Abstract
At the present, the term “glycoxidation” is recognized as the synergistic interaction between glycation and oxidative processes which, with the help of redox-active metals, consequently leads to the production of deleterious tissue modifications. The association between glycation and oxidation events is considered one of the major factors in the accumulation of non-functional damaged proteins, enhancing the oxidative damage at the cellular level. Because of the central role of insulin in the biology of diabetes, we investigated the site-specific oxidation of native and glycated insulin (mono, di, and tri-glycated forms), through metal-catalyzed oxidation, with a combination of liquid chromatography and mass spectrometry. With this approach we were able to identify the residues that were mainly oxidized, and peptide sequences resulting from oxidative cleavage of insulin. Tyrosine, phenylalanine, and cysteine were the main affected residues. Time-course analysis (0–48 h) of the oxidative damage enabled to detect more pronounced and earlier oxidative modifications in the case of glycated insulin. We also observed more severe oxidative damage as the number of glycation sites increased in insulin. These oxidative modifications included other oxidized residues, namely proline, histidine, valine, leucine, and glycine, which were shown to be carbonylated. In addition, we identified new sites of peptide cleavage with the formation of new fragments, derived mainly from chain B, which were both glycated and oxidatively modified. Peptide fragmentation occurred mainly between the residues phenylalanine, glycine, leucine, and tyrosine. Moreover, for diglycated and triglycated forms we observed further oxidative cleavage occurring in both chains, with oxidation and fragmentation of residues occurring near cysteine bridges, especially in chain A.

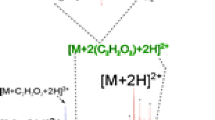
Schematic structure of glycated insulin showing modified residues resulting from oxidative damage marked with red circles and one possible glycated residues (bold and underlined). Two examples of tandem mass spectra that allowed the identification of the modified residues are shown.






Similar content being viewed by others
References
Beal MF (2002) Oxidatively modified proteins in aging and disease. Free Radic Biol Med 32:797–803
Levine RL, Stadtman ER (2001) Oxidative modification of proteins during aging. Exp Gerontol 36:1495–1502
Stadtman ER, Berlett BS (1997) Reactive oxygen-mediated protein oxidation in aging and disease. Chem Res Toxicol 10:485–494
Berlett BS, Stadtman ER (1997) Protein oxidation in aging, disease, and oxidative stress. J Biol Chem 272:20313–20316
Davies KJ (1987) Protein damage and degradation by oxygen radicals. I. general aspects. J Biol Chem 262:9895–9901
Ghezzi P, Bonetto V (2003) Redox proteomics: identification of oxidatively modified proteins. Proteomics 3:1145–1153
Mirzaei H, Regnier F (2008) Protein:protein aggregation induced by protein oxidation. J Chromatogr B 873:8–14
Bochkov VN, Leitinger N (2003) Anti-inflammatory properties of lipid oxidation products. J Mol Med 81:613–626
Nair U, Bartsch H, Nair J (2007) Lipid peroxidation-induced DNA damage in cancer-prone inflammatory diseases: a review of published adduct types and levels in humans. Free Radic Biol Med 43:1109–1120
Lovell MA, Markesbery WR (2007) Oxidative DNA damage in mild cognitive impairment and late-stage Alzheimer’s disease. Nucleic Acids Res 35:7497–7504
Davies MJ, Fu S, Wang H, Dean RT (1999) Stable markers of oxidant damage to proteins and their application in the study of human disease. Free Radic Biol Med 27:1151–1163
Bridgewater JD, Lim J, Vachet RW (2006) Transition metal–peptide binding studied by metal-catalyzed oxidation reactions and mass spectrometry. Anal Chem 78:2432–2438
Detweiler CD, Lardinois OM, Deterding LJ, de Montellano PR, Tomer KB, Mason RP (2005) Identification of the myoglobin tyrosyl radical by immuno-spin trapping and its dimerization. Free Radic Biol Med 38:969–976
Finch JW, Crouch RK, Knapp DR, Schey KL (1993) Mass spectrometric identification of modifications to human serum albumin treated with hydrogen peroxide. Arch Biochem Biophys 305:595–599
Garrison WM, Jayko ME, Bennett W (1962) Radiation-induced oxidation of protein in aqueous solution. Radiat Res 16:483–502
Inoue K, Garner C, Ackermann BL, Oe T, Blair IA (2006) Liquid chromatography/tandem mass spectrometry characterization of oxidized amyloid beta peptides as potential biomarkers of Alzheimer’s disease. Rapid Commun Mass Spectrom 20:911–918
Jain R, Freund HG, Budzinsky E, Sharma M (1997) Radiation-induced formation of 3, 4-dihydroxyphenylalanine in tyrosine-containing peptides and proteins as a function of X-irradiation dose. Bioconjug Chem 8:173–178
Kato Y, Kitamoto N, Kawai Y, Osawa T (2001) The hydrogen peroxide/copper ion system, but not other metal-catalyzed oxidation systems, produces protein-bound dityrosine. Free Radic Biol Med 31:624–632
Kato Y, Uchida K, Kawakishi S (1992) Oxidative fragmentation of collagen and prolyl peptide by Cu(II)/H2O2. Conversion of proline residue to 2-pyrrolidone. J Biol Chem 267:23646–23651
Kim J, Rodriguez ME, Guo M, Kenney ME, Oleinick NL, Anderson VE (2008) Oxidative modification of cytochrome c by singlet oxygen. Free Radic Biol Med 44:1700–1711
Li S, Nguyen TH, Schoneich C, Borchardt RT (1995) Aggregation and precipitation of human relaxin induced by metal-catalyzed oxidation. Biochemistry 34:5762–5772
Schoneich C, Williams TD (2002) Cu(II)-catalyzed oxidation of beta-amyloid peptide targets His13 and His14 over His6: Detection of 2-Oxo-histidine by HPLC–MS/MS. Chem Res Toxicol 15:717–722
Smith JB, Jiang X, Abraham EC (1997) Identification of hydrogen peroxide oxidation sites of alpha A- and alpha B-crystallins. Free Radic Res 26:103–111
Wright A, Bubb WA, Hawkins CL, Davies MJ (2002) Singlet oxygen-mediated protein oxidation: evidence for the formation of reactive side chain peroxides on tyrosine residues. Photochem Photobiol 76:35–46
Dalle-Donne I, Scaloni A, Giustarini D, Cavarra E, Tell G, Lungarella G, Colombo R, Rossi R, Milzani A (2005) Proteins as biomarkers of oxidative/nitrosative stress in diseases: the contribution of redox proteomics. Mass Spectrom Rev 24:55–99
Stadtman ER, Levine RL (2003) Free radical-mediated oxidation of free amino acids and amino acid residues in proteins. Amino Acids 25:207–218
Olivares-Corichi IM, Ceballos G, Ortega-Camarillo C, Guzman-Grenfell AM, Hicks JJ (2005) Reactive oxygen species (ROS) induce chemical and structural changes on human insulin in vitro, including alterations in its immunoreactivity. Front Biosci 10:838–843
Guedes S, Vitorino R, Domingues R, Amado F, Domingues P (2009) Oxidation of bovine serum albumin: identification of oxidation products and structural modifications. Rapid Commun Mass Spectrom 23:2307–2315
Schalkwijk CG, Brouwers O, Stehouwer CD (2008) Modulation of insulin action by advanced glycation endproducts: a new player in the field. Horm Metab Res 40:614–619
Abdel-Wahab YH, O’Harte FP, Boyd AC, Barnett CR, Flatt PR (1997) Glycation of insulin results in reduced biological activity in mice. Acta Diabetol 34:265–270
Boyd AC, Abdel-Wahab YH, McKillop AM, McNulty H, Barnett CR, O’Harte FP, Flatt PR (2000) Impaired ability of glycated insulin to regulate plasma glucose and stimulate glucose transport and metabolism in mouse abdominal muscle. Biochim Biophys Acta 1523:128–134
Ceriello A, Motz E (2004) Is oxidative stress the pathogenic mechanism underlying insulin resistance, diabetes, and cardiovascular disease? The common soil hypothesis revisited. Arterioscler Thromb Vasc Biol 24:816–823
Guedes S, Vitorino R, Domingues MR, Amado F, Domingues P (2009) Mass spectrometry characterization of the glycation sites of bovine insulin by tandem mass spectrometry. J Am Soc Mass Spectrom 20:1319–1326
Cheng RZ, Kawakishi S (1994) Site-specific oxidation of histidine residues in glycated insulin mediated by Cu2+. Eur J Biochem 223:759–764
O’Harte FP, Hojrup P, Barnett CR, Flatt PR (1996) Identification of the site of glycation of human insulin. Peptides 17:1323–1330
Guan JQ, Chance MR (2005) Structural proteomics of macromolecular assemblies using oxidative footprinting and mass spectrometry. Trends Biochem Sci 30:583–592
Hawkins CL, Davies MJ (2001) Generation and propagation of radical reactions on proteins. Biochim Biophys Acta 1504:196–219
Maleknia SD, Brenowitz M, Chance MR (1999) Millisecond radiolytic modification of peptides by synchrotron X-rays identified by mass spectrometry. Anal Chem 71:3965–3973
Sharp JS, Becker JM, Hettich RL (2004) Analysis of protein solvent accessible surfaces by photochemical oxidation and mass spectrometry. Anal Chem 76:672–683
Traverso N, Menini S, Cottalasso D, Odetti P, Marinari UM, Pronzato MA (1997) Mutual interaction between glycation and oxidation during non-enzymatic protein modification. Biochim Biophys Acta 1336:409–418
Rondeau P, Singh NR, Caillens H, Tallet F, Bourdon E (2008) Oxidative stresses induced by glycoxidized human or bovine serum albumin on human monocytes. Free Radic Biol Med 45:799–812
Sobal G, Menzel J, Sinzinger H (2000) Why is glycated LDL more sensitive to oxidation than native LDL? A comparative study. Prostaglandins Leukot Essent Fatty Acids 63:177–186
Chetyrkin SV, Mathis ME, Ham AJ, Hachey DL, Hudson BG, Voziyan PA (2008) Propagation of protein glycation damage involves modification of tryptophan residues via reactive oxygen species: inhibition by pyridoxamine. Free Radic Biol Med 44:1276–1285
Lapolla A, Fedele D, Seraglia R, Traldi P (2006) The role of mass spectrometry in the study of non-enzymatic protein glycation in diabetes: an update. Mass Spectrom Rev 25:775–797
Selvaraj N, Bobby Z, Sridhar MG (2008) Oxidative stress: does it play a role in the genesis of early glycated proteins? Med Hypotheses 70:265–268
Sen S, Kar M, Roy A, Chakraborti AS (2005) Effect of nonenzymatic glycation on functional and structural properties of hemoglobin. Biophys Chem 113:289–298
Thornalley PJ, Battah S, Ahmed N, Karachalias N, Agalou S, Babaei-Jadidi R, Dawnay A (2003) Quantitative screening of advanced glycation endproducts in cellular and extracellular proteins by tandem mass spectrometry. Biochem J 375:581–592
Traldi P, Favretto D, Seraglia R, Lapolla A (1997) Mass spectrometry in the study of non-enzymatic glyco-oxidation of proteins. Rapid Commun Mass Spectrom 11:673–678
Zeng J, Dunlop RA, Rodgers KJ, Davies MJ (2006) Evidence for inactivation of cysteine proteases by reactive carbonyls via glycation of active site thiols. Biochem J 398:197–206
Morgan PE, Dean RT, Davies MJ (2002) Inactivation of cellular enzymes by carbonyls and protein-bound glycation/glycoxidation products. Arch Biochem Biophys 403:259–269
Acknowledgments
The authors express their appreciation for the financial support provided by the “Fundação para a Ciência e Tecnologia” (grants SFRH/BD/14963/2004 and PTDC/QUI/68408/2006).
Author information
Authors and Affiliations
Corresponding author
Supplementary electronic material
Below is the link to the electronic supplementary material.
ESM Table 2
(DOC 448 kb)
Rights and permissions
About this article
Cite this article
Guedes, S., Vitorino, R., Domingues, M.R.M. et al. Oxidative modifications in glycated insulin. Anal Bioanal Chem 397, 1985–1995 (2010). https://doi.org/10.1007/s00216-010-3757-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-010-3757-x