Summary
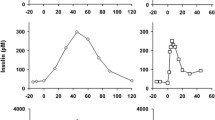
In five totally pancreatectomized human subjects the secretion of gut-derived glucagons was stimulated by ingestion of a meal rich in fat and carbohydrates. Glucagon-like immunoreactivity in plasma, measured with an antiserum against the 6–15 sequence, increased fivefold in response to the meal. Glucagon like immunoreactivity measured with an antiserum against the C-terminal sequence was initially normal (12–13 pmol/l), increased slightly (to 20 pmol/l), and then decreased (to approximately 6 pmol/1). The Chromatographic profile of glucagon-like immunoreactivity in plasma at maximum stimulation was studied after concentration by affinity chromatography. Both assay systems identified two peaks (at Kd-values of 0.30 and 0.60–0.65, and 0.30 and 0.70, respectively). The position at Kd 0.70 corresponds to that of glucagon 1–29. The same components may be identified in plasma from normal subjects. It is concluded that the human intestine is capable of generating all of the molecular forms of glucagon which normally are present in plasma.
Article PDF
Similar content being viewed by others
References
Holst JJ (1978) Extrapancreatic glucagons. Digestion 17: 168–190
Tiengo A, Bessioud M, Valverde I, Tabbi-Anneni A, Delprato S, Alexandre J, Assan R (1982) Absence of islet alpha cell function in pancreatectomized patients. Diabetologia 22: 25–32
Holst JJ (1983) Molecular heterogeneity of glucagon in normal subjects and in patients with glucagon producing tumours. Diabetologia 24: 359–365
Faber OK, Binder C (1977) C-peptide response to glucagon. A test for the residual beta-cell function in diabetes mellitus. Diabetes 26: 605–610
Heding LG (1971) Radioimmunological determination of gut and pancreatic glucagon in plasma. Diabetologia 7: 10–19
Hoist JJ, Åsted B (1974) Production and evaluation of glucagon antibodies for radioimmunoassay. Acta Endocrinol (Copenh) 77: 715–726
Hoist JJ (1980) Evidence that glicentin contains the entire glucagon sequence. Biochem J 187: 337–343
Hoist JJ (1977) Extraction, gel filtration and receptor binding of porcine gastrointestinal glucagon-like immunoreactivity. Diabetologia 13: 159–169
Holst JJ (1977) Interactions of hepatocyte membrane receptors with pancreatic and gut glucagon. In: Foa PP, Bajaj JS, Foa NL (eds) Glucagon: its role in physiology and clinical medicine. Springer Verlag, New York, pp 287–303
Hoist JJ (1982) Evidence that peak II GLI or enteroglucagon is identical to the C-terminal sequence (residues 33–69) of glicentin. Biochem J 207: 381–388
Jørgensen KH, Larsen UD (1972) Purification of 125-I-glucagon by anion exchange chromatography. Horm Metab Res 4: 223–224
Hoist JJ, Kreutzfeldt M, Holm G, Jensen E, Poulsen JSD, Sparsø B, Sparsø B, Schmidt A (1978) Absence of true pancreatic glucagon but persistence of circulating pancreatic glucagon-like immunoreactivity after pancreatectomy in pigs. Diabete Metab 4: 75–79
Stadil F, Rehfeld JF (1973) Determination of gastrin in serum. An evaluation of the reliability of a radioimmunoassay. Scand J Gastroenterol 8: 101–112
Grønholt R, Holst JJ (1983) Stripping of endogenous ligands from antisera against glucagon. J Immunol Methods 58: 83–91
Albano JDM, Ekins RP, Maritz G, Turner RC (1972) A sensitive precise radioimmunoassay of serum insulin relying on charcoal separation of bound and free hormone moieties. Acta Endocrinol (Koh) 70: 487–509
Hoist JJ (1983) Gut glucagon, enteroglucagon, gut GLI, glicentin — current status. Gastroenterology 84: 1602–1613
Baldissera FA, Hoist JJ (1983) Glucagon-related peptides in the human gastrointestinal mucosa. Diabetologia (in press)
Thim L, Moody AJ (1981) The primary structure of porcine glicentin (proglucagon). Regulatory Peptides 2: 139–150
Yanaihara N, Yanaihara C, Nishida T, Hiraiwa T, Mihara S, Sakagami M, Ozaki J, Imagawa K, Shin S (1979) Synthesis of glicentinand proglucagon-related peptides and their immunological properties. In: Rosselin G, Fromageot P, Bonfils S (eds) Hormone receptors in digestion and nutrition. Biomedical Press, Elsevier/North Holland, pp 65–68
Koranyi L, Peterfy F, Szabo J, Török A, Guoth M, Tamas GY (1981) Evidence for transformation of glucagon-like immunoreactivity of the gut into pancreatic glucagon in vivo. Diabetes 30: 792–794
Bataille D, Tatemoto K, Gespach C, Jörnvall H, Rosselin G, Mutt V (1982) Isolation of glucagon-37 (bioactive enteroglucagon/ oxyntomodulin) from porcine jejuno-ileum. Characterization of the peptide. FEBS Lett 146: 79–86
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Holst, J.J., Holst Pedersen, J., Baldissera, F. et al. Circulating glucagon after total pancreatectomy in man. Diabetologia 25, 396–399 (1983). https://doi.org/10.1007/BF00282517
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00282517