Abstract
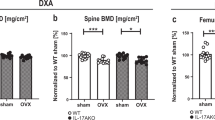
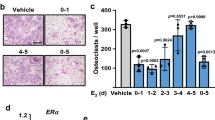
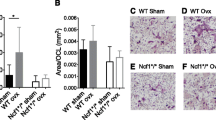
Estrogen deficiency causes a marked bone loss by stimulating osteoclastic bone resorption. Recent studies on the role of estrogen in bone resorption have focused on the involvement of at least two cytokines, IL-1 and IL-6. It is well known that IL-1 stimulates osteoclastic bone resorptionin vitro andin vivo by inducing the differentiation of osteoclast precursors into mature osteoclasts and by stimulating their activity. On the other hand, we reported that IL-6 greatly stimulated the differentiation of osteoclast precursors into mature osteoclast in the presence of soluble IL-6 receptors in co-cultures of mouse bone marrow and primary osteoblastic cells. Using the systems of ovariectomized (OVX) animals, recent studies have indicated that IL-1 and/or IL-6 are over-produced by bone marrow cells in estrogen deficiency. We examined endogenous bone-resorbing factors present in the supernatants fraction of mouse bone marrows in OVX mice. The endogenous bone-resorbing activity in bone marrow was much greater in OVX mice than in sham mice. The bone-resorbing activity of bone marrow supernatants was significantly decreased by adding indomethacin or neutralizing antibodies against bone-resorbing cytokines such as IL-1α and IL-6. These results suggest that the enhanced bone-resorption that occurs during estrogen deficiency is due to several cytokines including IL-1, IL-6 and prostaglandins rather than to a single factor. Estrogen deficiency induced by OVX stimulated hemopoiesis and caused a marked increase in bone marrow cells. OVX selectively stimulated accumulation of B lymphocyte precursors. This suggests that estrogen modulates the microenvironment for hemopoiesis and bone metabolism in bone marrow.
Similar content being viewed by others
References
Mundy GR, Roodman GD: Osteoclast ontogeny and function. J Bone Miner Res 5: 209–281, 1987
Suda T, Takahashi N, Martin TJ: Modulation of osteoclast differentiation. Endocr Rev 13: 66–80, 1992
Udagawa N, Takahashi N, Akatsu T et al.: Origin of osteoclast: mature monocytes and macrophages are capable of differentiating into osteoclasts under a suitable microenvironment prepared by bone marrow-derived stromal cells. Proc Natl Acad Sci USA 87: 7260–7264, 1990
Pacifici R, Brown C, Puscheck E et al.: Effect of surgical menopause and estrogen replacement on cytokine release from human blood mononuclear cells. Proc Natl Acad Sci USA 88: 5134–5138, 1991
Kimble RB, Vannice JL, Bloedow DC et al.: Interleukin-1 receptor antagonist decreases bone loss and bone resorption in ovariectomized rats. J Clin Invest 93: 1959–1967, 1994
Gowen M, Wood DD, Ihrie EJ et al.: An interleukin-1-like factor stimulates bone resorption in vitro. Nature (Lond.) 306: 378–380, 1983
Akatsu T, Takahashi N, Udagawa N et al.: Role of prostaglandins in interleukin-1-induced bone resorption in mice in vitro. J Bone Miner Res 6: 183–190, 1991
Rosen GD, Birkenmeier TM, Raz A et al.: Identification of a cyclooxygenase-related gene and its potential role in prostaglandin formation. Biochem Biophys Res Commun 164: 1358–1365, 1989
Xie W, Chipman JG, Robertson DL et al.: Expression of a mitogen-responsive gene encoding prostaglandin synthase is regulated by mRNA splicing. Proc Natl Acad Sci USA 88: 2692–2696, 1991
O'Banion MK, Winn VD, Young DA: cDNA cloning and functional activity of a glucocorticoid-regulated inflammatory cyclooxygenase. Proc Natl Acad Sci USA 89: 4888–4892, 1992
Brum-Fernandes AJ, Laport S, Heroux Me et al.: Expression of prostaglandin endoperoxide synthase-1 and prostaglandin endoperoxide synthase-2 in human osteoblasts. Biochem Biophys Res Commun 198: 955–960, 1994
Harrison JR, Lorenzo JA, Kawaguchi H et al.: Stimulation of prostaglandin E2 production by interleukin-1α and transforming growth factorα in osteoblastic MC3T3-E1 cells. J Bone Miner Res 9: 817–823, 1994
Kawaguchi H, Raisz LG, Voznesensky OD et al.: Regulation of the two prostaglandin G/H synthases by parathyroid hormone, interleukin-1, cortisol, and prostaglandin E2 in cultured neonatal mouse calvariae. Endocrinology 135: 1157–1164, 1994
Ishimi Y, Miyaura C, Jin CH et al.: IL-6 is produced by osteoblasts and induces bone resorption. J Immunol 145: 3297–3303, 1990
Ishimi Y, Abe E, Jin CH et al.: Leukemia inhibitory factor/differentiation-stimulating factor (LIF/D-factor): Regulation of its production and possible roles in bone metabolism. J Cell Physiol 152: 71–78, 1992
Horowitz M, Fields A, Zamparo J et al.: IL-11 secretion and its regulation in bone cells. J Bone Miner Res 8: S143, 1993
Girasole G, Passeri G, Jilka RL et al.: Interleukin-11: A new cytokine critical for osteoclast development. J Clin Invest 93: 1516–1524, 1994
Tamura T, Udagawa N, Takahashi N et al.: Soluble interleukin-6 receptor triggers osteoclast formation by interleukin-6. Proc Natl Acad Sci USA 90: 11924–11928, 1993
Lowik CW, Pluijm GMG, Bloys H et al.: Parathyroid hormone (PTH) and PTH-like protein (PLP) stimulate interleukin-6 production by osteogenic cells: a possible role of interleukin-6 in osteoclastogenesis. Biochem Biophys Res Commun 162: 1546–1552, 1989
Black K, Garrett IR, Mundy GR: Chinese hamster ovarian cells transfected with the murine interleukin-6 gene cause hypercalcemia as well as cachexia, leukocytosis and thrombocytosis in tumor-bearing nude mice. Endocrinology 128: 2657–2659, 1991
Roodman GD, Kurihara N, Ohsaki Y et al.: Interleukin 6. A potential autocrine/paracrine factor in Paget's disease of bone. J Clin Invest 89: 46–52, 1992
Girasole G, Jilka RL, Passeri G et al.: 17β-Estradiol inhibits interleukin-6 production by bone marrow-derived stromal cells and osteoblasts in vitro: A potential mechanism for the antiosteoporotic effect of estrogens. J Clin Invest 89: 883–891, 1992
Jilka RL, Hangoc G, Girasole G et al.: Increased osteoclast development after estrogen loss: Mediation by interleukin-6. Science 257: 88–91, 1992
Poli V, Balena R, Fattori E et al.: Interleukin-6 deficient mice are protected from bone loss caused by estrogen depletion. EMBO J 13: 1189–1196, 1994
Miyaura C, Owan I, Masuzawa T et al.: IL-1α is a major endogenous bone-resorbing factor in estrogen deficiency. J Bone Miner Res 8: S130, 1993
Screpanti I, Morrone S, Meco D et al.: Steroid sensitivity of thymocyte subpopulations during intrathymic differentiation. Effects of 17β-estradiol and dexamethasone on subsets expressing T cell antigen receptor or IL-2 receptor. J Immunol 142: 3378–3383, 1989
Masuzawa T, Miyaura C, Onoe Y et al.: Estrogen deficiency stimulates B lymphopoiesis in mouse bone marrow. J Clin Invest 94: 1090–1097, 1994
Landreth K, Dorshkind K: Pre-B cell generation potentiated by soluble factors from a bone marrow stromal cell line. J Immunol 140: 845–852, 1988
Namen AE, Lupton S, Hjerrild K et al.: Stimulation of B-cell progenitors by cloned murine interleukin-7. Nature 333: 571–573, 1988
Kincade PW: Experimental models for understanding B lymphocyte formation. Adv Immunol 41: 181–267, 1987
Miyake K, Weissman IL, Greenberger JS et al.: Evidence for a role of the integrin VLA-4 in lymphohemopoiesis. J Exp Med 173: 599–607, 1991
Miyake K, Medina KL, Hayashi SI et al.: Monoclonal antibodies to Pgp-1/CD44 block lymphohemopoiesis in long-term bone marrow cultures. J Exp Med 171: 477–488, 1990
Okada S, Nakauchi H, Nagayoshi K et al.: Enrichment and characterization of murine hematopoietic stem cells that express c-kit molecule. Blood 78: 1706–1712, 1991
Author information
Authors and Affiliations
About this article
Cite this article
Miyaura, C. Mechanism of bone resorption induced by estrogen deficiency. J Bone Miner Metab 12 (Suppl 2), S3–S7 (1994). https://doi.org/10.1007/BF02383388
Issue Date:
DOI: https://doi.org/10.1007/BF02383388