Summary
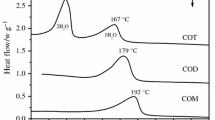
Procedures to obtain calcium oxalate monohydrate, dihydrate and trihydrate are presented and discussed. The influence of several additives and conditions in the formation of calcium oxalate dihydrate crystals are compartatively evaluated. It seems that the presence of colloidal phosphate favours the formation of calcium oxalate dihydrate crystals through heterogeneous nucleation.
Similar content being viewed by others
References
Hesse A, Berg W, Schneider HJ, Hienzsch E (1976) In vitro experiments concerning the theory of the formation of whewellite and weddellite urinary calculi. Urol Res 4:157
Lachance H, Tawashi R (1987) The effect of controlled diffusion of ions on the formation of hydrated calcium oxalate crystals. Scanning Microsc 1:563
Tomazic B, Nancollas GH (1979) The kinetics of dissolution of calcium oxalate hydrates. J Cryst Growth 46:355
Babic-Ivancic V, Furedi-Milhofer H, Purgaric B, Brnicevic N, Despotovic Z (1985) Precipitation of calcium oxalates from high ionic strength solutions. III. J Cryst Growth 71:655
Brecevic LJ, Skrtic D (1986) Transformation of calcium oxalate hydrates. J Cryst Growth 74:399
Nancollas GH, Gardner GL (1974) Kinetics of crystal growth of calcium oxalate monohydrate. J Cryst Growth 21:167
Skrtic D, Furedi-Milhofer H, Markovic M (1987) Precipitation of calcium oxalates from high ionic strength solutions. V.J Cryst Growth 80:113
Brecevic L, Kraj D (1986) The influence of some amino acids on calcium oxalate dihydrate transformation. J Cryst Growth 79:178
Oka T, Yoshioka T, Koide T, Takaha M, Sonoda T (1987) Role of magnesium in the growth of calcium oxalate monohidrate crystals. Urol Int 42:89
Ackermann D, Brown P, Finlayson B (1988) COD production. Urol Res 16:219
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grases, F., Millan, A. & Conte, A. Production of calcium oxalate monohydrate, dihydrate or trihydrate. Urol Res 18, 17–20 (1990). https://doi.org/10.1007/BF00294575
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00294575