Abstract
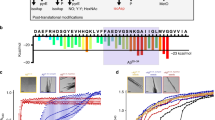
Normal protein-boundl-aspartyl/l-asparaginyl residues may undergo post-translational modification by racemization tod-aspartate, or by isomerization to thel-isoaspartyl form in which the peptide chain links through the beta carboxyl group of the residue. Based on preliminary results reported here, proteins associated with Alzheimer neurofibrillary tangle preparations contain a significantly greater number of these modified aspartyl residues than the unaffected proteins from the surrounding gray matter or in comparable preparations from normal brains.
Similar content being viewed by others
References
Katzman, R. 1986. Alzheimer's Disease. New Eng. J. Med. 314:964–973.
Stephenson, R. C., and Clarke, S. 1989. Succinimide formation from aspartyl and asparaginyl peptides as a model for the spontaneous degradation of proteins. J. Cell Biol. 264:6164–6170.
Clarke, S. 1985. Protein carboxyl methyltransferase: two distinct classes of enzymes. Ann. Rev. Biochem. 54:479–506.
Murray, E. D., and Clarke, S. 1984. Synthetic peptide substrates for the erythrocyte protein carboxyl methyltransferase: detection of a new site of methylation at isomerized L-aspartyl residues. J. Biol. Chem. 256:10722–10732.
Fisher, G. H., Payan, I. L., Chou, S.-J., Man, E. H., Cerwinski, S., Martin, T., Emory, C., and Frey, II, W. H. 1992. Racemized D-aspartate in Alzheimer neurofibrillary tangles. Brain Res. Bull. 28(1):127–131.
Shapira, R., and Chou, C.-H. 1987. Differential racemization of aspartate and serine in human myelin basic protein. Biochem. Biophys. Res. Commun. 146:1342–1349.
Wischik, C. M., Crowther, R. A., Stewart, M., and Roth, M. 1985. Subunit structure of paired helical filaments in Alzheimer's disease. J. Cell. Biol. 100:1095–1912.
Kirschner, D. A., Abraham, C., and Selkoe, D. J. 1986. X-Ray diffraction from intraneuronal paired helical filaments and extraneuronal amyloid fibers in Alzheimer's disease indicates crossbeta configuration. Proc. Natl. Acad. Sci. USA 83:503–507.
Selkoe, D. J., and Abraham, C. R. 1986. Isolation of paired helical filaments and amyloid fibers from human brain. Method. Enzymol. 134:388–404.
Iqbal, K., Zaidi, T., Thompson, C. H., Merz, P. A., and Wisniewski, H.M. 1984. Alzheimer paired helical filaments: bulk isolation, solubility, and protein composition. Acta Neuropathol. 62:167–177.
Man, E. H., Sandhouse, M., Burg, J., and Fisher, G. H. 1983. Accumulation of D-aspartic acid with age in human brain. Science 220:1407–1408.
Man, E. H., Fisher, G. H., Payan, I. L., Cadilla-Perezrios, R., Garcia, N. M., Chemburkar, R., Arends, G., and Frey, W. H. II. 1987.d-Aspartate in human brain. J. Neurochem. 48:510–515.
Fisher, G. H., Garcia, N. M., Payan, I. L., Cadilla-Perezrios, R., Sheremata, W. A., and Man, E. H. 1986.d-Aspartic acid in purified myelin and myelin basic protein. Biochem. Biophys. Res. Commun. 135:683–687.
Shapira, R., Austin, G. E., and Mirra, S. S. 1988. Neuritic plaque amyloid in Alzheimer's disease is highly racemized. J. Neurochem. 50:69–74.
Metuzals, J. 1986. On the question of enantiomer determination in paired helical filaments protein. Pages 25–27,in J. Metuzals, (ed.) Electron Microscopy and Alzheimer's Disease, San Francisco Press, San Francisco, CA.
Johnson, B. A., Shirokawa, J. M., Geddes, J. W., Choi, B. H., Kim, R. C., and Aswad, D. W. 1991. Protein L-isoaspartyl methyltransferase in postmortem brains of aged humans. Neurobiol. Aging 12:19–24.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Payan, I.L., Chou, SJ., Fisher, G.H. et al. Altered aspartate in Alzheimer neurofibrillary tangles. Neurochem Res 17, 187–191 (1992). https://doi.org/10.1007/BF00966798
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00966798