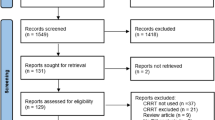
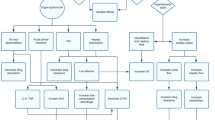
Conclusion
The appropriate dosage adjustment of drugs during continuous extracorporeal renal replacement therapy requires an understanding of the clearance characteristics of the different techniques and knowledge of the pharmacokinetic parameters of the drugs. Extracorporeal drug removal can be calculated from these data. However, since pharmacokinetics are different in critically ill patients and in healthy volunteers, these calculations can only yield rough estimates and monitoring of drug level and/or clinical effect is still essential.
Similar content being viewed by others
References
Bennett WN, Aronoff GR, Morrison G, Golper TA, Pulliam J, Wolfson M, Singer I (1983) Drug prescribing in renal failure: dosing guidelines for adults. Am J Kidney Dis 3:155–193
La Greca G, Biasoli S, Borin D, Brendolan A, Chiaramonte S, Fabris A, Feriani M, Pisani E, Ronco C (1983) Drugs and dialysis. Int J Artif Organs 6:139–156
Lee CC, Marbury TC (1984) Drug therapy in patients undergoing hemodialysis: clinical pharmacokinetic considerations. Clin Pharmacokinet 9:42–66
Fillastre JP, Singlas E (1991) Pharmacokinetics of newer drugs in patients with renal impairment. I. Clin Pharmacokinet 20:293–310
Singlas E, Fillastre JP (1991) Pharmacokinetics of newer drugs in patients with renal impairment. II. Clin Pharmacokinet 20:389–410
St Peter WL, Redic-Kill KA, Halstenson CE (1992) Clinical pharmacokinetics of antibiotics in patients with impaired renal function. Clin Pharmacokinet 22:169–210
Schetz M, Lauwers PM, Ferdinande P (1989) Extracorporeal treatment of acute renal failure in the intensive care unit: a critical view. Intensive Care Med 15:349–357
Reetze-Bonorden P, Bohler J, Keller E (1993) Drug dosage in patients during continuous renal replacement therapy: pharmacokinetic and therapeutic considerations. Clin Pharmacokinet 24:162–179
Cutler RE, Forland SC (1989) Changing drug dosage in renal insufficiency. 2. Dialysis of drugs. Dial Transplant 18:250–257
Rumpf KW, Rieger J, Ansorg R, Doht B, Scheler F (1977) Binding of antibiotics by dialysis membranes and its clinical relevance. Kidney Int 12:87
Kraft D, Lode H (197) Elimination of ampicillin and gentamicin by hemofiltration. Klin Wochenschr 57:195–196
Kronfol NO, Lau AH, Barakat MM (1987) Aminoglycoside binding to polyacrylonitrile hemofilter membranes during continuous hemofiltration. ASAIO Trans 33:300–303
Cigarran-Guldris S, Brier ME, Golper TA (1991) Tobramycin clearance during simulated continuous arteriovenous hemodialysis. Contrib Nephrol 93:120–123
Vincent HH, Akçahuseyin E, Vos MC, Van Ittersum FJ, Van Duyl WA, Schalekamp MAD (1990) Determinants of blood flow and ultrafiltration in continuous arteriovenous hemodiafiltration: theoretical predictions and laboratory and clinical observations. Nephrol Dial Transplant 5:1031–1037
Pallone TL, Petersen J (1988) Continuous arteriovenous hemofiltration: An in vitro simulation and mathematical model. Kidney Int 33:685–698
Ronco C, Brendolan A, Borin D, Bragantini L, Fabris A, Feriani M, Chiaramonte S, La Greca G (1984) Permeability characteristics of polysulfone membranes in CAVH. In: Siebert HG, Mann H (eds) Continuous arteriovenous hemofiltration (CAVH). Karger, Basel, pp 59–63
Golper TA (1991) Drug removal during continous hemofiltration or dialysis. Contrib Nephrol 93:110–116
Sprenger KGB, Stephan H, Kratz W, Huber K, Franz ME (1985) Optimising of hemodiafiltration with modern membranes? Contrib Nephrol 46:43–60
Sigler MH, Teehan BP, Van Valckenburgh D (1987) Solute transport in continuous hemodialysis: a new treatment for acute renal failure. Kidney Int 32:562–571
Davies SP, Kox WJ, Brown EA (1991) Clearance studies in patients with acute renal failure treated by continuous arteriovenous hemodialysis. Contrib Nephrol 93:117–119
Benet LZ, Williams RL (1992) Design and optimization of dosage regimens: pharmacokinetic data: In: Goodman Gilman A, Rall TW, Nies AS, Taylor P (eds) The pharmacological basis of therapeutics. McGraw-Hill International New York, pp 1650–1735
Brater DG (1983) Pharmacokinetics. In: Chernow B (ed) The pharmacologic approach to the critically ill patient. Williams & Wilkins, Baltimore, pp 1–21
Blye E, Lorch J, Cortell S (1984) Extracorporeal therapy in the treatment of intoxication. Am J Kidney Dis 3:321–328
Tillement JP, Lhoste F, Giudicelli JF (1978) Diseases and drug protein binding. Clin Pharmacokinet 3:144–154
Wedlund PJ, Branch RA (1983) Adjustment of medications in liver failure. In: Chernow B (ed) The pharmacologic approach to the critically ill patient. Williams & Wilkins, Baltimore, pp 84–114
Elston AC, Bayliss MK, Park GR (1993) Effect of renal failure on drug metabolism by the liver. Br J Anaesth 71:282–290
Bodenham A, Shelly NP, Park GR (1988) The altered pharmacokinetics and pharmacodynamics of drugs commonly used in critically ill patients. Clin Pharmacokinet 14:347–373
Runciman WB, Myburgh JA, Upton RN (1990) Pharmacokinetics and pharmacodynamics in the critically ill. Baillieres Clin Anaesthesiol 4:271–303
Vos MC, Vincent HH, Yzerman EPF (1992) Clearance of imipenem/cilastatin in acute renal failure patients treated by continuous hemodiafiltration (CAVHD). Intensive Care Med 18:282–285
Lanese DM, Alfrey PS, Molitoris BA (1989) Markedly increased clearance of vancomycin during hemodialyses using polysulfone dialysers. Kidney Int 35:1409–1412
Kong KL, Haynes S, Bion J (1990) Newer drugs in intensive care. Baillieres Clin Anaesthesiol 4:305–331
Feinfeld DA, Frishman WH (1987) Renal considerations in cardiovascular drug therapy. Cardiol Clin 5:675–688
Verresen L, Waer M, Vanrenterghem Y, Michielsen P (1990) Angiotensin-converting-enzyme inhibitors and anaphylactoid reactions to high-flux membrane dialysis. Lancet 336:1360–1362
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schetz, M., Ferdinande, P., Van den Berghe, G. et al. Pharmacokinetics of continuous renal replacement therapy. Intensive Care Med 21, 612–620 (1995). https://doi.org/10.1007/BF01700172
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01700172