Abstract
Background
The primary objective of the study was to identify early hematologic changes predictive for radiotherapy (RT)-associated genitourinary and gastrointestinal toxicity.
Methods
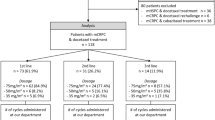
In a group of 91 prostate cancer patients presenting for primary (n = 51) or postoperative (n = 40) curative RT, blood samples (blood count, acute phase proteins, and cytokines) were analyzed before (T1), three times during (T2–T4), and 6–8 weeks after (T5) radiotherapy. Before RT (baseline), on the last day (acute toxicity), a median of 2 months and 16 months (late toxicity) after RT, patients responded to a validated questionnaire (Expanded Prostate Cancer Index Composite). Acute score changes > 20 points and late changes > 10 points were considered clinically relevant.
Results
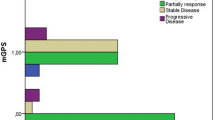
Radiotherapy resulted in significant changes of hematologic parameters, with the largest effect on lymphocytes (mean decrease of 31–45 %) and significant dependence on target volume. C-reactive protein (CRP) elevation > 5 mg/l and hemoglobin level decrease ≥ 5 G/1 at T2 were found to be independently predictive for acute urinary toxicity (p < 0.01, respectively). CRP elevation was predominantly detected in primary prostate RT (p = 0.02). Early lymphocyte level elevation ≥ 0.3G/l at T2 was protective against late urinary and bowel toxicity (p = 0.02, respectively). Other significant predictive factors for late bowel toxicity were decreasing hemoglobin levels (cut-off ≥ 5 G/l) at T2 (p = 0.04); changes of TNF-α (tumor necrosis factor; p = 0.03) and ferritin levels (p = 0.02) at T5. All patients with late bowel toxicity had interleukin (IL)-6 levels < 1.5 ng/l at T2 (63 % without; p = 0.01).
Conclusion
Early hematologic changes during prostate cancer radiotherapy are predictive for late urinary and bowel toxicity.
Zusammenfassung
Hintergrund
Das primäre Ziel der Studie war die Identifikation von frühen hämatologischen Veränderungen mit prädiktiver Bedeutung für radiotherapieassoziierte genitourinale und gastrointestinale Toxizität.
Methoden
In einer Gruppe von 91 Patienten mit einem Prostatakarzinom, die sich zur primären (n = 51) oder postoperativen (n = 40) Radiotherapie (RT) vorgestellt haben, wurden Blutproben (Blutbild, Akute-Phase-Proteine und Zytokine) vor (T1), dreimal während (T2–T4) und 6–8 Wochen nach RT (T5) untersucht. Vor RT (Ausgangsbefund), am letzten Tag (akute Toxizität), median 2 Monate und 16 Monate (Spättoxizität) nach RT haben die Patienten einen validierten Fragebogen beantwortet (Expanded Prostate Cancer Index Composite). Akute Scoreänderungen > 20 Punkte und späte Änderungen > 10 Punkte wurden als klinisch relevant bewertet.
Ergebnisse
Die Radiotherapie führte zu signifikanten Veränderungen der hämatologischen Parameter, mit dem größten Effekt auf Lymphozyten (mittlerer Abfall von 31–45 %) und signifikanter Abhängigkeit vom Zielvolumen. Eine Erhöhung des C-reaktiven Proteins (CRP) auf > 5 mg/l und ein Hämoglobinabfall ≥ 5 G/l zum Zeitpunkt T2 waren unabhängig prädiktiv für akute Miktionsbeschwerden (jeweils p < 0,01). Eine CRP-Erhöhung fand sich vorwiegend bei primärer RT der Prostata (p = 0,02). Ein früher Lymphozytenanstieg ≥ 0,3G/l bei T2 war protektiv gegen späte Miktions- und Darmbeschwerden (jeweils p = 0,02). Weitere signifikante prädiktive Faktoren für späte Darmbeschwerden waren abfallende Hämoglobinwerte (Grenzwert ≥ 5 G/l) bei T2 (p = 0,04); Veränderungen der TNFα- (Tumornekrosefaktor; p = 0,03) und Ferritinspiegel (p = 0,02) bei T5. Alle Patienten mit späten Darmbeschwerden hatten Interleukin-(IL-)6-Werte < 1,5 ng/l bei T2 (vs. 63 % ohne Darmbeschwerden; p = 0,01).
Schlussfolgerung
Frühe hämatologische Veränderungen während der Radiotherapie beim Prostatakarzinom sind prädiktiv für späte Miktions- und Darmbeschwerden.
Similar content being viewed by others
References
Blank KR, Cascardi MA, Kao GD (1999) The utility of serial complete blood count monitoring in patients receiving radiation therapy for localized prostate cancer. Int J Radiat Oncol Biol Phys 44:317–321
Bolla M, Van Poppel H, Tombal B et al (2012) Postoperative radiotherapy after radical prostatectomy for high-risk prostate cancer: long-term results of a randomised controlled trial (EORTC trial 22911). Lancet 380:2018–2027
Budäus L, Bolla M, Bossi A et al (2012) Functional outcomes and complications following radiation therapy for prostate cancer: a critical analysis of the literature. Eur Urol 61:112–127
Christensen E, Pintilie M, Evans KR et al (2009) Longitudinal cytokine expression during IMRT for prostate cancer and acute treatment toxicity. Clin Cancer Res 15:5576–5583
Christiansen H, Saile B, Hermann RM et al (2007) Increase of hepcidin plasma and urine levels is associated with acute proctitis and changes in hemoglobin levels in primary radiotherapy for prostate cancer. J Cancer Res Clin Oncol 133:297–304
Heidenreich A, Bellmunt J, Bolla M et al (2011) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and treatment of clinically localised disease. Eur Urol 59:61–71
Henriquez-Hernandez LA, Bordon E, Pinar B et al (2012) Prediction of normal tissue toxicity as part of the individualized treatment with radiotherapy in oncology patients. Surg Oncol 21:201–206
Indaram AV, Visvalingam V, Locke M et al (2000) Mucosal cytokine production in radiation-induced proctosigmoiditis compared with inflammatory bowel disease. Am J Gastroenterol 95:1221–1225
Kim YJ, Cho KH, Pyo HR et al (2014) Radical prostatectomy versus external beam radiotherapy for localized prostate cancer: comparison of treatment outcomes. Strahlenther Onkol (in press) published in 2015; Strahlenther Onkol 191: 321-329
Koc M, Taysi S, Sezen O et al (2003) Levels of some acute-phase proteins in the serum of patients with cancer during radiotherapy. Biol Pharm Bull 26:1494–1497
Li LY, Yang M, Gao X et al (2009) Prospective comparison of five mediators of the systemic response after high-intensity focused ultrasound and targeted cryoablation for localized prostate cancer. BJU Int 104:1063–1067
Lohm G, Lütcke J, Jamil B et al (2014) Salvage radiotherapy in patients with prostate cancer and biochemical relapse after radical prostatectomy: long-term follow-up of a single center survey. Strahlenther Onkol 190:727–731
Lopes CO, Callera F (2012) Three-dimensional conformal radiotherapy in prostate cancer patients: rise in interleukin 6 (IL-6) but not IL-2, IL-4, IL-5, tumor necrosis factor-alpha, MIP-1-alpha, and LIF levels. Int J Radiat Oncol Biol Phys 82:1385–1388
Osoba D, Rodrigues G, Myles J et al (1998) Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol 16:139–144
Peinemann F, Grouven U, Bartel C et al (2011) Permanent interstitial low-dose-rate brachytherapy for patients with localised prostate cancer: a systematic review of randomised and nonrandomised controlled clinical trials. Eur Urol 60:881–893
Petersen SE, Bentzen L, Emmertsen KJ et al (2014) Development and validation of a scoring system for late anorectal side-effects in patients treated with radiotherapy for prostate cancer. Radiother Oncol 111:94–99
Pinkawa M, Holy R, Piroth MD et al (2010) Consequential late effects after radiotherapy for prostate cancer—a prospective longitudinal quality of life study. Radiat Oncol 5:27
Pinkawa M, Eble MJ, Mottaghy FM (2011) PET and PET/CT in radiation treatment planning for prostate cancer. Expert Rev Anticancer Ther 11:1033–1039
Pinkawa M, Holy R, Piroth DM et al (2011) Intensity-modulated radiotherapy for prostate cancer implementing molecular imaging with 18F-choline PET-CT to define a simultaneous integrated boost. Strahlenther Onkol 186:600–606
Pinkawa M, Djukic V, Klotz J et al (2014) Hematologic chnages during radiation therapy are dependent on treatment volume. Future Oncol 10:835–843
Pinkawa M, Schubert C, Excobar-Corral N et al (2014) Application of a hydrogel spacer for postoperative slavage radiotherapy of prostate cancer. Strahlenther Onkol (in press) published in 2015: Strahlenther Onkol 191: 375-379
Schaake W, Wiegman EM, de Groot M et al (2014) The impact of gastrointestinal and genitourinary toxicity on health related quality of life among irradiated prostate cancer patients. Radiother Oncol 110:284–290
Thompson IM, Tangen CM, Paradelo J et al (2009) Adjuvant radiotherapy for pathological T3N0M0 prostate cancer significantly reduces risk of metastases and improves survival: long-term followup of a randomized clinical trial. J Urol 181:956–962
Wei JT, Dunn RL, Litwin MS et al (2000) Development and validation of the expanded prostate cancer index composite (EPIC) for comprehensive assessment of health-related quality of life in men with prostate cancer. Urology 56:899–905
Wiegel T, Bottke D, Steiner U et al (2009) Phase III postoperative adjuvant radiotherapy after radical prostatectomy compared with radical prostatectomy alone in pT3 prostate cancer with postoperative undetectable prostate-specific antigen: ARO 96-02/AUO AP 09/95. J Clin Oncol 27:2924–2930
Zachariah B, Jacob SS, Gwede C et al (2001) Effect of fractionated regional external beam radiotherapy on peripheral blood cell count. Int J Radiat Oncol Biol Phys 50:465–472
Acknowledgments
This work was supported by Philips Research, Aachen, Germany
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. Pinkawa, C. Ribbing, V. Djukic, J. Klotz, R. Holy, and M.J. Eble state that there are no conflicts of interest.
All studies on humans described in the present manuscript were carried out with the approval of the responsible ethics committee and in accordance with national law and the Helsinki Declaration of 1975 (in its current, revised form). Informed consent was obtained from all patients included in studies.
Rights and permissions
About this article
Cite this article
Pinkawa, M., Ribbing, C., Djukic, V. et al. Early hematologic changes during prostate cancer radiotherapy predictive for late urinary and bowel toxicity. Strahlenther Onkol 191, 771–777 (2015). https://doi.org/10.1007/s00066-015-0841-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-015-0841-3