Abstract
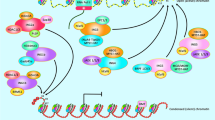
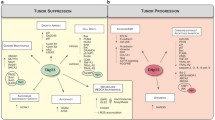
The inhibitor of growth (ING) family of tumor suppressors has five members and is implicated in the control of apoptosis, senescence, DNA repair, and cancer progression. However, little is known about ING activity in the regulation of cancer progression. ING members and splice variants seem to behave differently with respect to cancer invasion and metastasis. Interaction with histone trimethylated at lysine 4 (H3K4me3), hypoxia inducible factor-1 (HIF-1), p53, and nuclear factor kappa-B (NF-κB) are potential mechanisms by which ING members exert effects on invasion and metastasis. Subcellular mislocalization, rapid protein degradation, and to a lesser extent ING gene mutation are among the mechanisms responsible for inappropriate ING levels in cancer cells. The aim of this review is to summarize the different roles of ING family tumor suppressors in cancer progression and the molecular mechanisms involved.

Similar content being viewed by others
References
Unoki M, Kunamoto K, Takenoshita S, Harris CC (2009) Reviewing the current classification of inhibitor of growth family proteins. Cancer Sci 100:1173–1179
Ythier D, Larrieu D, Brambilla C, Brambilla E, Pedeux R (2008) The new tumor suppressor genes ING: genomic structure and status in cancer. Int J Cancer 123:1483–1490
Gong W, Suzuki K, Russell M, Riabowol K (2005) Function of the ING family of PHD proteins in cancer. Int J Biochem Cell Biol 37:1054–1065
Coles AH, Jones SN (2009) The ING gene family in the regulation of cell growth and tumorigenesis. J Cell Physiol 218:45–57
Soliman MA, Riabowol K (2007) After a decade of study-ING, a PHD for a versatile family of proteins. Trends Biochem Sci 32:509–519
Campos E, Chine MY, Kuo WH, Li G (2004) Biological functions of the ING family tumor suppressors. Cell Mol Life Sci 61:2597–2613
Larrieu D, Ythier D, Binet R, Brambilla C, Brambilla E, Sengupta S, Pedeux R (2009) Ing2 controls the progression of DNA replication forks to maintain genome stability. EMBO Rep 10:1168–1174
Garkavtsev I, Grigorian IA, Ossovskaya VS, Chernov MV, Chumakov PM, Gudkov AV (1998) The candidate tumour suppressor p33ING1 cooperates with p53 in cell growth control. Nature 391:295–298
Takahashi M, Seki N, Ozaki T, Kato M, Kuno T, Nakagawa T, Watanabe K-i, Miyazaki K, Ohira M, Hayashi S, Hosoda M, Tokita H, Mizuguchi H, Hayakawa T, Todo S, Nakagawara A (2002) Identification of the p33ING1-regulated genes that include cyclin B1 and proto-oncogene DEK by using cDNA microarray in a mouse mammary epithelial cell line NMuMG. Cancer Res 62:2203–2209
Shinoura N, Muramatsu Y, Nishimura M, Yoshida Y, Saito A, Yokoyama T, Furukawa T, Horii A, Hashimoto M, Asai A, Kirino T, Hamada H (1999) Adenovirus-mediated transfer of p33ING1 with p53 drastically augments apoptosis in gliomas. Cancer Res 59:5521–5528
Shimada H, Liu TL, Ochiai T, Shimizu T, Haupt Y, Hamada H, Abe T, Oka M, Takiguchi M, Hiwasa T (2002) Facilitation of adenoviral wild-type p53-induced apoptotic cell death by overexpression of p33ING1 in T.Tn human esophageal carcinoma cells. Oncogene 21:1208–1216
Nagashima M, Shiseki M, Pedeux RM, Okamura S, Kitahama-Shiseki M, Miura K, Yokota J, Harris CC (2003) A novel PHD-finger motif protein, p47ING3, modulates p53-mediated transcription, cell cycle control, and apoptosis. Oncogene 22:343–350
Cheung KJ, Li G (2002) p33ING1 enhances UVB-induced apoptosis in melanoma cells. Exp Cell Res 279:291–298
Cheung K-J Jr, Mitchell D, Lin P, Li G (2001) The tumor suppressor candidate p33ING1 mediates repair of UV-damaged DNA. Cancer Res 61:4974–4977
Kuo W-HW, Wang Y, Wong RPC, Campos EI, Li G (2007) The ING1b tumor suppressor facilitates nucleotide excision repair by promoting chromatin accessibility to XPA. Exp Cell Res 313:1628–1638
Wang Y, Wang J, Li G (2006) Leucine zipper-like domain is required for tumor suppressor ING2-mediated nucleotide excision repair and apoptosis. FEBS Lett 580:3787–3793
Wang J, Chin MY, Li G (2006) The novel tumor suppressor p33ING2 enhances nucleotide excision repair via inducement of histone H4 acetylation and chromatin relaxation. Cancer Res 66:1906–1911
Doyon Y, Cayrou C, Ullah M, Landry A-J, Côté V, Selleck W, Lane WS, Tan S, Yang X-J, Côté J (2006) ING tumor suppressor proteins are critical regulators of chromatin acetylation required for genome expression and perpetuation. Mol Cell 21:51–64
Loewith R, Meijer M, Lees-Miller SP, Riabowol K, Young D (2000) Three yeast proteins related to the human candidate tumor suppressor p33(ING1) are associated with histone acetyltransferase activities. Mol Cell Biol 20:3807–3816
Menendez C, Abad M, Gomez-Cabello D, Moreno A, Palmero I (2009) ING proteins in cellular senescence. Curr Drug Targets 10:406–417
Shah S, Riabowol K (2009) Signaling pathways of the ING proteins in apoptosis. Curr Drug Targets 10:385–391
Gunduz M, Ouchida M, Fukushima K, Hanafusa H, Etani T, Nishioka S, Nishizaki K, Shimizu K (2000) Genomic structure of the human ING1 gene and tumor-specific mutations detected in head and neck squamous cell carcinomas. Cancer Res 60:3143–3146
Ding H, Yang D, Wu FR, Li H, Feng K, Zheng SJ, Ni CR, Yu GZ (2003) P33(ING1) gene expression and mutation in stomach cancer tissues and precarcinomatous tissues. Zhonghua Yi Xue Za Zhi 83:320–323
Chen L, Wei JB, Zhou YC, Zhang S, Liang JL, Cao YF, Tang ZJ, Zhang XL, Gao F (2005) Genetic alterations and expression of inhibitor of growth 1 in human sporadic colorectal cancer. World J Gastroenterol 11:6120–6124
Nouman G, Anderson JJ, Wood KM, Lunec J, Hall AG, Reid MM, Angus B (2002) Loss of nuclear expression of the p33ING1b inhibitor of growth protein in childhood acute lymphoblastic leukaemia. J Clin Pathol 55:596–601
Nouman G, Anderson JJ, Crosier S, Shrimankar J, Lunec J, Angus B (2003) Downregulation of nuclear expression of the p33ING1b inhibitor of growth protein in invasive carcinoma of the breast. J Clin Pathol 56:507–511
Ohgi T, Masaki T, Nakai S, Morishita A, Yukimasa S, Nagai M, Miyauchi Y, Funaki T, Kurokohchi K, Watanabe S, Kuriyama S (2002) Expression of p33(ING1) in hepatocellular carcinoma: relationships to tumour differentiation and cyclin E kinase activity. Scand J Gastroenterol 37:1440–1448
Oki E, Maehara Y, Tokunaga E, Kakeji Y, Sugimachi K (1999) Reduced expression of p33(ING1) and the relationship with p53 expression in human gastric cancer. Cancer Lett 147:157–162
Tallen G, Kaiser I, Krabbe S, Lass U, Hartmann C, Henze G, Riabowol K, von Deimling A (2004) No ING1 mutations in human brain tumours but reduced expression in high malignancy grades of astrocytoma. Int J Cancer 109:476–479
Tokunaga E, Maehara Y, Oki E, Kitamura K, Kakeji Y, Ohno S, Sugimachi K (2000) Diminished expression of ING1 mRNA and the correlation with p53 expression in breast cancers. Cancer Lett 152:15–22
Zhu Z, Luo Z, Li Y, Ni C, Li H, Zhu M (2009) Human inhibitor of growth 1 inhibits hepatoma cell growth and influences p53 stability in a variant-dependent manner. Hepatology 49:504–512
Vieyra D, Loewith R, Scott M, Bonnefin P, Boisvert FM, Cheema P, Pastyryeva S, Meijer M, Johnston RN, Bazett-Jones DP, McMahon S, Cole MD, Young D, Riabowol K (2002) Human ING1 proteins differentially regulate histone acetylation. J Biol Chem 277:29832–29839
Scott M, Bonnefin P, Vieyra D, Boisvert F-M, Young D, Bazett-Jones DP, Riabowol K (2001) UV-induced binding of ING1 to PCNA regulates the induction of apoptosis. J Cell Sci 114:3455–3462
Skowyra D, Zeremski M, Nezanov N, Li M, Choi Y, Uesugi M, Hauser CA, Gu W, Gudkov AV, Qin J (2001) Differential association of products of alternative transcripts of the candidate tumor suppressor ING1 with the mSin3/HDAC1 transcriptional corepressor complex. J Biol Chem 276:8734–8739
Zeremski M, Hill JE, Kwek SSS, Grigorian IA, Gurova KV, Garkavtsev IV, Diatchenko L, Koonin EV, Gudkov AV (1999) Structure and regulation of the mouse ING1 gene. J Biol Chem 274:32172–32181
Toyama T, Hirotaka I, Watson P, Muzik H, Saettler E, Magliocco A, DiFrancesco L, Forsyth P, Garkavtsev I, Kobayashi S, Riabowol K (1999) Suppression of ING1 expression in sporadic breast cancer. Oncogene 18:5187–5193
Ahmed I, Kelly S, Anderson J, Angus B, Challen C, Lunec J (2007) The predictive value of p53 and p33ING1b in patients with Dukes’C colorectal cancer. Colorectal Dis 10:344–351
Nouman G, Anderson JJ, Mathers ME, Leonard N, Crosier S, Lunec J, Angus B (2002) Nuclear to cytoplasmic compartment shift of the p33ING1b tumour suppressor protein is associated with malignancy in melanocytic lesions. Histopathology 40:360–366
Zhang J, Wang DW, Li QX, Zhu ZL, Wang MW, Cui DS, Yang YH, Gu YX, Sun XF (2008) Nuclear to cytoplasmic shift of p33ING1b protein from normal oral mucosa to oral squamous cell carcinoma in relation to clinicopathological variables. J Cancer Res Clin Oncol 134:421–426
Gong W, Russell M, Suzuki K, Riabowol K (2006) Subcellular targeting of p33ING1b by phosphorylation-dependent 14–3-3 binding regulates p21WAF1 expression. Mol Cell Biol 26:2947–2954
Cheung K, Li G (2002) The tumour suppressor p33ING1 does not regulate migration and angiogenesis in melanoma cells. Int J Oncol 21:1361–1365
Chin MY, Ng KCP, Li G (2005) The novel tumor suppressor p33ING2 enhances UVB-induced apoptosis in human melanoma cells. Exp Cell Res 304:531–543
Nagashima M, Shiseki M, Miura K, Hagiwara K, Linke SP, Pedeux R, Wang XW, Yokota J, Riabowol K, Harris CC (2001) DNA damage-inducible gene p33ING2 negatively regulates cell proliferation through acetylation of p53. Proc Natl Acad Sci USA 98:9671–9676
Unoki M, Kumamoto K, Robles AI, Shen JC, Zheng Z-M, Harris CC (2008) A novel ING2 isoform, ING2b, synergizes with ING2a to prevent cell cycle arrest and apoptosis. FEBS Lett 582:3868–3874
Ythier D, Brambilla E, Binet R, Nissou D, Vesin A, de Fraipont F, Moro-Sibilot D, Lantuejoul S, Brambilla C, Gazzeri S, Pedeux R (2009) Expression of candidate tumor suppressor gene ING2 is lost in non-small cell lung carcinoma. Lung Cancer (in press)
Kumamoto K, Fujita K, Kurotani R, Saito M, Unoki M, Hagiwara N, Shia H, Bowman ED, Yanaihara N, Okamura S, Nagashima M, Miyamoto K, Takenoshita S, Yokota J, Harris CC (2009) ING2 is upregulated in colon cancer and increases invasion by enhanced mmp13 expression. Int J Cancer 125:1306–1315
Borkosky S, Gunduz M, Nagatsuka H, Beder LB, Gunduz E, Ali MAL, Rodriguez AP, Cilek MZ, Tominaga S, Yamanada N, Shimizu K, Nagai N (2009) Frequent deletion of ING2 locus at 4q35.1 associates with advanced tumor stage in head and neck squamous cell carcinoma. J Cancer Res Clin Oncol 135:703–713
Lu F, Dai DL, Martinka M, Ho V, Li G (2006) Nuclear ING2 expression is reduced in human cutaneous melanomas. Br J Cancer 95:80–86
Okano T, Gemma A, Hosoya Y, Hosomi Y, Nara M, Kokubo Y, Yoshimura A, Shibuya M, Nagashima M, Harris CC, Kudoh S (2006) Alterations in novel candidate tumor suppressor genes, ING1, and ING2 in human lung cancer. Oncol Rep 15:545–549
Cetin E, Cengiz B, Gunduz E, Gunduz M, Nagatsuka H, Beder LB, Fukushima K, Pehlivan D, Ozaslan M, Yamanada N, Nishizaki K, Shimizu K, Nagai N (2008) Deletion mapping of chromosome 4q22–35 and identification of four frequently deleted regions in head and neck cancers. Neoplasma 55:299–304
Sironi E, Cerri A, Tomasini D, Sirchia SM, Porta G, Rossella F, Grati FR, Simoni G (2004) Loss of heterozygosity on chromosome 4q32–35 in sporadic basal cell carcinomas: evidence for the involvement of p33ING2/ING1L and SAP30 genes. J Cutan Pathol 31:318–322
Peña PV, Davrazou F, Shi X, Walter KL, Verkhusha VV, Gozani O, Zhao R, Kutateladze TG (2006) Molecular mechanism of histone H3K4me3 recognition by plant homeodomain of ING2. Nature 442:100–103
Luo J, Shah S, Riabowol K, Mains PE (2009) The caenorhabditis elegans ING-3 gene regulates ionizing radiation-induced germ-cell apoptosis in a p53-associated pathway. Genetics 181:473–482
Wang Y, Li G (2006) ING3 promotes UV-induced apoptosis via Fas/caspase-8 pathway in melanoma cells. J Biol Chem 281:11887–11893
Wang Y, Dai DL, Martinka M, Li G (2007) Prognostic significance of nuclear ING3 expression in human cutaneous melanoma. Clin Cancer Res 13:4111–4116
Chen G, Wang Y, Garate M, Zhou J, Li G (2009) The tumor suppressor ING3 is degraded by SCFSkp2-mediated ubiquitin-proteasome system. Oncogene (in press)
Hung W-C, Tseng W-L, Shiea J, Chang H-C (2009) Skp2 overexpression increases the expression of MMP-2 and MMP-9 and invasion of lung cancer cells. Cancer Lett (in press)
Wang X, Wu YP, Ye B, Lin DC, Feng YB, Zhang ZQ, Xu X, Han YL, Cai Y, Dong JT, Zhan QM, Wu M, Wang MR (2009) Suppression of anoikis by Skp2 amplification and overexpression promotes metastasis of esophageal squamous cell carcinoma. Mol Cancer Res 7:12–22
Liao Q, Zhong D, Chen Q (2008) Protein expression of Skp2 in osteosarcoma and its relation with prognosis. Zhong Nan Da Xue Xue Bao Yi Xue Ban 33:606–611
Fang F-M, Chien C-Y, Li C-F, Shiu W-Y, Chen C-H, Huang H-Y (2009) Effect of S-phase kinase-associated protein 2 expression on distant metastasis and survival in nasopharyngeal carcinoma patients. Int J Rad Oncol Biol Phys 73:202–207
Zhao J, Yang CL, Zhao SY (2007) Expression of Skp2 protein in lung carcinoma and its implication for prognosis. Zhonghua Zhong Liu Za Zhi 29:289–292
Einama T, Kagata Y, Tsuda H, Morita D, Ogata S, Ueda S, Takigawa T, Kawarabayashi N, Fukatsu K, Sugiura Y, Matsubara O, Hatsuse K (2006) High-level Skp2 expression in pancreatic ductal adenocarcinoma: correlation with the extent of lymph node metastasis, higher histological grade, and poorer patient outcome. Pancreas 32:376–381
Sui L, Dong Y, Watanabe Y, Yamaguchi F, Sugimoto K, Tokuda M (2006) Clinical significance of Skp2 expression, alone and combined with Jab1 and p27 in epithelial ovarian tumors. Oncol Rep 15:765–771
Harada K, Supriatno K, Kawaguchi S, Kawashima Y, Itashiki Y, Yoshida H, Sato M (2005) High expression of S-phase kinase-associated protein 2 (Skp2) is a strong prognostic marker in oral squamous cell carcinoma patients treated by UFT in combination with radiation. Anticancer Res 25:2471–2475
Kamata Y, Watanabe J, Nishimura Y, Arai T, Kawaguchi M, Hattori M, Obokata A, Kuramoto H (2005) High expression of Skp2 correlates with poor prognosis in endometrial endometrioid adenocarcinoma. J Cancer Res Clin Oncol 131:591–596
Li H, Jiang X, Zhou X (2005) Expression of Skp2 and p27 proteins in laryngeal squamous cell carcinoma and their significance. Lin Chuang Er Bi Yan Hou Ke Za Zhi 19:409–411
Zheng W-Q, Zheng J-M, Ma R, Meng F-F, Ni C-R (2005) Relationship between levels of Skp2 and p27 in breast carcinomas and possible role of Skp2 as targeted therapy. Steroids 70:770–774
Takanami I (2005) The prognostic value of overexpression of Skp2 mRNA in non-small cell lung cancer. Oncol Rep 13:727–731
Yokoi S, Yasui K, Mori M, Iizasa T, Fujisawa T, Inazawa J (2004) Amplification and overexpression of Skp2 are associated with metastasis of non-small-cell lung cancers to lymph nodes. Am J Pathol 165:175–180
Li J, Masaki T, Kubo A, Fujita J, Dixon DA, Beauchamp RD, Ishida T, Kuriyama S, Imaida K (2004) Correlation of Skp2 with carcinogenesis, invasion, metastasis, and prognosis in colorectal tumors. Int J Oncol 25:87–95
Dong Y, Sui L, Watanabe Y, Sugimoto K, Tokuda M (2003) S-phase kinase-associated protein 2 expression in laryngeal squamous cell carcinomas and its prognostic implications. Oncol Rep 10:321–325
Gunduz M, Beder LB, Gunduz E, Nagatsuka H, Fukushima K, Pahlivan D, Cetin E, Yamanaka N, Nishizaki K, Shimizu K, Nagai N (2008) Downregulation of ING3 mRNA expression predicts poor prognosis in head and neck cancer. Cancer Sci 99:531–538
Gunduz M, Ouchida M, Fukushima K, Ito S, Jitsumori Y, Nakashima T, Nagai N, Nishizaki K, Shimizu K (2002) Allelic loss and reduced expression of the ING3, a candidate tumor suppressor gene at 7q31 in human head and neck cancers. Oncogene 21:4462–4470
Li M, Jin Y, Sun W-j, Yu Y, Bai J, Tong D-d, Qi J-p, Du J-r, Geng J-s, Huang Q, Huang X-y, Huang Y, Han F-f, Meng X-n, Rosales JL, Lee K-Y, Fu S-b (2009) Reduced expression and novel splice variants of ING4 in human gastric adenocarcinoma. J Pathol 219:87–95
Li X, Cai L, Chen H, Zhang Q, Zhang S, Wang Y, Dong Y, Cheng H, Qi J (2009) Inhibitor of growth 4 induces growth suppression and apoptosis in glioma U87MG. Pathobiology 76:181–192
Li J, Martinka M, Li G (2008) Role of ING4 in human melanoma cell migration, invasion and patient survival. Carcinogenesis 29:1373–1379
Cai L, Li X, Zheng S, Wang Y, Wang Y, Li H, Yang J, Sun J (2009) Inhibitor of growth 4 is involved in melanomagenesis and induces growth suppression and apoptosis in melanoma cell line M14. Melanoma Res 19:1–7
Gunduz M, Nagatsuka H, Demircan K, Gunduz E, Cengiz B, Ouchida M, Tsujigiwa H, Yamachika E, Fukushima K, Beder L, Hirohata S, Ninomiya Y, Nishizaki K, Shimizu K, Nagai N (2005) Frequent deletion and down-regulation of ING4, a candidate tumor suppressor gene at 12p13, in head and neck squamous cell carcinomas. Gene 356:109–117
Xie Y, Zhang H, Shen W, Xiang J, Ye Z, Yang J (2008) Adenovirus-mediated ING4 expression suppresses lung carcinoma cell growth via induction of cell cycle alteration and apoptosis and inhibition of tumor invasion and angiogenesis. Cancer Lett 271:105–116
Li X, Cai L, Liang M, Wang Y, Yang J, Zhao Y (2008) ING4 induces cell growth inhibition in human lung adenocarcinoma A549 cells by means of Wnt-1/beta-Catenin signaling pathway. Anatomical Rec 291:593–600
Zhang X, Xu L-S, Wang Z-Q, Wang K-S, Li N, Cheng Z-H, Huang S-Z, Wei D-Z, Han Z-G (2004) ING4 induces G2/M cell cycle arrest and enhances the chemosensitivity to DNA-damage agents in HepG2 cells. FEBS Lett 570:7–12
Fang F, Luo LB, Tao YM, Wu F, Yang LY (2009) Decreased expression of inhibitor of growth 4 correlated with poor prognosis of hepatocellular carcinoma. Cancer Epidemiol Biomarkers Prev 18:409–416
Kim S, Chin K, Gray JW, Bishop JM (2004) A screen for genes that suppress loss of contact inhibition: identification of ING4 as a candidate tumor suppressor gene in human cancer. Proc Natl Acad Sci USA 101:16251–16256
Kim S (2005) HuntING4 new tumor suppressors. Cell Cycle 4:516–517
Hung T, Binda O, Champagne KS, Kuo AJ, Johnson K, Chang HY, Simon MD, Kutateladze TG, Gozani O (2009) ING4 mediates crosstalk between histone H3K4 trimethylation and H3 acetylation to attenuate cellular transformation. Mol Cell 33:248–256
Ozer A, Bruik RK (2005) Regulation of HIF by prolyl hydroxylases: recruitment of the candidate tumor suppressor protein ING4. Cell Cycle 4:1153–1156
Ozer A, Wu LC, Bruick RK (2005) The candidate tumor suppressor ING4 represses activation of the hypoxia inducible factor (HIF). Proc Natl Acad Sci USA 102:7481–7486
Colla S, Tagliaferri S, Morandi F, Lunghi P, Donofrio G, Martorana D, Mancini C, Lazzaretti M, Mazzera L, Ravanetti L, Bonomini S, Ferrari L, Miranda C, Ladetto M, Neri TM, Neri A, Greco A, Mangoni M, Bonati A, Rizzoli V, Guiliani N (2007) The new tumor-suppressor gene inhibitor of growth family member 4 (ING4) regulates the production of proangiogenic molecules by myeloma cells and suppresses hypoxia-inducible factor-1 alpha (HIF-1 alpha) activity: involvement in myeloma-induced angiogenesis. Blood 110:4464–4475
Garkavtsev I, Kozin SV, Chernova O, Xu L, Winkler F, Brown E, Barnett GH, Jain RK (2004) The candidate tumour suppressor protein ING4 regulates brain tumour growth and angiogenesis. Nature 428:328–332
Nozell S, Laver T, Moseley D, Nowoslawski L, DeVos M, Atkinson GP, Harrison K, Nabors LB, Benveniste EN (2008) The ING4 tumor suppressor attenuates NF-κB activity at the promoters of target genes. Mol Cell Biol 28:6632–6645
Gadea G, deToledo M, Anguille C, Roux P (2007) Loss of p53 promotes Rhoa-ROCK-dependent cell migration and invasion in 3D matrices. J Cell Biol 178:23–30
Gao K, Dai DL, Martinka M, Li G (2006) Prognostic significance of nuclear factor-kappaB p105/p50 in human melanoma and its role in cell migration. Cancer Res 66:8382–8388
Shiseki M, Nagashima M, Pedeux RM, Kitahama-Shiseki M, Miura K, Okamura S, Onogi H, Higashimoto Y, Appella E, Yokota J, Harris CC (2003) P29ING4 and p28ING5 bind to p53 and p300, and enhance p53 activity. Cancer Res 63:2373–2378
Cengiz B, Gunduz M, Nagatsuka H, Beder L, Gunduz E, Tamamura R, Mahmut N, Fukushima K, Ali MAS, Naomoto Y, Shimizu K, Nagai N (2007) Fine deletion mapping of chromosome 2q21–37 shows three preferentially deleted regions in oral cancer. Oral Oncol 43:241–247
Shen J, Unoki M, Ythier D, Duperray A, Varticovski L, Kumamoto K, Pedeux R, Harris CC (2007) Inhibitor of growth 4 suppresses cell spreading and cell migration by interacting with novel binding partner, liprin alpha 1. Cancer Res 67:2552–2558
Unoki M, Shen JC, Zheng ZM, Harris CC (2006) Novel splice variants of ING4 and their possible roles in the regulation of cell growth and motility. J Biol Chem 281:34677–34686
Acknowledgments
This work was supported by Canadian Institutes of Health Research (MOP-84559 and MOP-93810) and Canadian Dermatology Foundation to G. Li. B. Piche is a recipient of the trainee award from CIHR Skin Research Training Centre.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piche, B., Li, G. Inhibitor of growth tumor suppressors in cancer progression. Cell. Mol. Life Sci. 67, 1987–1999 (2010). https://doi.org/10.1007/s00018-010-0312-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-010-0312-z