Abstract
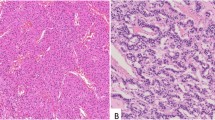
Despite the widespread distribution of chromogranin A (CgA) in neuroendocrine tissues, the biological function of CgA has not yet been elucidated. The primary amino acid sequence of CgA, elucidated by cDNA analysis, has been revealed to include several pairs of basic amino acid residues that are homologous to the bioactive peptides, such as pancreastatin (PST) and chromostatin (CST). Using antibodies for human PST and CST, the immunohistochemical localization of these peptides was investigated in neuroendocrine tissues, including human pituitary glands, pancreas, adrenal medulla, various types of neuroendocrine neoplasms (13 pheochromocytomas, 10 medullary thyroid carcinomas, 11 pancreatic endocrine tumors, and 19 carcinoid tumors), and the cell line QGP-1N derived from human somatostatin-producing pancreatic endocrine tumor.
Variable immunoreactive intensities of PST and CST were seen, but both peptides were detectable in all neuroendocrine tissues and in most of the neoplasms. Immunoreactivity for both PST and CST was observed in 100 and 73%, respectively, of pancreatic endocrine tumors, all pheochromocytomas, and 80 and 40%, respectively, of medullary thyroid carcinomas, as well as all nonrectal carcinoid tumors. In rectal carcinoids, cells immunoreactive for PST and CST were sparse. The distribution of PST and CST was similar to that of CgA, and it is considered that these peptides are simultaneously processed from CgA, and may play roles in autocrine and paracrine regulation on various hormones in addition to their previously known functions.
Similar content being viewed by others
References
Simon JP, Aunis D. Biochemistry of the chromogranin A protein family. Biochem J 262: 1–13, 1989.
Tatemoto K, Efendic S, Mutt V, Makk G, Feistner G, Barchas HD. Pancreastatin, a novel pancreatic peptide that inhibits insulin secretion. Nature 324: 476–478, 1986.
Konecki DS, Benedum UM, Gerdes H-H, Hutter WB. The primary structure of human chromogranin A and pancreastatin. J Biol Chem 262: 17,026–17,030, 1987.
Funakoshi A, Jimi A, Yasunami Y, Tateishi K, Funakoshi S, Tamamura H, Yajima H. Bioactivity of human pancreastatin and its localization in pancreas. Biochem Biophys Res Commun 159: 913–918, 1989.
Funakoshi A, Miyasaka K, Nakamura R, Kitani K, Funakoshi S, Tamamura H, Fujii N, Yajima H. Bioactivity of synthetic human pancreastatin on exocrine pancreas. Biochem Biophys Res Commun 156: 1237–1242, 1988.
Funakoshi A, Tateishi K, Kitayama N, Jimi A, Matuoka Y, Kono A. Parallel secretion of pancreastatin and somatostatin from human pancreastatin producing cell line (QGP-1N). Pancreas 8: 375–382, 1993.
Tateishi K, Funakoshi A, Jimi A, et al. High plasma pancreastatin-like immunoreactivity in a patient with malignant insulinoma. Gastroenterology 97: 1313–1318, 1989.
Tateishi K, Funakoshi A, Wakasugi H, Iguchi H, Shinozaki H, Abe M, Funakoshi S, Tamamura H, Yajima H, Matsuoka Y. Plasma pancreastatin-like immunoreactivity in various diseases. J Clin Endocrinol Metab 69: 1305–1308, 1989.
Galindo E, Rill A, Bader M-F, Aunis D. Chromostatin, a 20-amino acid peptide derived from chromogranin A, inhibits chromaffin cell secretion. Proc Natl Acad Sci USA 88: 1426–1430, 1991.
Simon J-P, Bader M-F, Aunis D. Secretion from chromaffin cells is controlled by chromogranin A-derived peptides. Proc Natl Acad Sci USA 85: 1712–1716, 1988.
Kimura N, Sasano N, Yamada R, Satoh J. Immunohistochemical study of chromogranin in 100 cases of phaeochromocytoma, carotid body tumor, medullary thyroid carcinoma and carcinoid tumour. Virchow Arch A Pathol Anat 413: 33–38, 1988.
Lloyd RV, Iacangelo A, Eiden LE, Cano M, Grimes M. Chromogranin A and B messenger ribonucleic acids in pituitary and other normal and neoplastic human endocrine tissues. Lab Invest 60: 548–556, 1989.
Cetin Y, Aunis D, Bader M-F, Galindo E, Jorns A, Bargsten G, Grube D. Chromostatin, a chromogranin A-derived bioactive peptide, is present in human pancreatic insulin (β) cells. Proc Natl Acad Sci USA 90: 2360–2364, 1993.
Schmidt WE, Siegel EG, Lamberts R, Gallwitz B, Creutzfeldt W. Pancreastatin: molecular and immunocytochemical characterization of a novel peptide in porcine and human tissues. Endocrinology 123: 1395–1404, 1988.
Kimura N, Mizokami A, Oonuma T, Sasano H, Nagura H. Immunocytochemical localization of androgen receptor with polyclonal antibody in paraffin embedded human tissues. J Histochem Cytochem 41: 671–678, 1993.
O’Connor DT. Chromogranin: wide-spread immunoreactivity in polypeptide hormone producing tissue and serum. Regul Peptides 6: 263–280, 1983.
Lloyd RV, Wilson BS. Specific endocrine tissue marker defined by a monochronal antibody. Science 222: 628–630, 1983.
Vandell IM, Lloyd RV, Wilson BS, Polak JM. Ultrastructural localization of chromogranin: a potential marker for the electronmicroscopical recognition of endocrine cell secretory granules. Histochem J 17: 981–992, 1985.
Cohen DV, Elting JJ, Frick M, Elde R. Selective localization of the parathyroid secretory protein/adrenal medulla chromogranin A protein family in a wide variety of endocrine cells of the rat. Endocrinology 114:1963–1974, 1984.
Grube D, Aunis D, Bader F, Cetin Y, Jorns A, Yoshie S. Chromogranin A (CGA) in the gastro-entero-pancreatic (GEP) endocrine system I. CGA in the mammalian endocrine pancreas. Histochemistry 85:441–452, 1986.
Seidah NG, Chretien M. Proprotein and prohormone convertases of the subtilisin fam- ily. Recent developments and future perspectives. Trends Endocrinol Metab 3: 133–140, 1992.
Kirchmair R, Egger C, Gee P, Hogue-Angeletti R, Fischer-Colbri R, Laslop A, Winkler H. Differential subcellular distribution of PCI, PC2 and furin in bovine adrenal medulla and secretion of PCI and PC2 from this tissue. Neurosci Lett 143: 143–145, 1992.
Arden SD, Rutherford NG, Guest PC, Curry WJ, Bailyes EM, Johnston CF, Hutton JC. The post-translational processing of chromogranin A in the pancreatic islet: involvement of the eukaryote subtilisin PC2. Biochem J 298:521–528, 1994.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kimura, N., Funakoshi, A., Aunis, D. et al. Immunohistochemical localization of chromostatin and pancreastatin, chromogranin A-Derived bioactive peptides, in normal and neoplastic neuroendocrine tissues. Endocr Pathol 6, 35–43 (1995). https://doi.org/10.1007/BF02914987
Issue Date:
DOI: https://doi.org/10.1007/BF02914987