Abstract
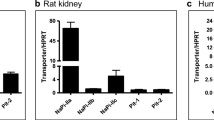
This study was designed to evaluate the expression and function of the organic anion transporters, Oat1 and Oat3, in rats exposed to a nephrotoxic dose of HgCl2. Oat1 protein expression increased in renal homogenates and decreased in renal basolateral membranes from HgCl2 rats, while Oat3 protein abundance decreased in both kidney homogenates and basolateral membranes. The lower protein levels of Oat1 and Oat3 in basolateral membranes explain the lower uptake capacity for p-aminohippurate (in vitro assays) and the diminution of the systemic clearance of this organic anion (in vivo studies) observed in treated rats. Since both transporters mediate mercury access to the renal cells, their down-regulation in basolateral membranes might be a defensive mechanism developed by the cell to protect itself against mercury injury. The pharmacological modulation of the expression and/or the function of Oat1 and Oat3 might be an effective therapeutic strategy for reducing the nephrotoxicity of mercury.






Similar content being viewed by others
References
Aleo MF, Morandini F, Bettoni F et al (2005) Endogenous thiols and MRP transporters contribute to Hg2+ efflux in HgCl2-treated tubular MDCK cells. Toxicology 206:137–151
Anzai N, Kanai Y, Endou H (2006) Organic anion transporter family: current knowledge. J Pharmacol Sci 100:411–426
Brady HR, Brenner BM, Lieberthal W (2004) Acute renal failure. In: Brenner BB, Rector FC (eds) The kidney, 7th edn. Saunders, Philadelphia, pp 1215–1292
Brandoni A, Quaglia NB, Torres AM (2003) Compensation increase in organic anion excretion in rats with acute biliary obstruction: role of the renal organic anion transporter 1. Pharmacology 68:57–63
Brandoni A, Anzai N, Kanai Y et al (2006a) Renal elimination of p-aminohippurate (PAH) in response to three days of biliary obstruction in the rat. The role of OAT1 and OAT3. Biochim Biophys Acta 1762:673–682
Brandoni A, Villar SR, Picena JC et al (2006b) Expression of rat renal cortical OAT1 and OAT3 in response to acute biliary obstruction. Hepatology 43:1092–1100
Bridges CC, Bauch C, Verrey F et al (2004) Mercuric conjugates of cysteine are transported by the amino acid transporter system b(0, +): implications of molecular mimicry. J Am Soc Nephrol 15:663–673
Bridges CC, Zalups RK (2005) Molecular and ionic mimicry and the transport of toxic metals. Toxicol Appl Pharmacol 204:274–308
Bridges CC, Joshee L, Zalups RK (2008) Multidrug resistance proteins and the renal elimination of inorganic mercury mediated by 2,3-dimercaptopropane-1-sulfonic acid and meso-2,3-dimercaptosuccinic acid. J Pharmacol Exp Ther 324:383–390
Cerrutti JA, Quaglia NB, Torres AM (2001) Characterization of the mechanisms involved in the gender differences in p-aminohippurate renal elimination in rats. Can J Physiol Pharmacol 79:805–813
Clarkson TW (1997) The toxicology of mercury. Crit Rev Clin Lab Sci 34:369–403
Denk GU, Soroka CJ, Takeyama Y et al (2004) Multidrug resistance-associated protein 4 is up-regulated in liver but down-regulated in kidneys in obstructive cholestasis in the rat. J Hepatol 40:585–591
Di Giusto G, Anzai N, Endou H et al (2008) Elimination of organic anions in response to an early stage of renal ischemia-reperfusion in the rat. Role of basolateral plasma membrane transporters and cortical renal blood flow. Pharmacology 81:127–136
Di Giusto G, Anzai N, Endou H et al (2009) Oat5 and NaDC1 protein abundances in kidney and urine following renal ischemic reperfusion injury. J Histochem Cytochem 57:17–27
Drasch G, O’ Bose, Reilly S et al (2001) The Mt. Diwata study on the Philippines 1999 assessing mercury intoxication of the population by small scale gold mining. Sci Total Environ 267:151–158
Girardi G, Torres AM, Elías MM (1989) The implication of renal glutathione levels in mercuric chloride nephrotoxicity. Toxicology 58:187–195
Green J, Abassi Z, Winaver J (2000) Acute renal failure: clinical and pathophysiologic aspects. In: Seldin DW, Giebisch G et al (eds) The kidney: physiology and pathophysiology, 3rd edn. Philadelphia, Lippincott, Williams & Wilkins, pp 2329–2373
Jensen RE, Berndt WO (1988) Epinephrine and norephinephrine enhance p-aminohippurate transport into basolateral membrane vesicles. J Pharmacol Exp Ther 244:543–549
Kojima R, Sekine T, Kawachi M et al (2002) Immunolocalization of multispecific organic anion transporters, OAT1, OAT2, and OAT3, in rat kidney. J Am Soc Nephrol 13:848–857
Magos L, Clarkson TW (2006) Overview of the clinical toxicity of mercury. Ann Clin Biochem 43:254–268
Motulsky H (1995) Using nonlinear regression to fit curves. In: Motulsky H (ed) Intuitive biostatistics. Oxford University Press, New York, pp 227–283
Pelis RM, Dangprapai Y, Wunz TM et al (2007) Inorganic mercury interacts with cysteine residues (C451 and C474) of hOCT2 to reduce its transport activity. Am J Physiol 292:F1583–F1591
Rizwan AN, Burckhardt G (2007) Organic anion transporters of the SLC22 family: biopharmaceutical, physiological, and pathological roles. Pharm Res 24:450–470
Sabolic I (2006) Common mechanisms in nephropathy induced by toxic metals. Nephron Physiol 104:107–114
Sedmak JJ, Grossberg SE (1977) A rapid, sensitive and versatile assay for protein using Coomassie Brilliant Blue G250. Anal Biochem 79:544–552
Shumaker R (1986) A basic interactive computer program for statistical and pharmacokinetic analysis of data. Drug Metab Rev 17:331–348
Stacchiotti A, Borsani E, Rodella L et al (2003) Dose-dependent mercuric chloride tubular injury in rat kidney. Ultrastruct Pathol 27:253–259
Torres AM, Mac Laughlin M, Muller A et al (2005) Altered renal elimination of organic anions in rats with chronic renal failure. Biochim Biophys Acta 1740:29–37
Villar SR, Brandoni A, Quaglia N et al (2004) Renal elimination of organic anions in rats with bilateral ureteral obstruction. Biochim Biophys Acta 1688:204–209
Villar SR, Brandoni A, Anzai N et al (2005) Altered expression of rat renal cortical OAT1 and OAT3 in response to bilateral ureteral obstruction. Kidney Int 68:2704–2713
Villar SR, Brandoni A, Torres AM (2008) Time course of organic anion excretion in rats with bilateral ureteral obstruction. Role of organic anion transporters (Oat1 and Oat3). Nephron Physiol 110:45–56
Waugh WH, Beall PT (1974) Simplified measurement of PAH and other arylamines in plasma and urine. Kidney Int 5:429–432
Wen JG, Frokiaer J, Jorgensen TM et al (1999) Obstructive nephropathy: an update of the experimental research. Urol Res 27:29–39
Wright SH, Dantzler WH (2004) Molecular and cellular physiology of renal organic cation and anion transport. Physiol Rev 84:987–1049
Zalups RK (1998a) Basolateral uptake of inorganic mercury in the kidney. Toxicol Appl Pharmacol 150:1–8
Zalups RK (1998b) Basolateral uptake of mercuric conjugates of N-acetylcysteine and cysteine involves the organic anion transport system. J Toxicol Environ Health 54:101–117
Zalups RK (2000) Molecular interactions with mercury in the kidney. Pharmacol Rev 52:113–143
Zalups RK, Ahmad S (2004) Homocysteine and the renal epithelial transport and toxicity of inorganic mercury: role of basolateral transporter organic anion transporter 1. J Am Soc Nephrol 15:2023–2031
Zalups RK, Parks LD, Cannon VT et al (1998) Mechanisms of action of 2,3-dimercaptopropane-1-sulfonate and the transport, disposition, and toxicity of inorganic mercury in isolated perfused segments of rabbit proximal tubules. Mol Pharmacol 54:353–363
Acknowledgments
This study was supported by the following grants: FONCYT and CONICET. The authors thank to Prof. Héctor R. Girolami (Area Toxicología, Facultad de Ciencias Bioquímicas y Farmacéuticas, U.N.R.) for his important collaboration in this work and to Mrs Alejandra Martínez (Area Morfología, Facultad de Ciencias Bioquímicas y Farmacéuticas, U.N.R.) for her technical assistance with the histological studies. The authors also thank Wiener Lab Argentina for analytical kits.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Giusto, G., Anzai, N., Ruiz, M.L. et al. Expression and function of Oat1 and Oat3 in rat kidney exposed to mercuric chloride. Arch Toxicol 83, 887–897 (2009). https://doi.org/10.1007/s00204-009-0445-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-009-0445-8