of human soleus during volitional and postural tasks studied using transcranial magnetic stimulation (abstract). J Physiol (Lond) 475: 38–39P
Maertens de Noordhoot A, Pepin JL, Gerard P, Delwaide PJ (1992) Facilitation of responses to motor cortex stimulation: effects of voluntary isometeic contraction. Ann Neurol 32: 365–370
Marsden CD, Merton PA, Morton HB (1982) Percutaneous stimulation of spinal cord and brain; pyramidal tract conduction velocities in man (abstract). J Physiol (Lond) 328: 6P
Matthews PBC (1972) Mammalian muscle receptors and their central actions. Arnold, London
Mazzocchio R, Rothwell JC, Day BL, Thompson PD (1994) Effect of tonic voluntary activity on the exitability human motor cortex. J Physiol (Lond) 474: 261–267
Merton PA, Morton HB (1980) Stimulation of the cerebral cortex in the intact human subject. Nature 285: 287
Milner-Brown HS, Stein RB, Yemm R (1973a) The orderly recruitment of human motor units during voluntary isometric ontractions. J Physiol (Lond) 230: 359–370
Milner-Brown HS, Stein RB, Yemm R (1973b) Changes in firing rate of human motor units during linearly changing voluntary contractions. J Physiol (Lond) 230: 371–390
Nashner LM (1976) Adapting reflexes controlling the human posture. Exp Brain Res 26: 59–72
Nashner LM (1978) Analysis of movement control in man using the movable platform. In: Desmedt JE (ed) Motor control mechanisms in health and disease. Raven, New York, pp 607–620
Nielsen J, Petersen N, Deuschl G, Ballegaard M (1993) Task-related changes in the effect of magnetic brain stimulation on spinal neurones in man. J Physiol (Lond) 471: 223–243
Phillips CG (1978) Significance of the monosynaptic cortical projection to spinal motoneurones in primates. In: Desmedt JE (ed) Motor control mechanisms in health and disease. Raven, New York, pp 1–9
Phillips CG, Porter R (1964) The pyramidal projection to motoneurones of some muscle groups of the baboon's forelimb. Prog Brain Res 12: 222–242
Preston JB, Shende MC, Uemura K (1967) The motor cortex-pyramidal system: patterns of facilitation and inhibition on motoneurones innervating limb musculature of the cat and baboon and their possible adaptive significance. In: Yahr M, Purpura D (ed) Neurophysiological basis of normal and abnormal motor activities. Raven, Hewlett, pp 61–74
Roick H, Geisen von HJ, Benecke R (1993) On the origin of postexcitatory inhibition seen after transcranial magnetic brain stimulation in awake human subjects. Exp Brain Res 94: 489–498
Ziemann U, Netz J, Szelény A, Hömberg V (1993) Spinal and supraspinal mechanisms contribute to the silent period in the contracting soleus muscle after transcranial magnetic stimulation of human motor cortex. Neurosci Lett 156: 167–171
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lavoie, B.A., Cody, F.W.J. & Capaday, C. Cortical control of human soleus muscle during volitional and postural activities studied using focal magnetic stimulation. Exp Brain Res 103, 97–107 (1995). https://doi.org/10.1007/BF00241968
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00241968