Summary
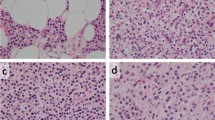
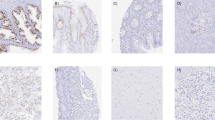
Twenty-five surgical specimens of malignant human prostate, 3 lymph nodes with metastatic prostate carcinoma, 11 normal human prostates, as well as 3 human prostate cell lines (DU-145, PC3 and LNCaP) were examined for the expression of the human matrix metalloproteinase-7 gene (MMP-7) from the human collagenase family (originally called PUMP-1 for putative metalloproteinase-1) [Quantin et al. (1989) Biochemistry 28:5327–5334; Muller et al. (1988) Biochem J 253:187–192; Matrisian and Bowden (1990) Semin Cancer Biol 1:107–115]. Northern blots were prepared using total RNA extracted from 18 prostate adenocarcinomas, 2 lymph nodes with metastatic prostate carcinoma and 11 normal human prostates. When the northern blots were hybridized with a32P-labeledMMP-7 cDNA probe, a 1.2-kb mRNA was detected in 14 out of 18 prostate adenocarcinomas, 1 out of 2 metastatic lymph nodes, and 3 out of 11 normal prostates. The 3 human prostate cell lines did not show any evidence of theMMP-7 transcript. In situ hybridization was conducted to localize theMMP-7 mRNA to individual cells using a35S-labeledMMP-7 cRNA. In situ hybridization was carried out on snap-frozen tissue sections of 7 prostate adenocarcinomas and 3 lymph nodes containing metastatic prostate adenocarcinoma using the same tissues previously probed by northern analysis as well as new samples. In situ hybridization revealed that theMMP-7 gene was expressed in the epithelial cells of primary prostate adenocarcinoma as well as in invasive and metastatic cells.MMP-7 expression was also seen focally in some dysplastic glands but not in stroma. Additional northern blot analysis was performed using probes to human type-IV collagenase, type-I collagenase and stromelysin I in human prostate adenocarcinoma as well as normal prostate tissue. Our results indicated that 6 out of 10 adenocarcinoma samples and none of the 4 normal samples were positive for type-IV collagenase transcripts. Tissue samples were also examined for the expression of type-I collagenase (9 adenocarcinomas and 4 normal) and stromelysin I (13 adenocarcinomas) by northern analysis. None of the tissues was found to express the transcripts of interest at detectable levels. These data suggest that certain metalloproteinases are present in prostatic adenocarcinoma and may play a role in invasion and metastasis.
Similar content being viewed by others
Abbreviations
- SSC:
-
standard saline citrate
- SDS:
-
sodium dodecycl sulfate
References
Abramson M, Schilling W, Huang CC, Salome RG (1975) Collagenase activity in epidermoid carcinoma of the oval cavity and larynx. Ann Otol Rhinol Laryngol 84:158–163
Baur EA, Gordon JM, Reddick ME, Eisen AZ (1977) Quantitation and immunohistochemical localization of human skin collagenase in basal cell carcinoma. J Invest Dermatol 69:363–367
Bosland MC (1988) The ethiopathogenesis of prostatic cancer with special reference to environmental factor. Adv Cancer Res 51:1–107
Doble A, William G (1987) Prostate cancer: the depressing facts. Practioner 231:239–241
Frank IM (1954) Latent carcinoma of prostate. J Pathol Bacteriol 68:603–616
Fuchs MD, Brawer MK, Rennels MA, Nagle RB (1989) The relationship of basement membrane to histologic grade of human prostatic carcinoma. Mod Pathol 2:105–111
Holand B (1980) Latent prostatic cancer in consecutive autopsy series. Scand J Urol Nephrol 14:29–35
Horoszewicz JS, Leong SS, Kawinski E, Karr JP, Rosenthal H, Chu TM, Mirand EA, Murphy GP (1983) LNCaP model of human prostatic carcinoma. Cancer Res 43:1809–1818
Kaighan ME, Shanker Navayan K, Ohnuki Y, Lechner JF, Jones LW (1979) Establishment and characterization of a human prostatic carcinoma cell line (PC3). Invest Urol 17:16–23
Krieg P, Amtman E, Sauer G (1983) The simultaneous extraction of high molecular weight DNA and of RNA from solid tumor. Anal Biochem 134:288–294
Liotta LA (1986) Tumor invasion and metastasis-role of extracellular matrix. Cancer Res 46:1–7
Maniatis T, Fritsch EF, Sambrook J (1985) Molecular cloning, a laboratory manual. Cold Spring Harbor Laboratory Press, New York, pp 202–203
Matrisian LM, Bowden GT (1990) Stromelysin/transin and tumor progression. Semin Cancer Biol 1:107–115
Muller D, Quantin MC, Millon-Collard R, Abecassis J, Breathnach R (1988) The collagenase family in human consists of at least four members. Biochem J 253:187–192
Mullins DE, Rohlich ST (1983) The role of proteinases in cellular invasiveness. Biochim Biophys Acta 695:117–214
Quantin B, Murphy G, Breathnach R (1989) PUMP-1 cDNA codes for a protein with characteristics similar to those of classical collagenases family member. Biochemistry 28:5327–5334
Reese JH, McNeal JE, Redwine EA, Stamey TA, Freiha FS (1988) Tissue type plasminogen activator as a marker of functional zone. Prostate 12:47–53
Silverberg E, Lubera JA (1988) Cancer Statistics. CA-A Cancer Journal for Clinicians. American Cancer Society, New York, NY 38:5–22
Stone KR, Mickey DD, Wunderli H, Mickey GH, Paulson DF (1987) Isolation of a human prostate carcinoma cell line (DU-145). Int J Cancer 21:274–281
Tryggvason K, Hoyhtya M, Salo T (1987) Proteolytic degradation of extracellular matrix in tumor invasion. Biochim Biophys Acta 907:191–217
Woolley DE (1984) Collagenolytic mechanisms in tumor cell invasion. Cancer Mestastasis Rev 3:361–371
Woolley DE, Grafton CA (1980) Collagenase immunolocalization studies of cutaneous secondary melanoma. Br J Cancer 42:260–265
Author information
Authors and Affiliations
Additional information
Aided by grant PDT-338 from American Cancer Society to R.B.N. and USPH grant CA-40584 awarded to G.T.B. This work was also supported in part by the Arizona Disease Control Commission.
Rights and permissions
About this article
Cite this article
Pajouh, M.S., Nagle, R.B., Breathnach, R. et al. Expression of metalloproteinase genes in human prostate cancer. J Cancer Res Clin Oncol 117, 144–150 (1991). https://doi.org/10.1007/BF01613138
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01613138